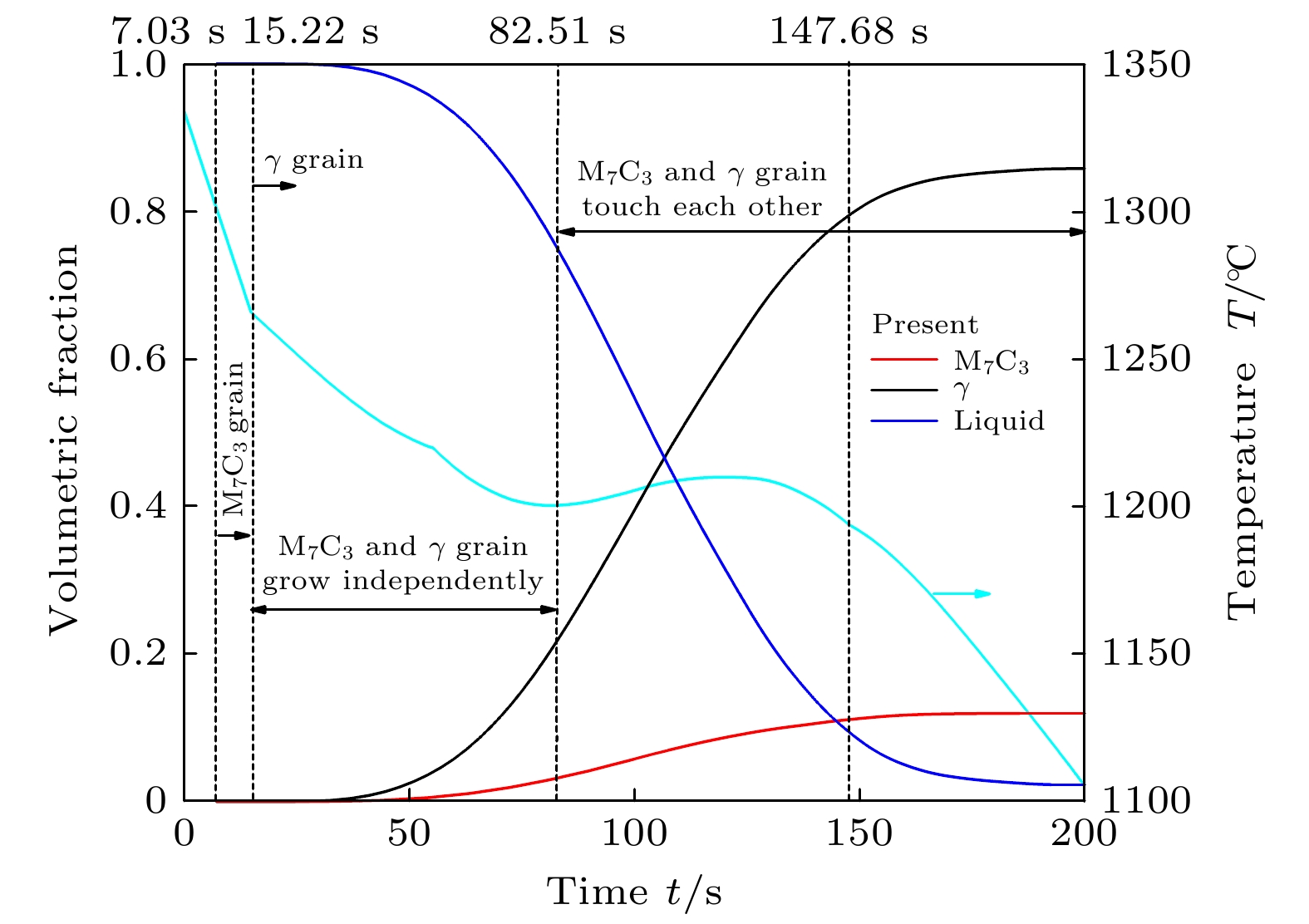
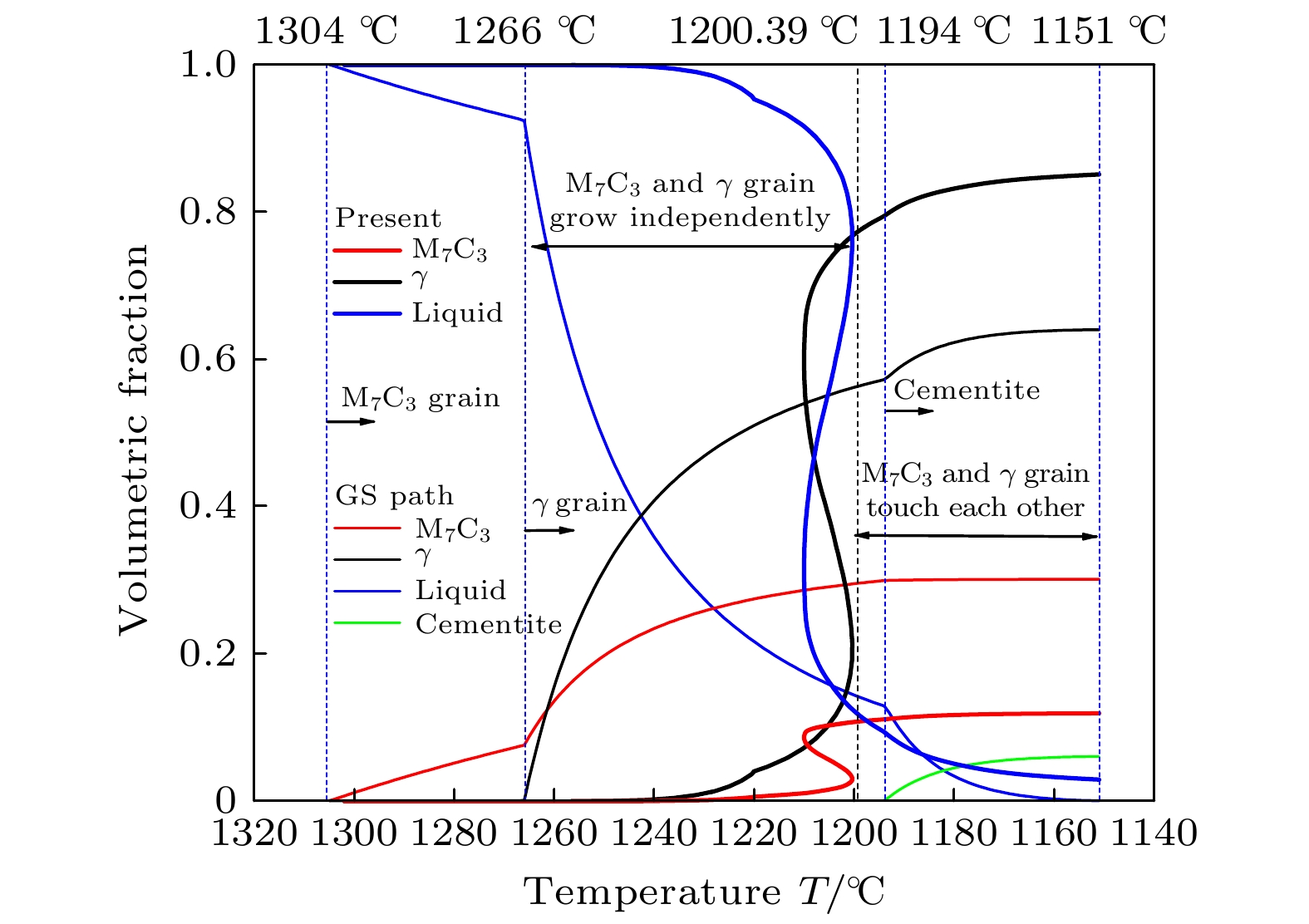
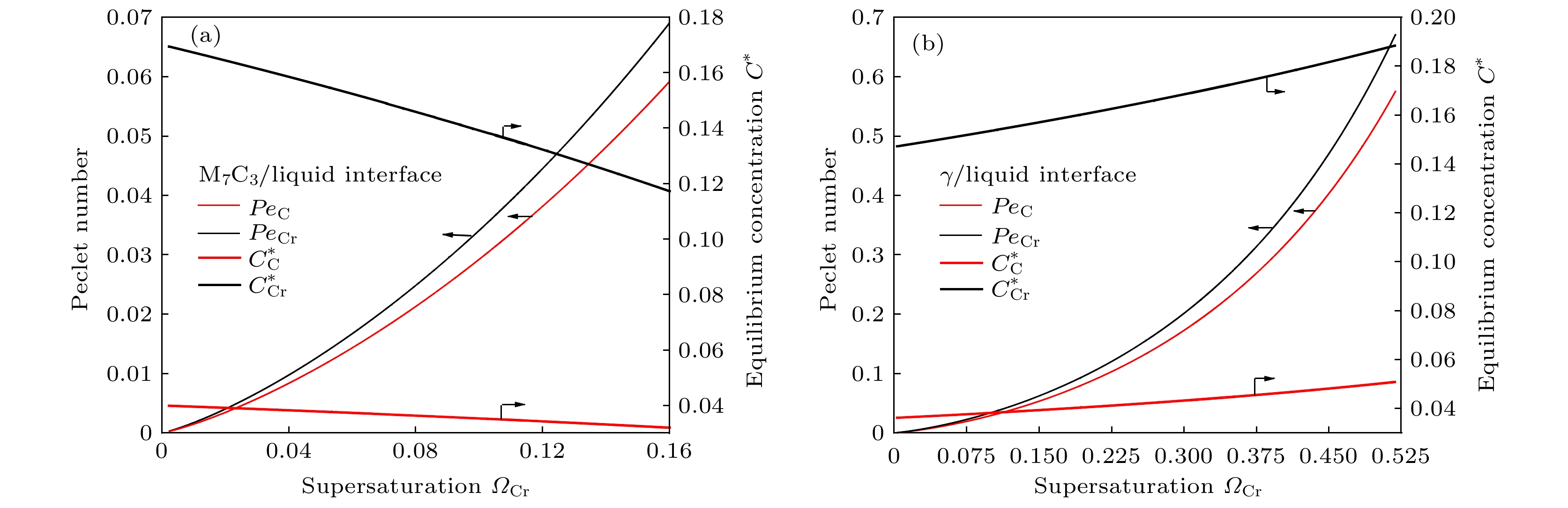
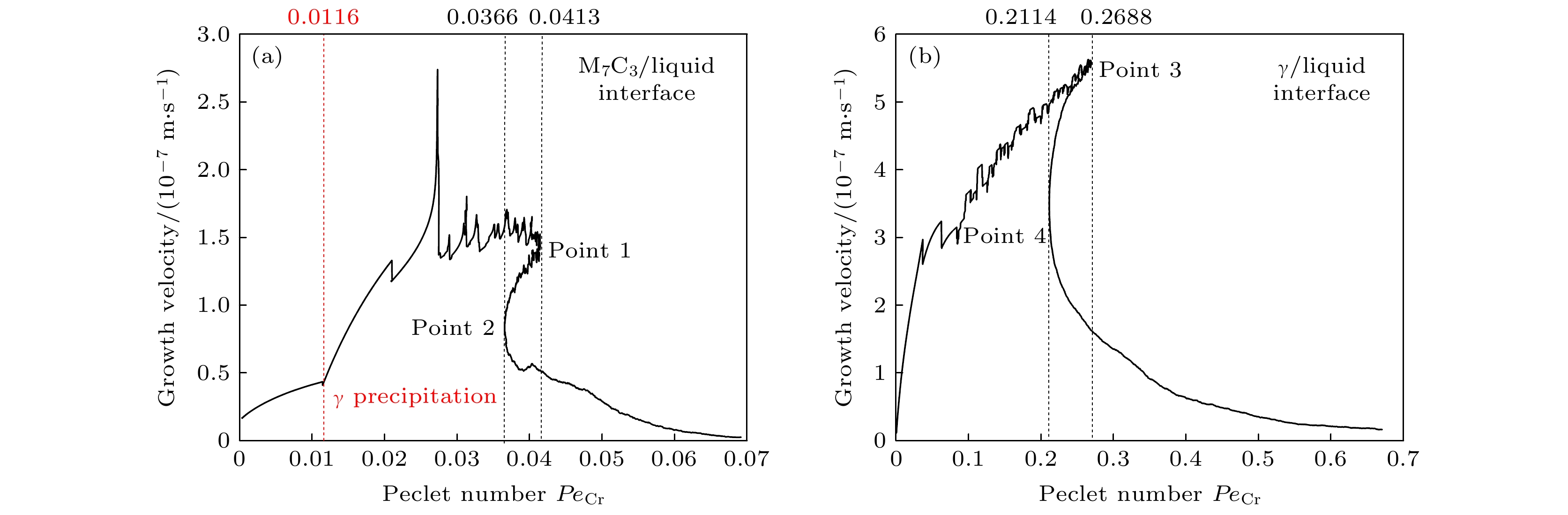
-
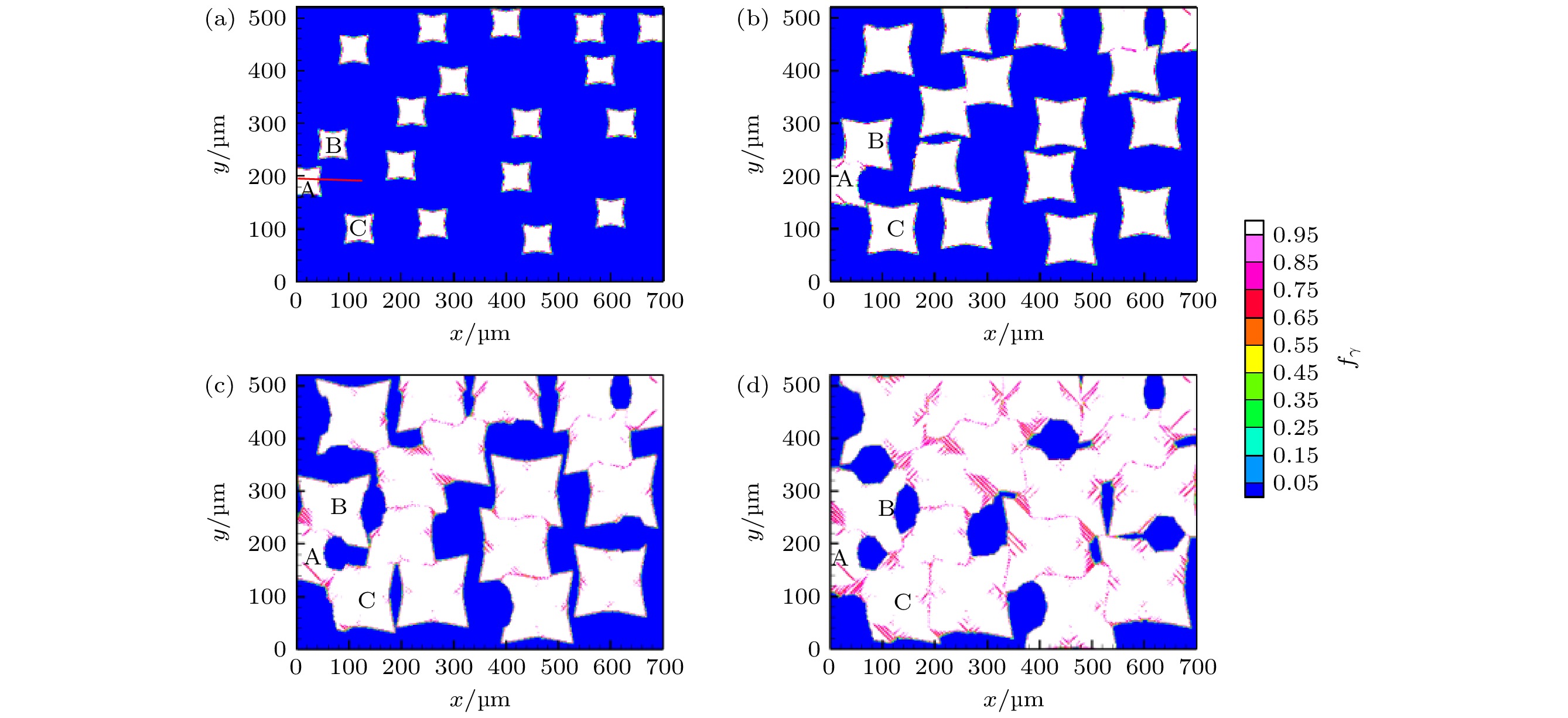
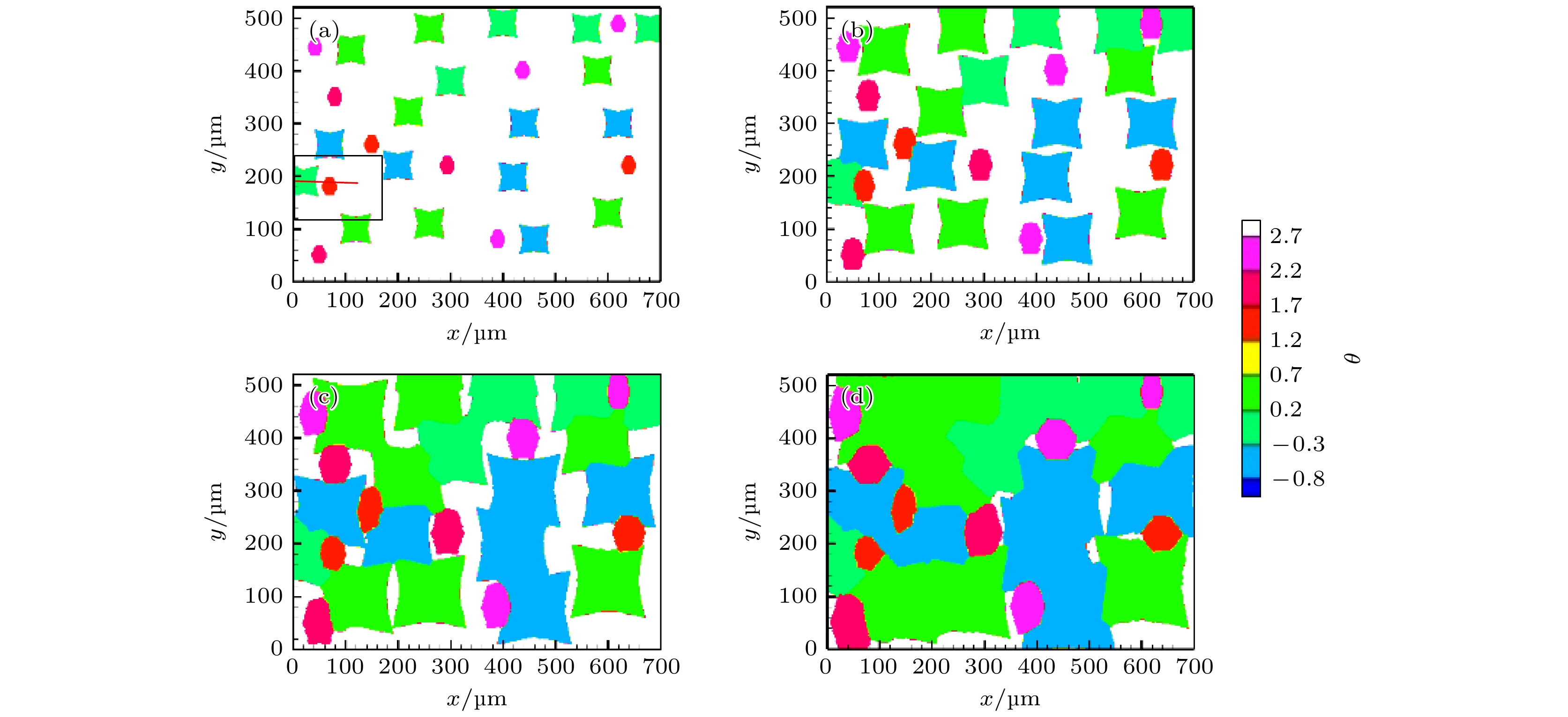
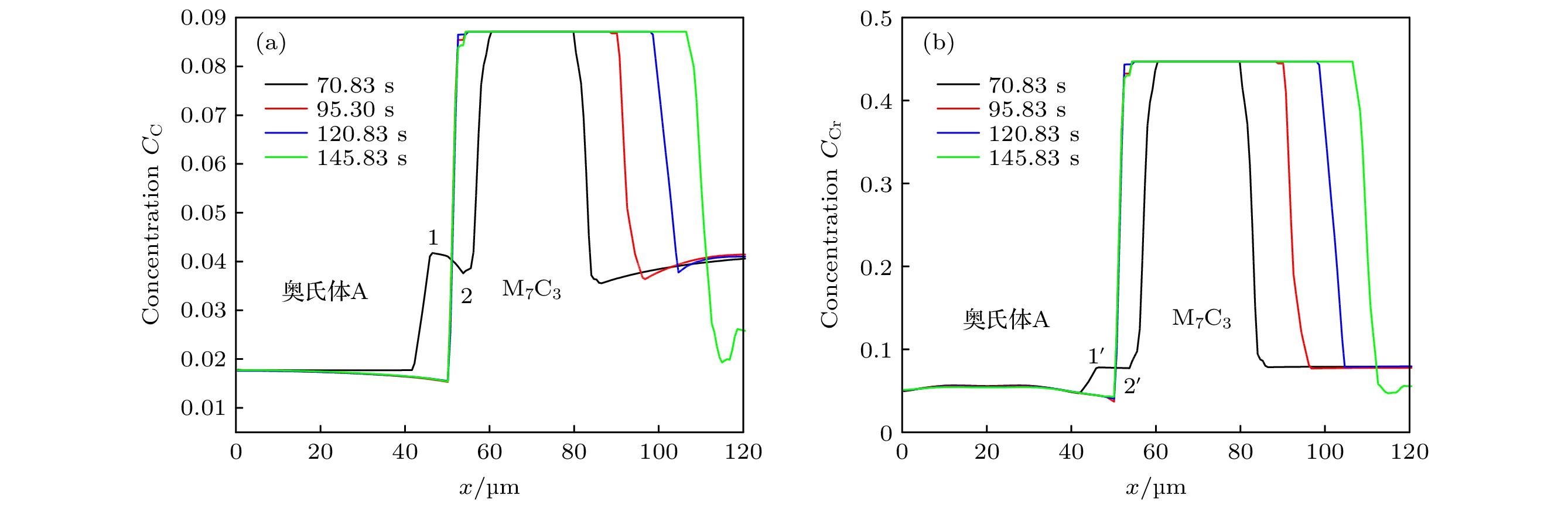
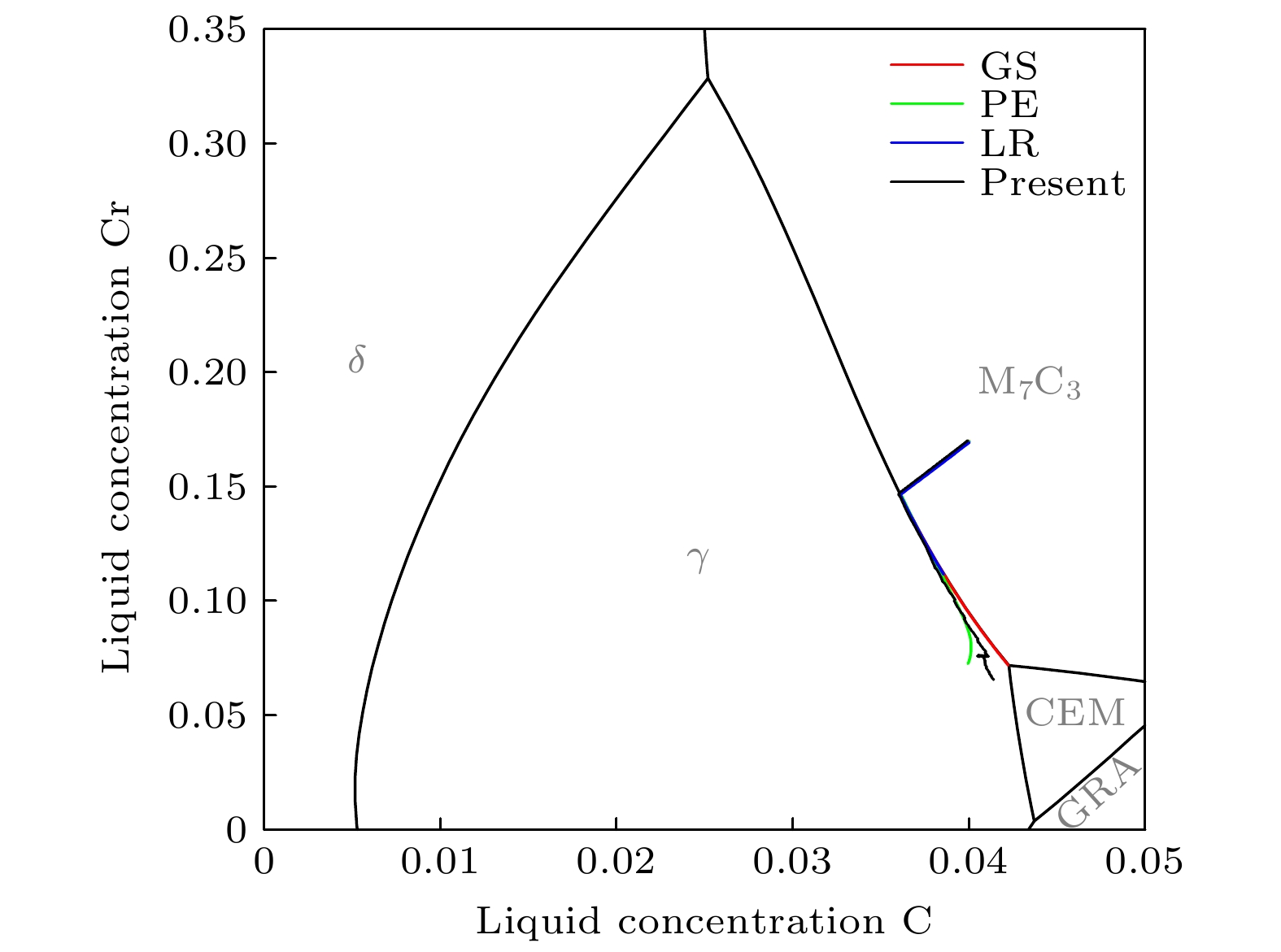
高铬铸铁中M7C3碳化物大小适中、弥散均匀分布, 有利于提高合金的耐磨性. 为分析凝固过程中M7C3碳化物晶粒在基体中的形貌及分布、M7C3碳化物与奥氏体晶粒生长的相互作用、引起的溶质偏聚对最终M7C3碳化物粒径分布的影响, 本文开发了Fe-C-Cr三元合金小面晶M7C3碳化物与奥氏体晶粒共生长的二维微观元胞自动机模型, 模型中加入潜热释放对凝固过程温度场的影响, 由C, Cr两溶质界面扩散共同确定晶体生长速度, 由凝固路径数据表插值获取液相元胞的溶质平衡浓度, 设定M7C3碳化物邻胞结构并优化形状因子来保持M7C3碳化物小面晶形貌, 模拟了Fe-4%C-17%Cr三元合金(C和Cr的质量分数分别为4%和17%)初生M7C3碳化物和共晶奥氏体晶粒的生长演变过程. 研究表明, M7C3碳化物和奥氏体晶粒各自的生长速度随着界面液相中C, Cr溶质的超饱和度和贝克列数的增大而增大; 随着奥氏体的析出和晶粒生长, M7C3碳化物晶粒的生长速度明显增快; 当奥氏体晶粒逐渐接触并包围M7C3碳化物晶粒时, 两相晶粒生长速度逐渐降低. 凝固过程中, 奥氏体晶粒生长向外排出C, Cr溶质, 与吸收C, Cr溶质生长的M7C3碳化物晶粒互补, 致使二者生长互相促进, 最终奥氏体晶粒包围M7C3碳化物晶粒生长. 预测的冷却曲线与实验冷却曲线变化趋势相符; 最终凝固组织形貌和M7C3碳化物体积分数与实验相符; 剩余液相、奥氏体中C, Cr溶质浓度演变也与Gulliver-Scheil, Partial Equilibrium, Lever Rule模型预测结果相符.
-
关键词:
- Fe-C-Cr三元合金 /
- M7C3小面晶 /
- 二相共生长 /
- 元胞自动机
M7C3 carbide’s amount, size, morphology and distribution in the microstructure contribute much to the wear resistance of high chromium cast irons. In the present paper, a two-dimensional microscopic cellular automaton model for the growth of the faceted M7C3 carbide together with the austenitic dendrite grains in an Fe-4%C-17%Cr ternary alloy is developed to obtain the evolution of M7C3 carbide grain morphology, the concentration redistribution and their interaction during the growth of M7C3 carbide and austenite grains, and also the total influence on the final M7C3 carbides’ size. The model includes the effect of latent heat release on the temperature drop. The grain growth velocity is determined by both the diffusion of C solute and the diffusion of Cr solute at the S/L interface. The equilibrium concentration in liquid cells is interpolated from the tablulated solidification path which is prescribed by Gulliver-Scheil approximation coupling with the thermodynamic equilibrium calculation. The morphology of the faceted M7C3 carbide is maintained through setting its neighborhood relations and optimizing its shape factor at grain growth. The results show that the individual grain growth velocity for M7C3 carbide and austenite increases with the increase of the supersaturation and Peclet number of solute C and Cr. The austenite precipitation and grain growth obviously speed up the growth velocity of M7C3 carbide grains. While with the austenite grains gradually touching and enveloping the M7C3 carbide grain, the growth velocities for both kinds of grains decrease. The rejection of solute C and Cr during austenite grain growth complements the absorption of solute C and Cr during M7C3 carbide grain growth, thus promoting their growth. The predicted cooling curve fits with the evolution tendency of the experimental one. The predicted final solidification microstructure and M7C3 carbide amount in volume fraction are in agreement with the experimental ones. Furthermore, both C solute concentration distribution and Cr solute concentration distribution in both residual liquid and austenite are consistent with the predictions by the Gulliver-Scheil, partial equilibrium and lever rule model.-
Keywords:
- Fe-C-Cr ternary alloy /
- faceted M7C3 /
- cooperative growth /
- cellular automaton
[1] Siekaniec D, Kopycinski D, Szczesny A, Guzik E, Tyraa E, Nowak A 2017 Nephron Clinical Practice 17 143
 Google Scholar
Google Scholar
[2] Liu H N, Sakamoto M, Nomura M, Ogi K 2001 Wear 250 71
 Google Scholar
Google Scholar
[3] Pokusova M, Gabrisova Z, Brusilova A, Pribulova A, Futa P 2020 Mater. Sci. Forum. 998 30
 Google Scholar
Google Scholar
[4] Filipovic M, Kamberovic Z, Korac M, Gavrilovski M 2013 Mater. Des. 47 41
 Google Scholar
Google Scholar
[5] Buytoz S, Yildirim M M, Eren H 2005 Mater. Lett. 59 607
 Google Scholar
Google Scholar
[6] 龚沛, 赵曜, 杨浩, 王宁 2017 金属热处理 42 137
 Google Scholar
Google Scholar
Gong P, Zhao Y, Yang H, Wang N 2017 Heat Treatment of Metals 42 137
 Google Scholar
Google Scholar
[7] 陈哲, 王亮亮, 张晗, 付永红, 钟黎声, 叶芳霞 2013 热加工工艺 42 8
 Google Scholar
Google Scholar
Chen Z, Wang L L, Zhang H, Fu Y H, Zhong L S, Ye F X 2013 Hot Working Technology 42 8
 Google Scholar
Google Scholar
[8] Tang X H, Chung R, Pang C J, Li D Y, Hinckley B, Dolman K 2011 Wear 271 1426
 Google Scholar
Google Scholar
[9] Zhang H W, Nakajima K, Gandin C A, He J C 2013 ISIJ Int. 53 493
 Google Scholar
Google Scholar
[10] 王同敏, 魏晶晶, 王旭东, 姚曼 2018 金属学报 54 193
 Google Scholar
Google Scholar
Wang T M, Wei J J, Wang X D, Yao M 2018 Acta Metall. Sin. 54 193
 Google Scholar
Google Scholar
[11] 朱鸣芳, 邢丽科, 方辉, 张庆宇, 汤倩玉, 潘诗琰 2018 金属学报 54 789
 Google Scholar
Google Scholar
Zhu M F, Xing L K, Fang H, Zhang Q Y, Tang Q Y, Pan S Y 2018 Acta. Metall. Sin. 54 789
 Google Scholar
Google Scholar
[12] Zhu M F, Cao W, Chen S L, Hong C P, Chang Y A 2007 J. Phase Equilib. Diffus. 28 130
 Google Scholar
Google Scholar
[13] 戴挺, 朱鸣芳, 陈双林, 曹伟生, 洪俊杓 2008 金属学报 10 1175
 Google Scholar
Google Scholar
Dai T, Zhu M F, Chen S L, Cao W S, Hong J Z 2008 Acta Metall. Sin. 10 1175
 Google Scholar
Google Scholar
[14] Zhang H W, Gandin C A, Hamouda H B, Tourret D, Nakajima K, He J C 2010 ISIJ Int. 50 1859
 Google Scholar
Google Scholar
[15] Zhang H W, Gandin C A, Nakajima K, He J C 2012 IOP Conf Series: Materials Science Engineering 33 012063
 Google Scholar
Google Scholar
[16] 石玉峰, 许庆彦, 柳百成 2012 物理学报 61 108101
 Google Scholar
Google Scholar
Shi Y F, Xu Q Y, Liu B C 2012 Acta Phys. Sin. 61 108101
 Google Scholar
Google Scholar
[17] Michelic S C, Thuswaldner J M, Bernhard C 2010 Acta Mater. 58 2738
 Google Scholar
Google Scholar
[18] 张晛韦 2019 硕士学位论文 (沈阳: 东北大学)
Zhang X W 2019 Master's Dissertation (Shenyang: Northeastern University) (in Chinese)
[19] Zhu M F, Hong C P, Stefanescu D M, Chang Y A 2007 Metall. Mater. Trans. B. 38 517
 Google Scholar
Google Scholar
[20] 张蕾, 赵红蕾, 朱鸣芳 2015 金属学报 51 148
 Google Scholar
Google Scholar
Zhang L, Zhao H L, Zhu M F 2015 Acta Metall. Sin. 51 148
 Google Scholar
Google Scholar
[21] Ma S Q, Xing J, He Y, Li Y F, Huang Z F, Liu G Z, Geng Q J 2015 Mater. Chem. Phys. 161 65
 Google Scholar
Google Scholar
[22] Liu Q, Zhang H W, Wang Q, Zhou X K, Jonsson P G, Nakajima K 2012 ISIJ Int. 52 2210
 Google Scholar
Google Scholar
[23] Stefan-Kharicha M, Kharicha A, Zaidat K, Reiss G, Essl W, Goodwin F, Wu M H, Ludwig A, Mugrauer C 2020 J. Cryst. Growth 541 125667
 Google Scholar
Google Scholar
[24] 付振南, 许庆彦, 熊守美 2007 中国有色金属学报 17 1567
 Google Scholar
Google Scholar
Fu Z N, Xu Q Y, Xiong S M 2007 J. Nonferrous Met. China 17 1567
 Google Scholar
Google Scholar
[25] 董祥雷, 邢辉, 陈长乐, 沙莎, 王建元, 金克新 2016 物理学报 65 58
 Google Scholar
Google Scholar
Dong X L, Xing H, Chen C L, Sha S, Wang J Y, Jin K X 2016 Acta Phys. Sin. 65 58
 Google Scholar
Google Scholar
[26] Zhang H W, Zhang S, Wang Y C, Hao Y Z, Miao M, Nakajima K, Lei H, Wang Q, He J C 2020 IOP Conf Series: Materials Science and Engineering 861 012046
 Google Scholar
Google Scholar
[27] Gandin C A 1993 Acta Metallurgica et Materialia 41 345
 Google Scholar
Google Scholar
[28] Flemings M C, Poirier D R, Barone R V, Brody H D 1970 J. Iron Steel Inst. 208 371
[29] Nastac L 1999 Acta Mater. 47 4253
 Google Scholar
Google Scholar
[30] Eggleston J J, Mcfadden G B, Voorhees P W 2001 Phys. D 150 91
 Google Scholar
Google Scholar
[31] Rappaz M, Boettinger W J 1999 Acta Mater. 47 3205
 Google Scholar
Google Scholar
[32] Burbelko A A, Gurgul D, Kaptur K W, Gorny M 2012 Arch. Foundry Eng. 12 11
 Google Scholar
Google Scholar
[33] 孙玉成 2012 博士学位论文 (山东: 山东大学)
Sun Y C 2012 Ph. D. Dissertation (Shandong: Shandong University) (in Chinese)
[34] Wang J, Qi X, Xing X, Shi Z J, Zhou Y F, Yang Q X 2019 Mater. Res. Express 6 0865h3
 Google Scholar
Google Scholar
[35] Liu S, Zhou Y F, Xing X, Wang J B, Ren X J, Yang Q X 2016 Sci. Rep. 6 1
 Google Scholar
Google Scholar
[36] Coronado J J 2011 Wear 270 287
 Google Scholar
Google Scholar
[37] Beltran S L, Stefanescu D M 2004 Metall. Mater. Trans. A. 35 2471
 Google Scholar
Google Scholar
[38] Liu S, Wang Z, Shi Z, Zhou Y F, Yang Q X 2017 J. Alloy. Compd. 713 108
 Google Scholar
Google Scholar
[39] Kopycinski D, Guzik E, Siekaniec, Szczesny A 2015 Arch Foundry Eng 15 35
 Google Scholar
Google Scholar
[40] Liu Q 2013 Ph. D. Dissertation (Stockholm, Sweden: KTH Royal Institute of Technology)
-
图 4 Fe-C伪二元合金相图(A 区, 液相; B区, 液相 + TiC; C 区, 液相 + M7C3; D区, 液相+ M7C3 + γ; E 区, M7C3 + γ; F区, 液相 + TiC + M7C3; G区, 液相+ TiC + γ; H区, 液相+ TiC + M7C3 + γ; I 区, TiC + γ + M7C3) (a) Fe-C-17%Cr; (b) Fe-C-23.8%Cr; (c) Fe-C-17%Cr-1.5%Ti; (d) Fe-C-23.8%Cr-4%Ti
Fig. 4. Fe-C pseudo binary phase diagram: (a) Fe-C-17%Cr; (b) Fe-C-23.8%Cr; (c) Fe-C-17%Cr-1.5%Ti; (d) Fe-C-23.8%Cr-4%Ti. A zone, Liquid; B zone, Liquid + TiC; C zone, Liquid + M7C3; D zone, Liquid + M7C3 + γ; E zone, M7C3 + γ; F zone, Liquid + TiC + M7C3; G zone, Liquid + TiC + γ; H zone, Liquid + TiC + M7C3 + γ; I zone, TiC + γ + M7C3.
图 14 Fe-4%C-17%Cr合金实验[22]和预测的凝固组织 (a)实验凝固形貌; (b)本模拟奥氏体质量分数; (c)本模拟C浓度场; (d)本模拟Cr浓度场
Fig. 14. Experimental[22] and predicted solidification microstructure for Fe-4%C-17%Cr alloy: (a) experimental microstructure; (b) predicted austenite mass fraction; (c) predicted C concentration field; (d) predicted Cr concentration field.
表 1 Fe-4%C-17%Cr三元合金模拟所用的物性参数(单位%是指质量分数)
Table 1. Physical properties used for Fe-4%C-17%Cr ternary alloy. Unit of % represents mass fraction (wt%).
Parameters Symbol Unit Value Note Initial composition C $ {C_{{\text{C0}}}} $ % 4.00 文献[22] Initial composition Cr $ {C_{{\text{Cr0}}}} $ % 17.00 文献[22] Austenite nucleation temperature $ \mathop T\nolimits_{{\gamma }} $ ℃ 1266.00 GS model M7C3 nucleation temperature $ \mathop T\nolimits_{\text{M}} $ ℃ 1304.00 GS model C partition coefficient at austenite/liquid interface ${k_{ { {{\rm p}, {\mathrm{\gamma }/\mathrm{L} } , {\rm C} } } } }$ — 0.407 GS model Cr partition coefficient at austenite/liquid interface ${k_{ { {{\rm p}, {\mathrm{\gamma }/\mathrm{L} } , {\rm Cr} } } } }$ — 0.576 GS model Liquidus slope of C at austenite/liquid interface ${m_{ { { {\mathrm{L}/\mathrm{\gamma } }, {\rm C} } } } }$ ℃/% –95.49 GS model Liquidus slope of Cr at austenite/liquid interface ${m_{ { { {\mathrm{L}/\mathrm{\gamma } }, {\rm Cr} } } } }$ ℃/% 6.14 GS model Liquidus slope of C at M7C3/liquid interface ${m_{ {{\rm L/M}, \text{C} } } }$ ℃/% 90.37 GS model Liquidus slope of Cr at M7C3/liquid interface ${m_{ {{\rm L/M},\text{Cr} } } }$ ℃/% 15.29 GS model Diffusion coefficient of C in austenite ${D_{ { \mathrm{\gamma }, {\rm C} } } }$ m2/s 2.57×10–10 GS model Diffusion coefficient of Cr in austenite ${D_{ { {\mathrm{\gamma } , \rm Cr} } } }$ m2/s 3.67×10–14 GS model Diffusion coefficient of C in liquid phase ${D_{ {\text{L,C} } } }$ m2/s 9.60×10–10 GS model Diffusion coefficient of Cr in liquid phase ${D_{ {\text{L,Cr} } } }$ m2/s 8.23×10–10 GS model Diffusion coefficient of C in M7C3 ${D_{ {\text{M,C} } } }$ m2/s 0.0 Diffusion coefficient of Cr in M7C3 ${D_{ {\text{M,Cr} } } }$ m2/s 0.0 Gibbs-Thomson coefficient at austenite/liquid interface ${\varGamma _{ {\gamma } } }$ $ {\text{m}} \cdot {\text{K}} $ 1.9×10–7 文献[37] Gibbs-Thomson coefficient at M7C3/liquid interface ${\varGamma _{\text{M} } }$ $ {\text{m}} \cdot {\text{K}} $ 6.213×10–7 文献[38] Latent heat of fusion for austenite $ \mathop L\nolimits_{{\gamma }} $ J/kg 1.86×105 GS model Latent heat of fusion for M7C3 $ \mathop L\nolimits_{\text{M}} $ J/kg 2.38×105 GS model Specific heat capacity $\mathop c\nolimits_{\rm p}$ J/(kg$ \cdot $℃) 839 GS model 表 2 GS模型和Fe-C伪二元相图中二种成分Fe-C-Cr合金析出相及析出温度
Table 2. Phase type and precipitation temperature in two Fe-C-Cr alloys by GS prediction and in Fe-C pseudo binary phase diagram.
Model Alloy composition M7C3 precipitation temperature/℃ γ precipitation temperature/℃ CEM precipitation temperature or solidus /℃ Phase volume fraction at CEM precipitation
temperature or solidusGS model Fe-4%C-17%Cr 1304 1266 1194(CEM) 29.91%(M7C3) 57.22%(γ) GS model Fe-4%C-17%Cr-1.5%Ti 1288 1271 1193(CEM) 26.38%(M7C3) 63.20%(γ) Fe-C phase diagram Fe-4%C-17%Cr 1305 1266 1239(solidus) — Fe-C phase diagram Fe-4%C-17%Cr-1.5%Ti 1285 1271 1248(solidus) — GS model Fe-3.23%C-23.8%Cr 1305 1297 1193(CEM) 28.18%(M7C3) 71.62%(γ) GS model Fe-3.23%C-23.8%Cr-4%Ti 1296 1324 1296(solidus) 17.80%(M7C3) 74.28%(γ) Fe-C phase diagram Fe-3.23%C-23.8%Cr 1305 1296 1292(solidus) — Fe-C phase diagram Fe-3.23%C-23.8%Cr-4%Ti 1295 1328 1295(solidus) — -
[1] Siekaniec D, Kopycinski D, Szczesny A, Guzik E, Tyraa E, Nowak A 2017 Nephron Clinical Practice 17 143
 Google Scholar
Google Scholar
[2] Liu H N, Sakamoto M, Nomura M, Ogi K 2001 Wear 250 71
 Google Scholar
Google Scholar
[3] Pokusova M, Gabrisova Z, Brusilova A, Pribulova A, Futa P 2020 Mater. Sci. Forum. 998 30
 Google Scholar
Google Scholar
[4] Filipovic M, Kamberovic Z, Korac M, Gavrilovski M 2013 Mater. Des. 47 41
 Google Scholar
Google Scholar
[5] Buytoz S, Yildirim M M, Eren H 2005 Mater. Lett. 59 607
 Google Scholar
Google Scholar
[6] 龚沛, 赵曜, 杨浩, 王宁 2017 金属热处理 42 137
 Google Scholar
Google Scholar
Gong P, Zhao Y, Yang H, Wang N 2017 Heat Treatment of Metals 42 137
 Google Scholar
Google Scholar
[7] 陈哲, 王亮亮, 张晗, 付永红, 钟黎声, 叶芳霞 2013 热加工工艺 42 8
 Google Scholar
Google Scholar
Chen Z, Wang L L, Zhang H, Fu Y H, Zhong L S, Ye F X 2013 Hot Working Technology 42 8
 Google Scholar
Google Scholar
[8] Tang X H, Chung R, Pang C J, Li D Y, Hinckley B, Dolman K 2011 Wear 271 1426
 Google Scholar
Google Scholar
[9] Zhang H W, Nakajima K, Gandin C A, He J C 2013 ISIJ Int. 53 493
 Google Scholar
Google Scholar
[10] 王同敏, 魏晶晶, 王旭东, 姚曼 2018 金属学报 54 193
 Google Scholar
Google Scholar
Wang T M, Wei J J, Wang X D, Yao M 2018 Acta Metall. Sin. 54 193
 Google Scholar
Google Scholar
[11] 朱鸣芳, 邢丽科, 方辉, 张庆宇, 汤倩玉, 潘诗琰 2018 金属学报 54 789
 Google Scholar
Google Scholar
Zhu M F, Xing L K, Fang H, Zhang Q Y, Tang Q Y, Pan S Y 2018 Acta. Metall. Sin. 54 789
 Google Scholar
Google Scholar
[12] Zhu M F, Cao W, Chen S L, Hong C P, Chang Y A 2007 J. Phase Equilib. Diffus. 28 130
 Google Scholar
Google Scholar
[13] 戴挺, 朱鸣芳, 陈双林, 曹伟生, 洪俊杓 2008 金属学报 10 1175
 Google Scholar
Google Scholar
Dai T, Zhu M F, Chen S L, Cao W S, Hong J Z 2008 Acta Metall. Sin. 10 1175
 Google Scholar
Google Scholar
[14] Zhang H W, Gandin C A, Hamouda H B, Tourret D, Nakajima K, He J C 2010 ISIJ Int. 50 1859
 Google Scholar
Google Scholar
[15] Zhang H W, Gandin C A, Nakajima K, He J C 2012 IOP Conf Series: Materials Science Engineering 33 012063
 Google Scholar
Google Scholar
[16] 石玉峰, 许庆彦, 柳百成 2012 物理学报 61 108101
 Google Scholar
Google Scholar
Shi Y F, Xu Q Y, Liu B C 2012 Acta Phys. Sin. 61 108101
 Google Scholar
Google Scholar
[17] Michelic S C, Thuswaldner J M, Bernhard C 2010 Acta Mater. 58 2738
 Google Scholar
Google Scholar
[18] 张晛韦 2019 硕士学位论文 (沈阳: 东北大学)
Zhang X W 2019 Master's Dissertation (Shenyang: Northeastern University) (in Chinese)
[19] Zhu M F, Hong C P, Stefanescu D M, Chang Y A 2007 Metall. Mater. Trans. B. 38 517
 Google Scholar
Google Scholar
[20] 张蕾, 赵红蕾, 朱鸣芳 2015 金属学报 51 148
 Google Scholar
Google Scholar
Zhang L, Zhao H L, Zhu M F 2015 Acta Metall. Sin. 51 148
 Google Scholar
Google Scholar
[21] Ma S Q, Xing J, He Y, Li Y F, Huang Z F, Liu G Z, Geng Q J 2015 Mater. Chem. Phys. 161 65
 Google Scholar
Google Scholar
[22] Liu Q, Zhang H W, Wang Q, Zhou X K, Jonsson P G, Nakajima K 2012 ISIJ Int. 52 2210
 Google Scholar
Google Scholar
[23] Stefan-Kharicha M, Kharicha A, Zaidat K, Reiss G, Essl W, Goodwin F, Wu M H, Ludwig A, Mugrauer C 2020 J. Cryst. Growth 541 125667
 Google Scholar
Google Scholar
[24] 付振南, 许庆彦, 熊守美 2007 中国有色金属学报 17 1567
 Google Scholar
Google Scholar
Fu Z N, Xu Q Y, Xiong S M 2007 J. Nonferrous Met. China 17 1567
 Google Scholar
Google Scholar
[25] 董祥雷, 邢辉, 陈长乐, 沙莎, 王建元, 金克新 2016 物理学报 65 58
 Google Scholar
Google Scholar
Dong X L, Xing H, Chen C L, Sha S, Wang J Y, Jin K X 2016 Acta Phys. Sin. 65 58
 Google Scholar
Google Scholar
[26] Zhang H W, Zhang S, Wang Y C, Hao Y Z, Miao M, Nakajima K, Lei H, Wang Q, He J C 2020 IOP Conf Series: Materials Science and Engineering 861 012046
 Google Scholar
Google Scholar
[27] Gandin C A 1993 Acta Metallurgica et Materialia 41 345
 Google Scholar
Google Scholar
[28] Flemings M C, Poirier D R, Barone R V, Brody H D 1970 J. Iron Steel Inst. 208 371
[29] Nastac L 1999 Acta Mater. 47 4253
 Google Scholar
Google Scholar
[30] Eggleston J J, Mcfadden G B, Voorhees P W 2001 Phys. D 150 91
 Google Scholar
Google Scholar
[31] Rappaz M, Boettinger W J 1999 Acta Mater. 47 3205
 Google Scholar
Google Scholar
[32] Burbelko A A, Gurgul D, Kaptur K W, Gorny M 2012 Arch. Foundry Eng. 12 11
 Google Scholar
Google Scholar
[33] 孙玉成 2012 博士学位论文 (山东: 山东大学)
Sun Y C 2012 Ph. D. Dissertation (Shandong: Shandong University) (in Chinese)
[34] Wang J, Qi X, Xing X, Shi Z J, Zhou Y F, Yang Q X 2019 Mater. Res. Express 6 0865h3
 Google Scholar
Google Scholar
[35] Liu S, Zhou Y F, Xing X, Wang J B, Ren X J, Yang Q X 2016 Sci. Rep. 6 1
 Google Scholar
Google Scholar
[36] Coronado J J 2011 Wear 270 287
 Google Scholar
Google Scholar
[37] Beltran S L, Stefanescu D M 2004 Metall. Mater. Trans. A. 35 2471
 Google Scholar
Google Scholar
[38] Liu S, Wang Z, Shi Z, Zhou Y F, Yang Q X 2017 J. Alloy. Compd. 713 108
 Google Scholar
Google Scholar
[39] Kopycinski D, Guzik E, Siekaniec, Szczesny A 2015 Arch Foundry Eng 15 35
 Google Scholar
Google Scholar
[40] Liu Q 2013 Ph. D. Dissertation (Stockholm, Sweden: KTH Royal Institute of Technology)
计量
- 文章访问数: 7421
- PDF下载量: 82
- 被引次数: 0














 下载:
下载: