-
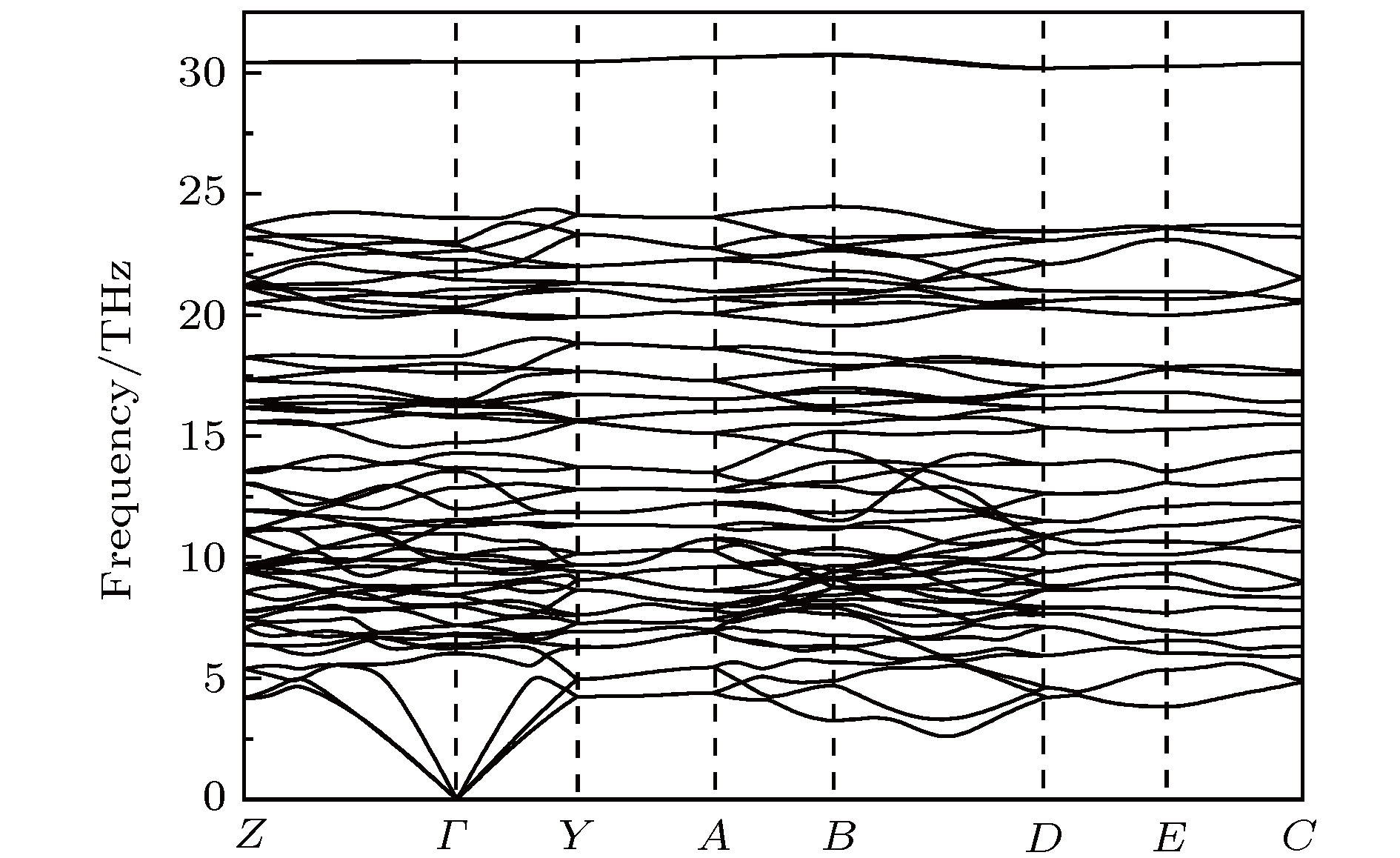
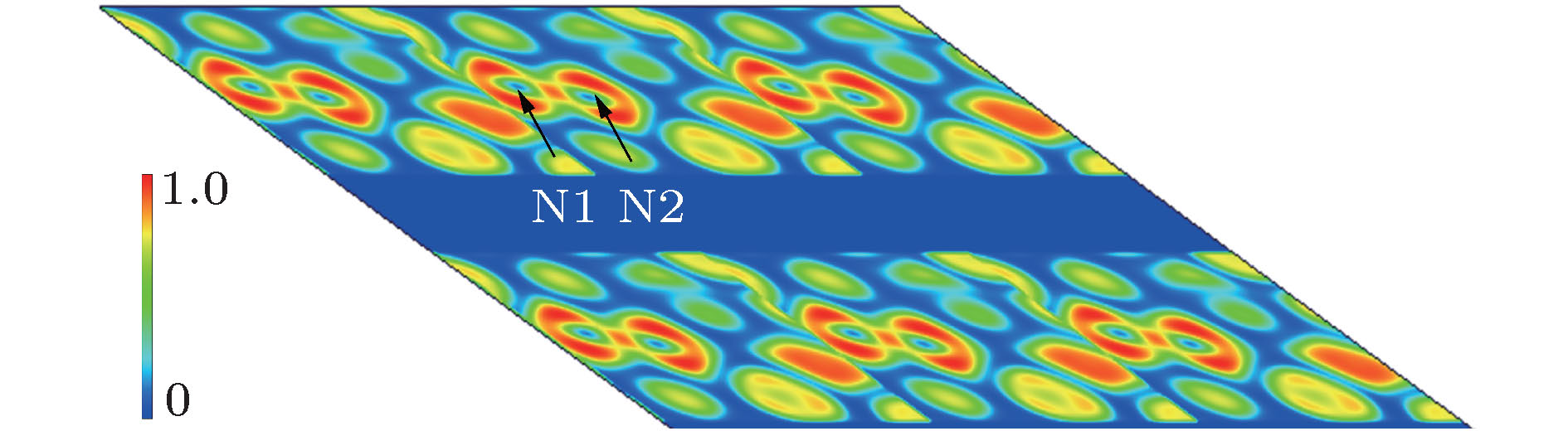
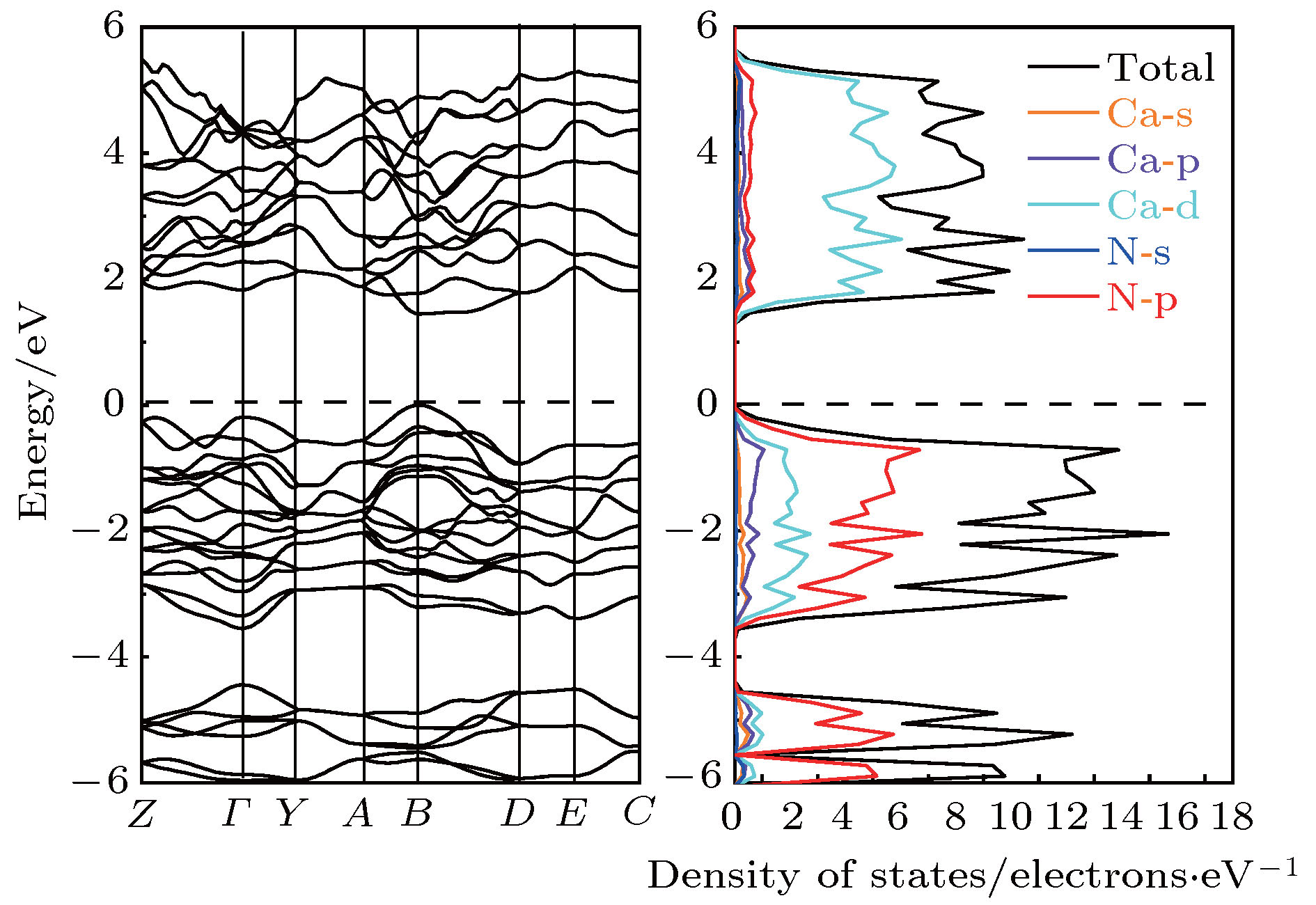
通过在氮中引入杂质离子, 利用高压手段获得具有新奇结构的多氮化合物是目前被广泛应用的研究方法. 钙氮材料在催化、光电方面有着广泛的应用. 具有较低电离能的钙(Ca)元素很容易和氮原子形成离子键钙氮化物. 高压为寻找新型钙氮化合物提供了全新的技术途径. 因此, 利用高压方法, 通过改变配比的方式, 寻找具有新奇特性的钙氮高压结构, 是一项非常有意义的工作. 本文利用基于密度泛函理论的结构搜索方法, 在100 GPa条件下, 通过预测得到了一个稳定的Ca5N4相. 该结构内部氮原子之间以N—N共价单键键合, 氮原子和钙原子之间是离子键相互作用, 且钙氮之间的电荷转移量为1.26 e/N atom. 能带结构计算表明P 21/c-Ca5N4是一个直接带隙为1.447 eV的半导体结构. 最后, 系统地给出了该结构的拉曼振动光谱, 并指认了拉曼振动模式, 为实验合成该结构提供了理论指导.Recent studies have shown that introducing metal elements into nitrogen matrix can induce more stable poly-nitrogen structures than the pure nitrogen phase due to the ionic interaction between metal elements and nitrogen matrix. Many types of poly-nitrogen structures have been reported by using the alkaline earth metal elements (M = Be, Mg, Ca, Sr, Ba) as the coordinate elements. For example, the one-dimensional (1D) infinite armchair poly-nitrogen chain (N∞) structure and N6 ring structure are obtained for the MN4 and MN3 chemical stoichiometry, respectively. Interestingly, the stabilities of theses MNx structures are enhanced 2–3 times compared with that of the pure nitrogen. Therefore, exploring the novel and stable poly-nitrogen structure by introducing alkaline earth metal elements under high pressure is a great significant job. As an alkaline earth element, Ca is abundant in the earth. Its ionization energy (I1 = 590 kJ/mol) is far lower than that of Be (900 kJ/mol) and Mg (738 kJ/mol), which means that Ca can form calcium nitrides more easily. Zhu et al. (Zhu S, Peng F, Liu H, Majumdar A, Gao T, Yao Y 2016Inorg. Chem. 55 7550) proposed that the Ca-N system can obtain poly-nitrogen structures under high pressure, such as CaN4 structure with armchair nitrogen chain, CaN5 and CaN3 consisting of pentazolate “N5” and benzene-like “N6” anions. These poly-nitrogen structures have potential applications in the field of high energy density materials. Here, we report the prediction of Ca-N system at 100 GPa by using particle swarm optimization algorithm technique for crystal structure prediction. A new thermal stable phase with P 21/c-Ca5N4 space group is found at 100 GPa, which enriches the phase of Ca-N system under high pressure. The dynamic stability and mechanical stability of new phase are confirmed by phono dispersion spectrum and elastic constant calculations. The electron localization function analysis shows that the nitrogen atoms in P 21/c-Ca5N4 are bonded by N—N single bond and electron transfer from Ca atom to N atom enables Ca5N4 to serve as an ionic-bonding interaction structure. Band structure calculation shows that the Ca5N4 has a semiconductor structure with a direct band gap of 1.447 eV. The PDOS calculation shows the valence band near Fermi energy is mainly contributed by N_p electrons, while the conduction band is mainly contributed by Ca_d electrons, indicating that electrons are transferred from Ca atom to N atom. Bader calculation shows that each N atom obtains 1.26e from Ca atom in P 21/c-Ca5N4. The Raman spectrum and X-ray diffraction spectrum are calculated and detailed Raman vibration modes are identified, which provides theoretical guidance for experimental synthesis.
-
Keywords:
- high pressure /
- alkaline earth metal /
- calcium nitrides
[1] Pickard C J, Needs R J 2009 Phys. Rev. Lett. 102 125702
 Google Scholar
Google Scholar
[2] Erba A, Maschio L, Pisani C, Casassa S 2011 Phys. Rev. B 84 012101
 Google Scholar
Google Scholar
[3] Hirshberg B, Gerber R B, Krylov A I 2014 Nat. Chem. 6 52
 Google Scholar
Google Scholar
[4] Özçelik V O, Aktürk O Ü, Durgun E, Ciraci S 2015 Phys. Rev. B 92 125420
 Google Scholar
Google Scholar
[5] Plašienka D, Martoňák R 2015 J. Chem. Phys. 142 094505
 Google Scholar
Google Scholar
[6] Yakub L N 2016 Low Temp. Phys. 42 1
 Google Scholar
Google Scholar
[7] Martin R M, Needs R J 1986 Phys. Rev. B 34 5082
 Google Scholar
Google Scholar
[8] Mailhiot C, Yang L H, McMahan A K 1992 Phys. Rev. B 46 14419
 Google Scholar
Google Scholar
[9] Wang X, Wang Y, Miao M, Zhong X, Lv J, Cui T, Li J, Chen L, Pickard C J, Ma Y 2012 Phys. Rev. Lett. 109 175502
 Google Scholar
Google Scholar
[10] Ma Y, Oganov A R, Li Z, Xie Y, Kotakoski J 2009 Phys. Rev. Lett. 102 065501
 Google Scholar
Google Scholar
[11] Bondarchuk S V, Minaev B F 2017 Comput. Mater. Sci. 133 122
 Google Scholar
Google Scholar
[12] Eremets M I, Gavriliuk A G, Trojan I A, Dzivenko D A, Boehler R 2004 Nat. Mater. 3 558
 Google Scholar
Google Scholar
[13] Tomasino D, Kim M, Smith J, Yoo C S 2014 Phys. Rev. Lett. 113 205502
 Google Scholar
Google Scholar
[14] Zhao J F, Li N, Li Q S 2003 Theor. Chem. Acc. 110 10
 Google Scholar
Google Scholar
[15] Steele B A, Oleynik I I 2016 Chem. Phys.Lett. 643 21
 Google Scholar
Google Scholar
[16] Zhang M, Yin K, Zhang X, Wang H, Li Q, Wu Z 2013 Solid State Commun. 161 13
 Google Scholar
Google Scholar
[17] Peng F, Yao Y, Liu H, Ma Y 2015 J. Phys. Chem. Lett. 6 2363
 Google Scholar
Google Scholar
[18] Zhang J, Zeng Z, Lin H Q, Li Y L 2015 Sci. Rep. 4 4358
 Google Scholar
Google Scholar
[19] Wang X, Li J, Zhu H, Chen L, Lin H 2014 J. Chem. Phys. 141 044717
 Google Scholar
Google Scholar
[20] Williams A S, Steele B A, Oleynik I I 2017 J. Chem. Phys. 147 234701
 Google Scholar
Google Scholar
[21] Wei S, Li D, Liu Z, Wang W, Tian F, Bao K, Duan D, Liu B, Cui T 2017 J. Phys. Chem. C 121 9766
 Google Scholar
Google Scholar
[22] Yu S, Huang B, Zeng Q, Oganov A R, Zhang L, Frapper G 2017 J. Phys. Chem. C 121 11037
 Google Scholar
Google Scholar
[23] Wei S, Li D, Liu Z, Li X, Tian F, Duan D, Liu B, Cui T 2017 Phys. Chem. Chem. Phys. 19 9246
 Google Scholar
Google Scholar
[24] Hou P, Lian L, Cai Y, Liu B, Wang B, Wei S, Li D 2018 RSC Adv. 8 4314
 Google Scholar
Google Scholar
[25] Braun C, Börger S L, Boyko T D, Miehe G, Ehrenberg H, Höhn P, Moewes A, Schnick W 2011 J. Am. Chem. Soc. 133 4307
 Google Scholar
Google Scholar
[26] Hao J, Li Y, Wang J, Ma C, Huang L, Liu R, Cui Q, Zou G, Liu J, Li X 2010 J. Phys. Chem. C 114 16750
 Google Scholar
Google Scholar
[27] Römer S R, Schnick W, Kroll P 2009 J. Phys. Chem. C 113 2943
 Google Scholar
Google Scholar
[28] Gregory D H, Bowman A, Baker C F, Weston D P 2000 J. Mater. Chem. 10 1635
 Google Scholar
Google Scholar
[29] Zhu S, Peng F, Liu H, Majumdar A, Gao T, Yao Y 2016 Inorg. Chem. 55 7550
 Google Scholar
Google Scholar
[30] Dong X, Oganov A R, Goncharov A F, Stavrou E, Lobanov S, Saleh G, Qian G R, Zhu Q, Gatti C, Deringer V L, Dronskowski R, Zhou X F, Prakapenka V B, Konôpková Z, Popov I A, Boldyrev A I, Wang H T 2017 Nat. Chem. 9 440
 Google Scholar
Google Scholar
[31] Ma Y, Eremets M, Oganov A R, Xie Y, Trojan I, Medvedev S, Lyakhov A O, Valle M, Prakapenka V 2009 Nature 458 182
 Google Scholar
Google Scholar
[32] Einaga M, Sakata M, Ishikawa T, Shimizu K, Eremets M I, Drozdov A P, Troyan I A, Hirao N, Ohishi Y 2016 Nat. Phys. 12 835
 Google Scholar
Google Scholar
[33] Wang Y, Lv J, Zhu L, Ma Y 2012 Comput. Phys. Commun. 183 2063
 Google Scholar
Google Scholar
[34] Kresse G, Furthmüller J 1996 Phys. Rev. B 54 11169
 Google Scholar
Google Scholar
[35] Chaput L, Togo A, Tanaka I, Hug G 2011 Phys. Rev. B 84 094302
 Google Scholar
Google Scholar
[36] Henkelman G, Arnaldsson A, Jónsson H 2006 Comput. Mater. Sci. 36 354
 Google Scholar
Google Scholar
[37] Perdew J P, Burke K, Ernzerhof M 1996 Phys. Rev. Lett. 77 3865
 Google Scholar
Google Scholar
[38] Hammer B, Hansen L B, Nørskov J K 1999 Phys. Rev. B 59 7413
 Google Scholar
Google Scholar
-
-
[1] Pickard C J, Needs R J 2009 Phys. Rev. Lett. 102 125702
 Google Scholar
Google Scholar
[2] Erba A, Maschio L, Pisani C, Casassa S 2011 Phys. Rev. B 84 012101
 Google Scholar
Google Scholar
[3] Hirshberg B, Gerber R B, Krylov A I 2014 Nat. Chem. 6 52
 Google Scholar
Google Scholar
[4] Özçelik V O, Aktürk O Ü, Durgun E, Ciraci S 2015 Phys. Rev. B 92 125420
 Google Scholar
Google Scholar
[5] Plašienka D, Martoňák R 2015 J. Chem. Phys. 142 094505
 Google Scholar
Google Scholar
[6] Yakub L N 2016 Low Temp. Phys. 42 1
 Google Scholar
Google Scholar
[7] Martin R M, Needs R J 1986 Phys. Rev. B 34 5082
 Google Scholar
Google Scholar
[8] Mailhiot C, Yang L H, McMahan A K 1992 Phys. Rev. B 46 14419
 Google Scholar
Google Scholar
[9] Wang X, Wang Y, Miao M, Zhong X, Lv J, Cui T, Li J, Chen L, Pickard C J, Ma Y 2012 Phys. Rev. Lett. 109 175502
 Google Scholar
Google Scholar
[10] Ma Y, Oganov A R, Li Z, Xie Y, Kotakoski J 2009 Phys. Rev. Lett. 102 065501
 Google Scholar
Google Scholar
[11] Bondarchuk S V, Minaev B F 2017 Comput. Mater. Sci. 133 122
 Google Scholar
Google Scholar
[12] Eremets M I, Gavriliuk A G, Trojan I A, Dzivenko D A, Boehler R 2004 Nat. Mater. 3 558
 Google Scholar
Google Scholar
[13] Tomasino D, Kim M, Smith J, Yoo C S 2014 Phys. Rev. Lett. 113 205502
 Google Scholar
Google Scholar
[14] Zhao J F, Li N, Li Q S 2003 Theor. Chem. Acc. 110 10
 Google Scholar
Google Scholar
[15] Steele B A, Oleynik I I 2016 Chem. Phys.Lett. 643 21
 Google Scholar
Google Scholar
[16] Zhang M, Yin K, Zhang X, Wang H, Li Q, Wu Z 2013 Solid State Commun. 161 13
 Google Scholar
Google Scholar
[17] Peng F, Yao Y, Liu H, Ma Y 2015 J. Phys. Chem. Lett. 6 2363
 Google Scholar
Google Scholar
[18] Zhang J, Zeng Z, Lin H Q, Li Y L 2015 Sci. Rep. 4 4358
 Google Scholar
Google Scholar
[19] Wang X, Li J, Zhu H, Chen L, Lin H 2014 J. Chem. Phys. 141 044717
 Google Scholar
Google Scholar
[20] Williams A S, Steele B A, Oleynik I I 2017 J. Chem. Phys. 147 234701
 Google Scholar
Google Scholar
[21] Wei S, Li D, Liu Z, Wang W, Tian F, Bao K, Duan D, Liu B, Cui T 2017 J. Phys. Chem. C 121 9766
 Google Scholar
Google Scholar
[22] Yu S, Huang B, Zeng Q, Oganov A R, Zhang L, Frapper G 2017 J. Phys. Chem. C 121 11037
 Google Scholar
Google Scholar
[23] Wei S, Li D, Liu Z, Li X, Tian F, Duan D, Liu B, Cui T 2017 Phys. Chem. Chem. Phys. 19 9246
 Google Scholar
Google Scholar
[24] Hou P, Lian L, Cai Y, Liu B, Wang B, Wei S, Li D 2018 RSC Adv. 8 4314
 Google Scholar
Google Scholar
[25] Braun C, Börger S L, Boyko T D, Miehe G, Ehrenberg H, Höhn P, Moewes A, Schnick W 2011 J. Am. Chem. Soc. 133 4307
 Google Scholar
Google Scholar
[26] Hao J, Li Y, Wang J, Ma C, Huang L, Liu R, Cui Q, Zou G, Liu J, Li X 2010 J. Phys. Chem. C 114 16750
 Google Scholar
Google Scholar
[27] Römer S R, Schnick W, Kroll P 2009 J. Phys. Chem. C 113 2943
 Google Scholar
Google Scholar
[28] Gregory D H, Bowman A, Baker C F, Weston D P 2000 J. Mater. Chem. 10 1635
 Google Scholar
Google Scholar
[29] Zhu S, Peng F, Liu H, Majumdar A, Gao T, Yao Y 2016 Inorg. Chem. 55 7550
 Google Scholar
Google Scholar
[30] Dong X, Oganov A R, Goncharov A F, Stavrou E, Lobanov S, Saleh G, Qian G R, Zhu Q, Gatti C, Deringer V L, Dronskowski R, Zhou X F, Prakapenka V B, Konôpková Z, Popov I A, Boldyrev A I, Wang H T 2017 Nat. Chem. 9 440
 Google Scholar
Google Scholar
[31] Ma Y, Eremets M, Oganov A R, Xie Y, Trojan I, Medvedev S, Lyakhov A O, Valle M, Prakapenka V 2009 Nature 458 182
 Google Scholar
Google Scholar
[32] Einaga M, Sakata M, Ishikawa T, Shimizu K, Eremets M I, Drozdov A P, Troyan I A, Hirao N, Ohishi Y 2016 Nat. Phys. 12 835
 Google Scholar
Google Scholar
[33] Wang Y, Lv J, Zhu L, Ma Y 2012 Comput. Phys. Commun. 183 2063
 Google Scholar
Google Scholar
[34] Kresse G, Furthmüller J 1996 Phys. Rev. B 54 11169
 Google Scholar
Google Scholar
[35] Chaput L, Togo A, Tanaka I, Hug G 2011 Phys. Rev. B 84 094302
 Google Scholar
Google Scholar
[36] Henkelman G, Arnaldsson A, Jónsson H 2006 Comput. Mater. Sci. 36 354
 Google Scholar
Google Scholar
[37] Perdew J P, Burke K, Ernzerhof M 1996 Phys. Rev. Lett. 77 3865
 Google Scholar
Google Scholar
[38] Hammer B, Hansen L B, Nørskov J K 1999 Phys. Rev. B 59 7413
 Google Scholar
Google Scholar
计量
- 文章访问数: 15689
- PDF下载量: 327
- 被引次数: 0














 下载:
下载: