-
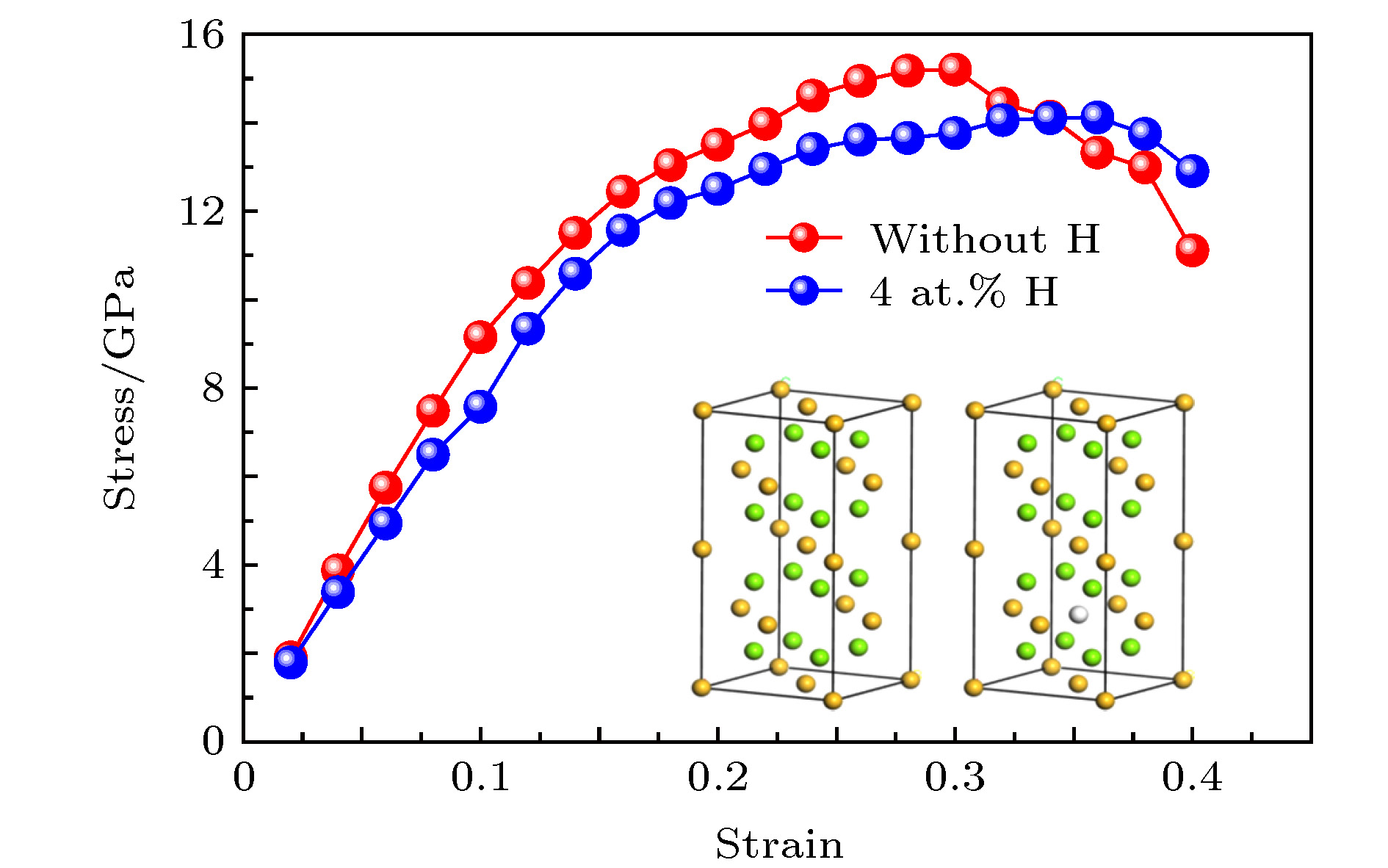
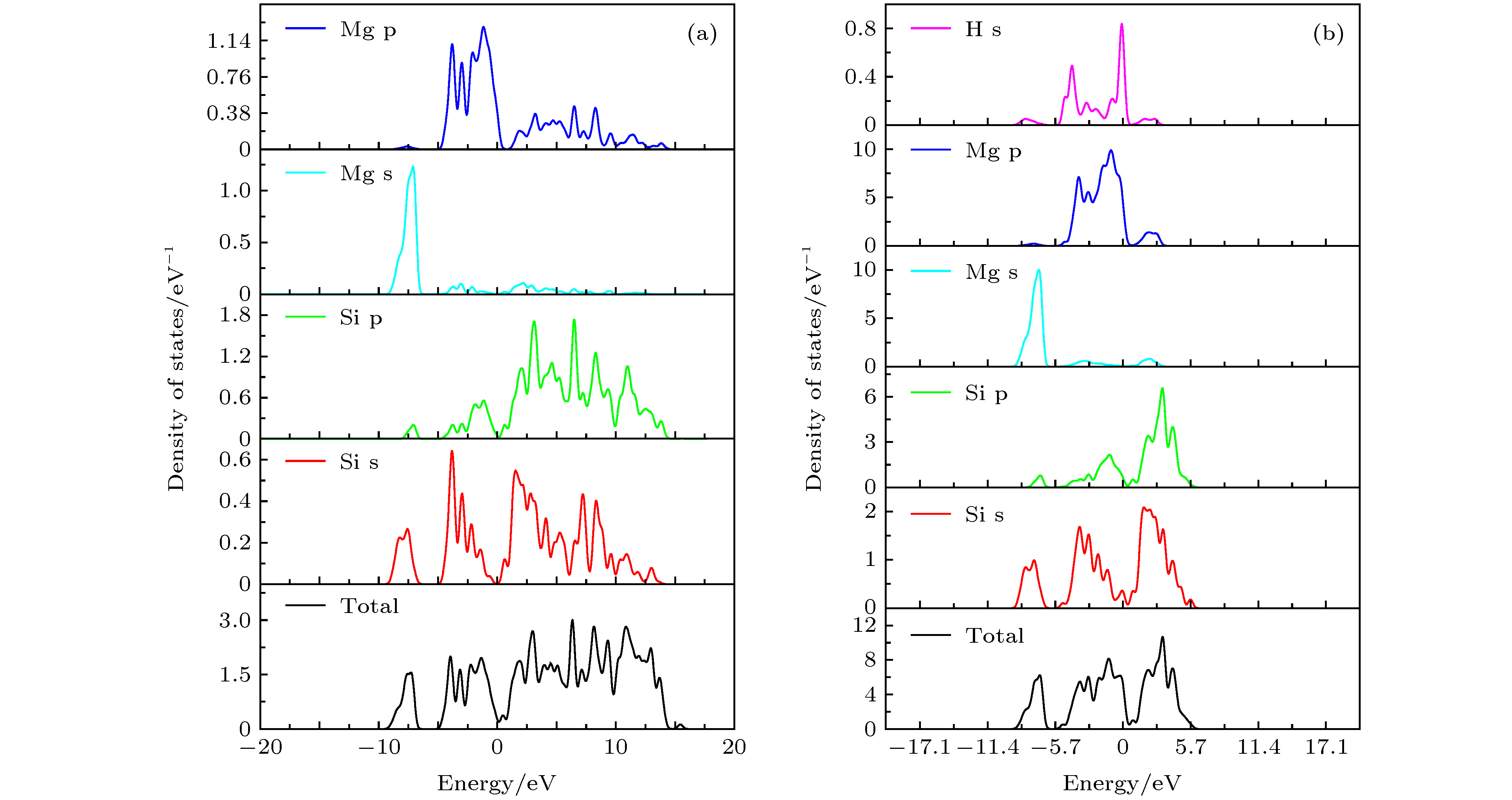
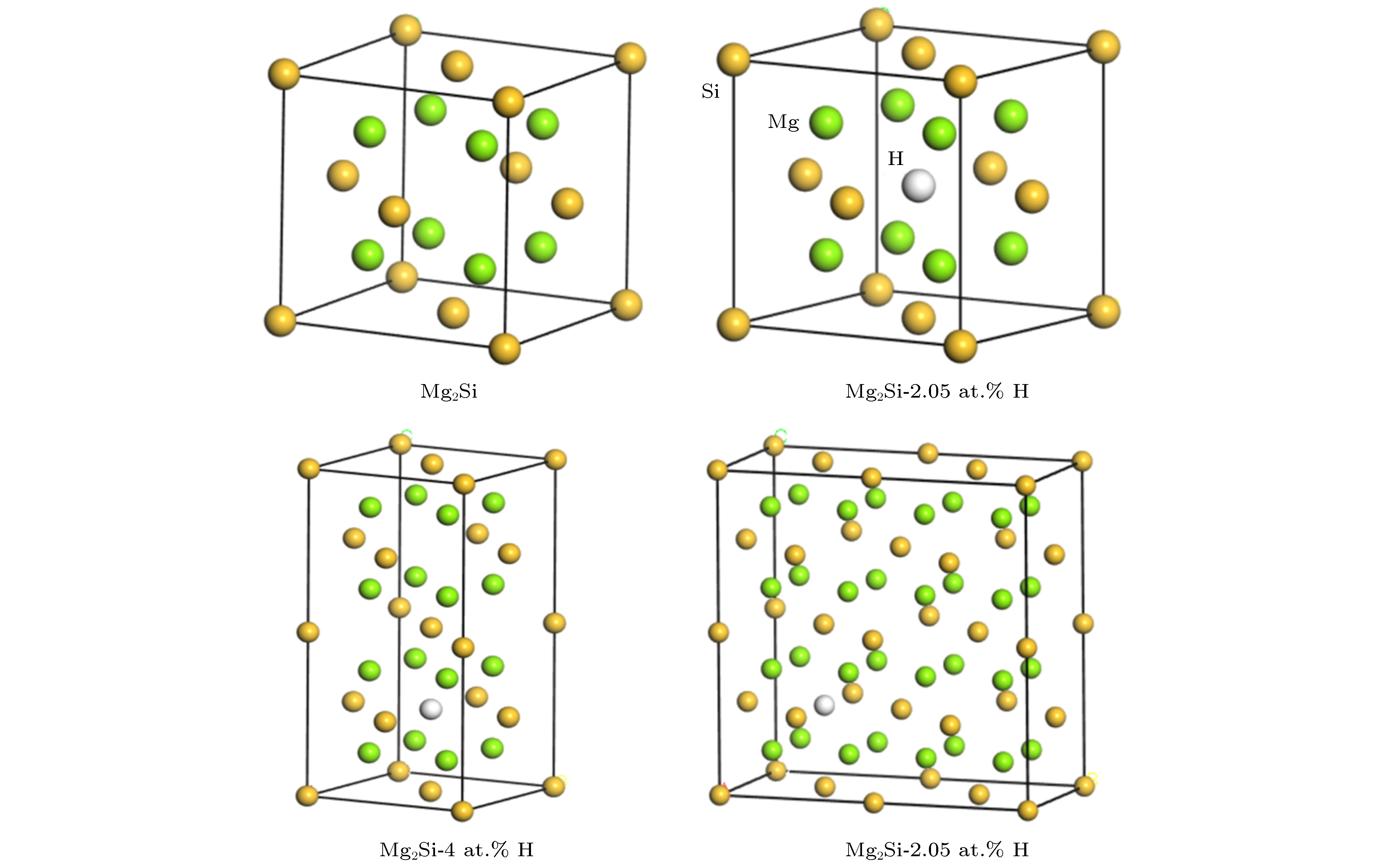
The mechanical properties of Al-Mg-Si-type aluminum alloys may degenerate due to the hydrogen damage during servicing in hydrogen environment. The Mg2Si is the main strengthening phases in Al-Mg-Si-type aluminum alloys. Therefore, the mechanical properties of Mg2Si directly determine the strengths of Al-Mg-Si-type aluminum alloys. In this work, the effects of hydrogen atoms on the mechanical properties of Mg2Si are investigated by first principle calculation, which is based on the density function theory. First of all, we calculate the single crystal elasticity constants of C11, C12 and C44. Then the elasticity modulus, Poisson’s ratio and hardness of polycrystalline are calculated by using the crystal elasticity constants. Furthermore, we also calculate the tensile properties of Mg2Si with and without H atoms. The difference between the densities of states with and without H atoms is used to investigate the change of Mg2Si induced by H atoms. The results show that hydrogen atoms significantly reduce the shear modulus and elastic modulus of Mg2Si, resulting in the strength and hardness decreasing, but the toughness increasing. The calculations of tensile properties indicate that H atoms reduce the fracture strength but enhance the fracture elongation of Mg2Si. The analysis of density of states indicates that hydrogen atoms will induce the properties of Mg2Si to transform from semiconductor to metal properties. The calculated results in this paper can provide a reference basis for revealing the mechanism of strength reduction of Mg2Si materials in a hydrogen environment.
-
Keywords:
- the first principles /
- Mg2Si /
- elastic properties /
- density of states
[1] 李建国, 谭红艳, 史子木, 何迁 2008 中国有色金属学报 18 1819
 Google Scholar
Google Scholar
Li J G, Tan H Y, Shi Z M, He Q 2008 Chin. J. Nonferrous. Met. 18 1819
 Google Scholar
Google Scholar
[2] Zeng F L, Wei Z L, Li J F, Li C X, Tan X, Zhang Z, Zheng Z Q 2011 T. Nonferr. Metal. Soc. 21 2559
 Google Scholar
Google Scholar
[3] Qin Q D, Li W X, Zhao K W, Qiu S L, Zhao Y G 2010 Mat. Sci. Eng. A 527 2253
 Google Scholar
Google Scholar
[4] Tong X, Zhang D, Wang K, Lin J, Liu Y, Shi Z, Li Y, Lin J, Wen C 2018 Mat. Sci. Eng. A 733 9
 Google Scholar
Google Scholar
[5] 任玉艳, 刘桐宇, 李英民 2016 中国科学: 物理学 力学 天文学 46 084611
Ren Y Y, Liu T Y, Li Y M 2016 Sci. Sin.: Phys. Mech. Astron. 46 084611
[6] 余本海, 刘墨林, 陈东 2011 物理学报 60 087105
 Google Scholar
Google Scholar
Yu B H, Liu M L, Chen D 2011 Acta Phys. Sin. 60 087105
 Google Scholar
Google Scholar
[7] 韩秀丽, 王清, 孙东立, 张红星 2008 中国有色金属学报 18 523
 Google Scholar
Google Scholar
Han S L, Wang Q, Sun L D, Zhang H X 2008 Chin. J. Nonferrous. Met. 18 523
 Google Scholar
Google Scholar
[8] 张凤春, 李春福, 文平, 罗强, 冉曾令 2014 物理学报 66 227101
 Google Scholar
Google Scholar
Zhang F C, Li C F, Wen P, Rang Z L 2014 Acta Phys. Sin. 66 227101
 Google Scholar
Google Scholar
[9] 马明光, 亢世江, 张红玲, 徐红彬 2015 热加工工艺 44 96
Ma G M, Kang S J, Zhang H L, Xu H B 2015 Hot Working Technology 44 96
[10] 姚宝殿, 胡桂青, 于治水 2016 物理学报 65 026202
 Google Scholar
Google Scholar
Yao B D, Hu G Q, Yu Z S 2016 Acta Phys. Sin. 65 026202
 Google Scholar
Google Scholar
[11] 韩秀丽 2010 博士学位论文 (哈尔滨: 哈尔滨工业大学)
Han X L 2010 Ph. D. Dissertation (Harbin: Harbin Institute of Technology) (in Chinese)
[12] 饶建平, 欧阳楚英, 雷敏生 2012 物理学报 61 047105
 Google Scholar
Google Scholar
Rao J P, Ouyang C Y, Lei M S 2012 Acta Phys. Sin. 61 047105
 Google Scholar
Google Scholar
[13] 石瑜, 白洋, 莫丽玢, 向青云, 黄亚丽, 曹江利 2015 物理学报 64 116301
 Google Scholar
Google Scholar
Shi Y, Bai Y, Mo L B, Xiang Q Y, Huang Y L, Cao J L 2015 Acta Phys. Sin. 64 116301
 Google Scholar
Google Scholar
[14] 刘娜娜, 孙翰英, 刘洪生 2009 材料导报: 纳米与新材料专辑 23 278
Liu N N, Sun H Y, Liu H S 2009 Mat. Rev. 23 278
[15] Li L Y, Yu W, Jin C Q 2005 J. Phys.: Condens. Mat. 17 5965
 Google Scholar
Google Scholar
[16] Hill R 2002 Proc. Phys. Soc. 65 349
[17] Jang S H, Chichibu S F 2012 J. Appl. Phys. 112 073503
 Google Scholar
Google Scholar
[18] Yu W Y, Wang N, Xiao X B 2009 Solid State Sci. 11 1400
 Google Scholar
Google Scholar
[19] Li Y, Gao Y, Xiao B 2010 J. Alloy Compd. 502 28
 Google Scholar
Google Scholar
[20] Bystricky J, LaFrance P, Lehar F, Perrot F, Winternitz P 1985 Phys. Rev. D 32 575
[21] Parks G S 1973 J. Chem. Educ. 30 82A
[22] Sung C M, Sung M 1996 Mater. Chem. Phys. 43 1
 Google Scholar
Google Scholar
[23] Leger J M, Djemia P, Ganot F 2001 Appl. Phys. Lett. 79 2169
 Google Scholar
Google Scholar
[24] Miao N, Sa B, Zhou J 2011 Comp. Mater. Sci. 50 1559
 Google Scholar
Google Scholar
[25] Senkov O N, Dubois M, Jonas J J 1996 Metall. Mater. Trans. A 27 3963
 Google Scholar
Google Scholar
[26] Maziyar A, Ahad M, Herbert D, Christian G M 2015 Metall. Mater. Trans. B 46 1471
 Google Scholar
Google Scholar
-
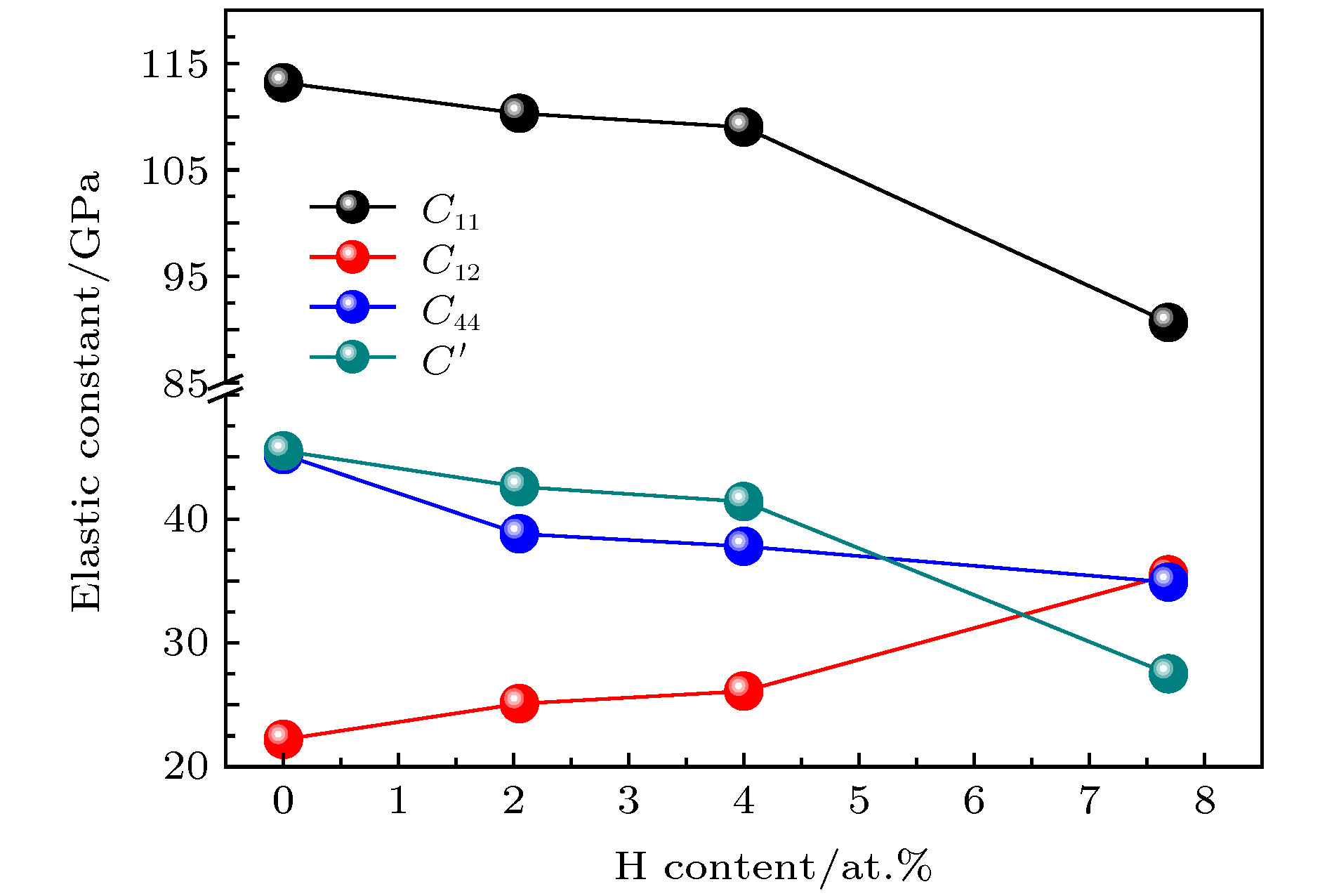
表 1 Mg2Si-H体系晶体弹常数计算结果
Table 1. Elastic constant of Mg2Si-H system.
Phase Source Elastic constants/GPa C11 C12 C44 C' C12–C44 Mg2Si This work 113.2 22.2 45.2 45.5 –23.0 Calculated [20] 113.7 22.8 43.5 Calculated [6] 114.5 21.5 45.6 Experimental [21] 126.0 26.0 48.5 Mg2Si-2.05 at.%H This work 110.3 25.1 38.8 42.6 –13.7 Mg2Si-4 at.%H This work 109.0 26.1 37.8 41.4 –11.7 Mg2Si-7.69 at.%H This work 90.6 35.5 34.9 27.5 0.6 表 2 Mg2Si-H体系模量
Table 2. Modulus of Mg2Si-H system.
Phase Source Modulus GH/GPa BH/GPa E/GPa ν BH/GH H/GPa Mg2Si This work 45.3 52.5 105.6 0.16 1.16 10.31 Calculated [20] 46.2 52.5 107.1 0.16 — — Calculated [6] 44.3 53.4 104.1 0.17 — — Experimental [21] — 59.0 120.0 — — — Mg2Si-2.05 at.%H This work 40.2 53.4 96.5 0.17 1.32 9.07 Mg2Si-4 at.%H This work 39.2 53.7 94.7 0.18 1.37 8.56 Mg2Si-7.69 at.%H This work 31.7 53.8 79.5 0.28 1.69 4.55 -
[1] 李建国, 谭红艳, 史子木, 何迁 2008 中国有色金属学报 18 1819
 Google Scholar
Google Scholar
Li J G, Tan H Y, Shi Z M, He Q 2008 Chin. J. Nonferrous. Met. 18 1819
 Google Scholar
Google Scholar
[2] Zeng F L, Wei Z L, Li J F, Li C X, Tan X, Zhang Z, Zheng Z Q 2011 T. Nonferr. Metal. Soc. 21 2559
 Google Scholar
Google Scholar
[3] Qin Q D, Li W X, Zhao K W, Qiu S L, Zhao Y G 2010 Mat. Sci. Eng. A 527 2253
 Google Scholar
Google Scholar
[4] Tong X, Zhang D, Wang K, Lin J, Liu Y, Shi Z, Li Y, Lin J, Wen C 2018 Mat. Sci. Eng. A 733 9
 Google Scholar
Google Scholar
[5] 任玉艳, 刘桐宇, 李英民 2016 中国科学: 物理学 力学 天文学 46 084611
Ren Y Y, Liu T Y, Li Y M 2016 Sci. Sin.: Phys. Mech. Astron. 46 084611
[6] 余本海, 刘墨林, 陈东 2011 物理学报 60 087105
 Google Scholar
Google Scholar
Yu B H, Liu M L, Chen D 2011 Acta Phys. Sin. 60 087105
 Google Scholar
Google Scholar
[7] 韩秀丽, 王清, 孙东立, 张红星 2008 中国有色金属学报 18 523
 Google Scholar
Google Scholar
Han S L, Wang Q, Sun L D, Zhang H X 2008 Chin. J. Nonferrous. Met. 18 523
 Google Scholar
Google Scholar
[8] 张凤春, 李春福, 文平, 罗强, 冉曾令 2014 物理学报 66 227101
 Google Scholar
Google Scholar
Zhang F C, Li C F, Wen P, Rang Z L 2014 Acta Phys. Sin. 66 227101
 Google Scholar
Google Scholar
[9] 马明光, 亢世江, 张红玲, 徐红彬 2015 热加工工艺 44 96
Ma G M, Kang S J, Zhang H L, Xu H B 2015 Hot Working Technology 44 96
[10] 姚宝殿, 胡桂青, 于治水 2016 物理学报 65 026202
 Google Scholar
Google Scholar
Yao B D, Hu G Q, Yu Z S 2016 Acta Phys. Sin. 65 026202
 Google Scholar
Google Scholar
[11] 韩秀丽 2010 博士学位论文 (哈尔滨: 哈尔滨工业大学)
Han X L 2010 Ph. D. Dissertation (Harbin: Harbin Institute of Technology) (in Chinese)
[12] 饶建平, 欧阳楚英, 雷敏生 2012 物理学报 61 047105
 Google Scholar
Google Scholar
Rao J P, Ouyang C Y, Lei M S 2012 Acta Phys. Sin. 61 047105
 Google Scholar
Google Scholar
[13] 石瑜, 白洋, 莫丽玢, 向青云, 黄亚丽, 曹江利 2015 物理学报 64 116301
 Google Scholar
Google Scholar
Shi Y, Bai Y, Mo L B, Xiang Q Y, Huang Y L, Cao J L 2015 Acta Phys. Sin. 64 116301
 Google Scholar
Google Scholar
[14] 刘娜娜, 孙翰英, 刘洪生 2009 材料导报: 纳米与新材料专辑 23 278
Liu N N, Sun H Y, Liu H S 2009 Mat. Rev. 23 278
[15] Li L Y, Yu W, Jin C Q 2005 J. Phys.: Condens. Mat. 17 5965
 Google Scholar
Google Scholar
[16] Hill R 2002 Proc. Phys. Soc. 65 349
[17] Jang S H, Chichibu S F 2012 J. Appl. Phys. 112 073503
 Google Scholar
Google Scholar
[18] Yu W Y, Wang N, Xiao X B 2009 Solid State Sci. 11 1400
 Google Scholar
Google Scholar
[19] Li Y, Gao Y, Xiao B 2010 J. Alloy Compd. 502 28
 Google Scholar
Google Scholar
[20] Bystricky J, LaFrance P, Lehar F, Perrot F, Winternitz P 1985 Phys. Rev. D 32 575
[21] Parks G S 1973 J. Chem. Educ. 30 82A
[22] Sung C M, Sung M 1996 Mater. Chem. Phys. 43 1
 Google Scholar
Google Scholar
[23] Leger J M, Djemia P, Ganot F 2001 Appl. Phys. Lett. 79 2169
 Google Scholar
Google Scholar
[24] Miao N, Sa B, Zhou J 2011 Comp. Mater. Sci. 50 1559
 Google Scholar
Google Scholar
[25] Senkov O N, Dubois M, Jonas J J 1996 Metall. Mater. Trans. A 27 3963
 Google Scholar
Google Scholar
[26] Maziyar A, Ahad M, Herbert D, Christian G M 2015 Metall. Mater. Trans. B 46 1471
 Google Scholar
Google Scholar
Catalog
Metrics
- Abstract views: 16731
- PDF Downloads: 121
- Cited By: 0














 DownLoad:
DownLoad: