-
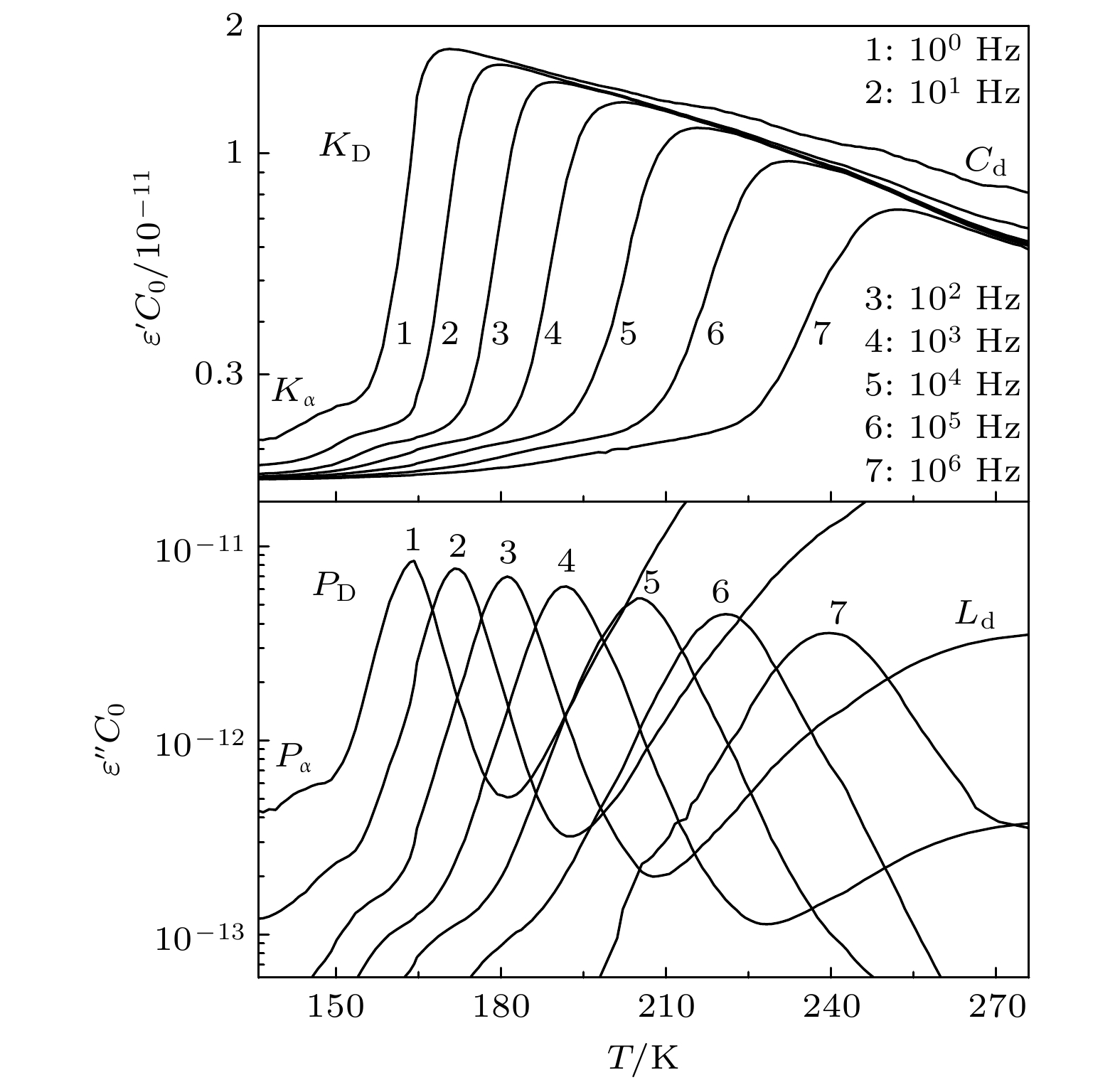
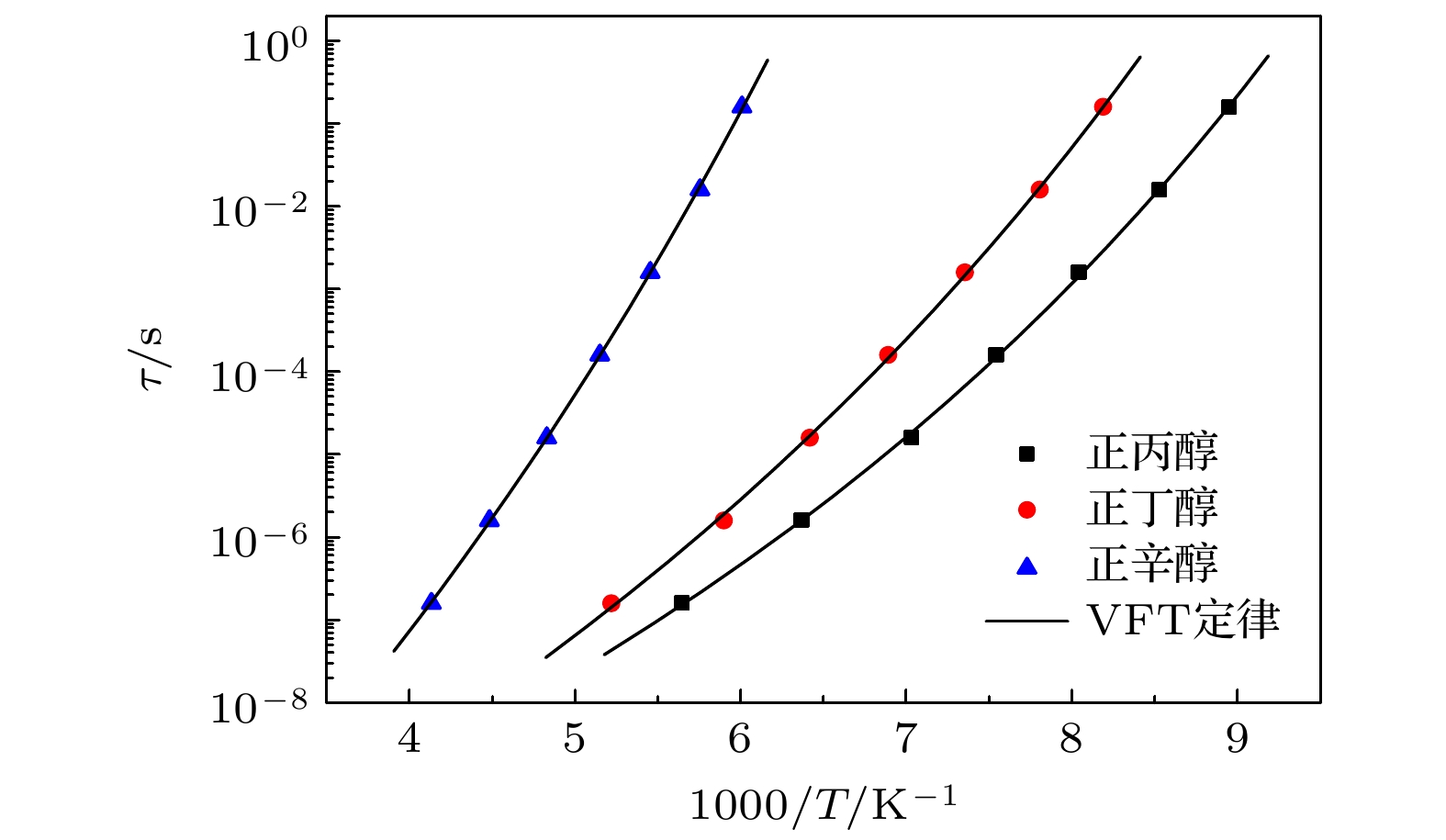
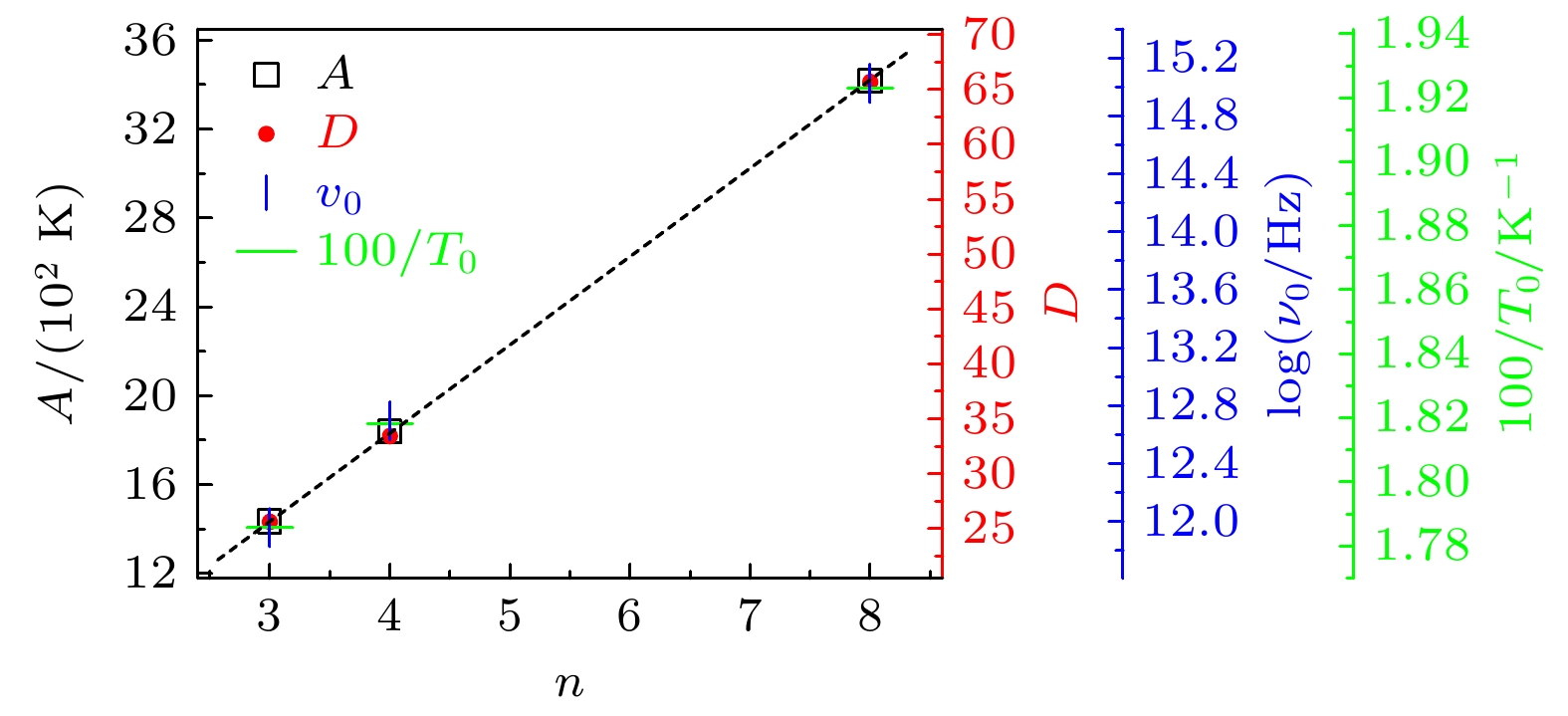
单羟基醇具有其他液体通常所不具备的Debye弛豫过程, 随着研究工作的开展, 一些与之相关的新现象和新问题也逐渐被发现, 深化了对物质结构和动力学的认知. 为了进一步研究Debye弛豫过程的动力学及其受分子构成影响的情况, 本文通过介电谱方法对3种无支链无侧基伯醇中的Debye弛豫进行测量与分析, 揭示了该过程的一些变化规律. 在正丙醇、正丁醇和正辛醇这3种伯醇中Debye过程的特征温度、Vogel-Fulcher-Tammann (VFT)温度的倒数和强度参量以及弛豫单元在高温极限下的激活能和固有振动频率的对数几乎都随分子内碳原子数的增加而线性增加. 但是VFT温度变化不大, 具有一致性, 表明这3种单羟基醇中Debye过程的弛豫单元应该相同, 进一步验证了Debye弛豫来源于氢键分子链中羟基的观点. 将样品的沸点和熔点等信息与上述激活能的变化进行对比, 表明氢键作用与分子之间的整体相互作用具有正相关性. 另外, 将强度参量的变化情况与相关理论进行结合, 给出了研究液体脆性的一个可能视角; 三者弛豫过程与乙醇结果的对比, 显示出Debye弛豫与α弛豫的分离程度会受到分子链长的影响, 也为Debye弛豫的研究提供了切入点. 这些结果将为单羟基醇Debye弛豫的进一步理解与研究产生促进作用, 也为相关理论提供检验性的实验信息.Monohydroxy alcohol has a Debye relaxation process that other liquids usually do not have, and with the further research, some new phenomena and new problems related to the process have been gradually discovered, deepening the understanding of material structure and dynamics. In order to further investigate the dynamics of Debye relaxation processes and the influence of molecular constitutions on them, the Debye processes of three primary alcohols without branched chains or side groups are studied by dielectric spectroscopy method, and some important information about the processes is revealed. A number of dynamic parameters of Debye relaxation in n-propanol, n-butanol and n-octanol almost all increase linearly with the rising of the number of carbon atoms in the molecules, which include the characteristic temperature, the reciprocal of Vogel-Fulcher-Tammann (VFT) temperature, the strength parameter of Debye process, the activation energy and the logarithm of the intrinsic vibration frequency of relaxation units under high temperature limit. However, the values of VFT temperatures change little and have consistency, illustrating that the relaxation units of Debye processes in these three monohydroxy alcohols should be the same, further verifying the view that the Debye relaxation originates from the hydroxyl groups in hydrogen bonded molecular chains. Comparing Boiling temperatures and melting temperatures of those samples with the evolution of the above activation energy, it is shown that there is a positive correlation between the interaction among hydrogen bonds and the whole one among molecules. In addition, combining the information about the strength parameter with that from the relevant theories, a possible perspective is gained for further investigation of liquid fragility. The comparison of those three samples with ethanol displays that the degree of separation of Debye relaxation and α relaxation is influenced by the molecular chain length, which can provide a breakthrough point to explore Debye relaxation. These results in this work will promote the further understanding and research of Debye relaxation in monohydroxy alcohols, and also provide experimental information for relevant theories.
-
Keywords:
- Debye relaxation /
- monohydroxy alcohol /
- dielectric spectroscopy /
- glass transition
[1] Böhmer R, Gainaru C, Richert R 2014 Phys. Rep. 545 125
 Google Scholar
Google Scholar
[2] Huth H, Wang L M, Schick C, Richert R 2007 J. Chem. Phys. 126 104503
 Google Scholar
Google Scholar
[3] Jakobsen B, Maggi C, Christensen T, Dyre J C 2008 J. Chem. Phys. 129 184502
 Google Scholar
Google Scholar
[4] Gainaru C, Kastner S, Mayr F, Lunkenheimer P, Schildmann S, Weber H J, Hiller W, Loidl A, Böhmer R 2011 Phys. Rev. Lett. 107 118304
 Google Scholar
Google Scholar
[5] Bauer S, Burlafinger K, Gainaru C, Lunkenheimer P, Hiller W, Loidl A, Böhmer R 2013 J. Chem. Phys. 138 94505
 Google Scholar
Google Scholar
[6] Wang L N, Zhao X Y, Huang Y N 2019 Int. J. Mod. Phys. B 33 1950313
 Google Scholar
Google Scholar
[7] Wang L N, Zhao X Y, Huang Y N 2019 Chin. Phys. Lett. 36 097701
 Google Scholar
Google Scholar
[8] Zhao X Y, Wang L N, He Y F, Zhou H W, Huang Y N 2020 Chem. Phys. 528 110473
 Google Scholar
Google Scholar
[9] Lu G H, Wang L N, Zhao X Y, He Y F, Huang Y N 2021 Int. J. Mod. Phys. B 35 2150014
 Google Scholar
Google Scholar
[10] Wang L M, Tian Y J, Liu R P, Richert R 2008 J. Chem. Phys. 128 084503
 Google Scholar
Google Scholar
[11] 李艳伟, 孙昭艳, 安立佳 2016 大学化学 31 1
 Google Scholar
Google Scholar
Li Y W, Sun Z Y, An L J 2016 Univ. Chem. 31 1
 Google Scholar
Google Scholar
[12] Dyre J C 2006 Rev. Mod. Phys. 78 953
 Google Scholar
Google Scholar
[13] Angell C A, Ngai K L, McKenna G B, McMillan P F, Martin S W 2000 J. Appl. Phys. 88 3113
 Google Scholar
Google Scholar
[14] 汪卫华 2013 物理学进展 33 177
Wang W H 2013 Prog. Phys. 33 177
[15] Gainaru C, Meier R, Schildmann S, Lederle C, Hiller W, Rössler E A, Böhmer R 2010 Phys. Rev. Lett. 105 258303
 Google Scholar
Google Scholar
[16] Gao Y Q, Tu W K, Chen Z M, Tian Y J, Liu R P, Wang L M 2013 J. Chem. Phys. 139 164504
 Google Scholar
Google Scholar
[17] Zhao X Y, Wang L N, Yin H M, Zhou H W, Huang Y N 2019 Chin. Phys. B 28 086601
 Google Scholar
Google Scholar
[18] Xu D, Feng S D, Wang J Q, Wang L M, Richert R 2020 J. Phys. Chem. Lett. 11 5792
 Google Scholar
Google Scholar
[19] Havriliak S, Negami S 1967 Polymer 8 161
 Google Scholar
Google Scholar
[20] Richert R 2010 J. Chem. Phys. 133 74502
 Google Scholar
Google Scholar
[21] Wang J, Zhao K S, Wu L X 2014 J. Chem. Phys. 141 54502
 Google Scholar
Google Scholar
[22] Ishai P B, Talary M S, Caduff A, Levy E, Feldman Y 2013 Meas. Sci. Technol. 24 102001
 Google Scholar
Google Scholar
[23] Brand R, Lunkenheimer P, Loidl A 2002 J. Chem. Phys. 116 10386
 Google Scholar
Google Scholar
[24] Chua Y Z, Young-Gonzales A R, Richert R, Ediger M D, Schick C 2017 J. Chem. Phys. 147 014502
 Google Scholar
Google Scholar
[25] Scherer G W 1992 J. Am. Ceram. Soc. 75 1060
 Google Scholar
Google Scholar
[26] Vogel H 1921 Phys. Zeit. 22 645
[27] Fulcher G S 1925 J. Am. Ceram. Soc. 8 339
 Google Scholar
Google Scholar
[28] Tammann G, Hesse W 1926 Z. Anorg. Allg. Chem. 156 245
 Google Scholar
Google Scholar
[29] Böhmer R, Ngai K L, Angell C A, Plazek D J 1993 J. Chem. Phys. 99 4201
 Google Scholar
Google Scholar
[30] Huang Y N, Wang C J, Riande E 2005 J. Chem. Phys. 122 144502
 Google Scholar
Google Scholar
[31] 赵兴宇, 王丽娜, 樊小辉, 张丽丽, 卫来, 张晋鲁, 黄以能 2011 物理学报 60 036403
 Google Scholar
Google Scholar
Zhao X Y, Wang L N, Fan X H, Zhang L L, Wei L, Zhang J-L, Huang Y N 2011 Acta Phys. Sin. 60 036403
 Google Scholar
Google Scholar
[32] Angell C A 1995 Science 267 1924
 Google Scholar
Google Scholar
[33] Lunkenheimer P, Schneider U, Brand R, Loidl A 2000 Contemp. Phys. 41 15
 Google Scholar
Google Scholar
[34] Tschamler H, Richter E, Wettig F 1949 Monatsh. Chem. 80 749
 Google Scholar
Google Scholar
[35] Wang L M, Richert R 2007 J. Phys. Chem. B 111 3201
 Google Scholar
Google Scholar
-
表 1 正丙醇、正丁醇和正辛醇的
$ n $ ,$ A $ ,$ {\nu }_{0} $ ,$ {T}_{0} $ ,$ {T}_{100} $ 以及D值Table 1. Values of
$ n $ ,$ A $ ,$ {\nu }_{0} $ ,$ {T}_{0} $ ,$ {T}_{100} $ and D in n-propanol, n-butanol and n-octanol.样品 $ n $ $ A $/K $ {\nu }_{0} $/Hz $ {T}_{0} $/K $ {T}_{100} $/K $ D $ 正丙醇 3 1434 9.08$ \times $1011 56 101 25.6 正丁醇 4 1839 4.97$ \times $1012 55 109 33.4 正辛醇 8 3416 1.06$ \times $1015 52 137 65.7 -
[1] Böhmer R, Gainaru C, Richert R 2014 Phys. Rep. 545 125
 Google Scholar
Google Scholar
[2] Huth H, Wang L M, Schick C, Richert R 2007 J. Chem. Phys. 126 104503
 Google Scholar
Google Scholar
[3] Jakobsen B, Maggi C, Christensen T, Dyre J C 2008 J. Chem. Phys. 129 184502
 Google Scholar
Google Scholar
[4] Gainaru C, Kastner S, Mayr F, Lunkenheimer P, Schildmann S, Weber H J, Hiller W, Loidl A, Böhmer R 2011 Phys. Rev. Lett. 107 118304
 Google Scholar
Google Scholar
[5] Bauer S, Burlafinger K, Gainaru C, Lunkenheimer P, Hiller W, Loidl A, Böhmer R 2013 J. Chem. Phys. 138 94505
 Google Scholar
Google Scholar
[6] Wang L N, Zhao X Y, Huang Y N 2019 Int. J. Mod. Phys. B 33 1950313
 Google Scholar
Google Scholar
[7] Wang L N, Zhao X Y, Huang Y N 2019 Chin. Phys. Lett. 36 097701
 Google Scholar
Google Scholar
[8] Zhao X Y, Wang L N, He Y F, Zhou H W, Huang Y N 2020 Chem. Phys. 528 110473
 Google Scholar
Google Scholar
[9] Lu G H, Wang L N, Zhao X Y, He Y F, Huang Y N 2021 Int. J. Mod. Phys. B 35 2150014
 Google Scholar
Google Scholar
[10] Wang L M, Tian Y J, Liu R P, Richert R 2008 J. Chem. Phys. 128 084503
 Google Scholar
Google Scholar
[11] 李艳伟, 孙昭艳, 安立佳 2016 大学化学 31 1
 Google Scholar
Google Scholar
Li Y W, Sun Z Y, An L J 2016 Univ. Chem. 31 1
 Google Scholar
Google Scholar
[12] Dyre J C 2006 Rev. Mod. Phys. 78 953
 Google Scholar
Google Scholar
[13] Angell C A, Ngai K L, McKenna G B, McMillan P F, Martin S W 2000 J. Appl. Phys. 88 3113
 Google Scholar
Google Scholar
[14] 汪卫华 2013 物理学进展 33 177
Wang W H 2013 Prog. Phys. 33 177
[15] Gainaru C, Meier R, Schildmann S, Lederle C, Hiller W, Rössler E A, Böhmer R 2010 Phys. Rev. Lett. 105 258303
 Google Scholar
Google Scholar
[16] Gao Y Q, Tu W K, Chen Z M, Tian Y J, Liu R P, Wang L M 2013 J. Chem. Phys. 139 164504
 Google Scholar
Google Scholar
[17] Zhao X Y, Wang L N, Yin H M, Zhou H W, Huang Y N 2019 Chin. Phys. B 28 086601
 Google Scholar
Google Scholar
[18] Xu D, Feng S D, Wang J Q, Wang L M, Richert R 2020 J. Phys. Chem. Lett. 11 5792
 Google Scholar
Google Scholar
[19] Havriliak S, Negami S 1967 Polymer 8 161
 Google Scholar
Google Scholar
[20] Richert R 2010 J. Chem. Phys. 133 74502
 Google Scholar
Google Scholar
[21] Wang J, Zhao K S, Wu L X 2014 J. Chem. Phys. 141 54502
 Google Scholar
Google Scholar
[22] Ishai P B, Talary M S, Caduff A, Levy E, Feldman Y 2013 Meas. Sci. Technol. 24 102001
 Google Scholar
Google Scholar
[23] Brand R, Lunkenheimer P, Loidl A 2002 J. Chem. Phys. 116 10386
 Google Scholar
Google Scholar
[24] Chua Y Z, Young-Gonzales A R, Richert R, Ediger M D, Schick C 2017 J. Chem. Phys. 147 014502
 Google Scholar
Google Scholar
[25] Scherer G W 1992 J. Am. Ceram. Soc. 75 1060
 Google Scholar
Google Scholar
[26] Vogel H 1921 Phys. Zeit. 22 645
[27] Fulcher G S 1925 J. Am. Ceram. Soc. 8 339
 Google Scholar
Google Scholar
[28] Tammann G, Hesse W 1926 Z. Anorg. Allg. Chem. 156 245
 Google Scholar
Google Scholar
[29] Böhmer R, Ngai K L, Angell C A, Plazek D J 1993 J. Chem. Phys. 99 4201
 Google Scholar
Google Scholar
[30] Huang Y N, Wang C J, Riande E 2005 J. Chem. Phys. 122 144502
 Google Scholar
Google Scholar
[31] 赵兴宇, 王丽娜, 樊小辉, 张丽丽, 卫来, 张晋鲁, 黄以能 2011 物理学报 60 036403
 Google Scholar
Google Scholar
Zhao X Y, Wang L N, Fan X H, Zhang L L, Wei L, Zhang J-L, Huang Y N 2011 Acta Phys. Sin. 60 036403
 Google Scholar
Google Scholar
[32] Angell C A 1995 Science 267 1924
 Google Scholar
Google Scholar
[33] Lunkenheimer P, Schneider U, Brand R, Loidl A 2000 Contemp. Phys. 41 15
 Google Scholar
Google Scholar
[34] Tschamler H, Richter E, Wettig F 1949 Monatsh. Chem. 80 749
 Google Scholar
Google Scholar
[35] Wang L M, Richert R 2007 J. Phys. Chem. B 111 3201
 Google Scholar
Google Scholar
计量
- 文章访问数: 6040
- PDF下载量: 83
- 被引次数: 0














 下载:
下载: