-
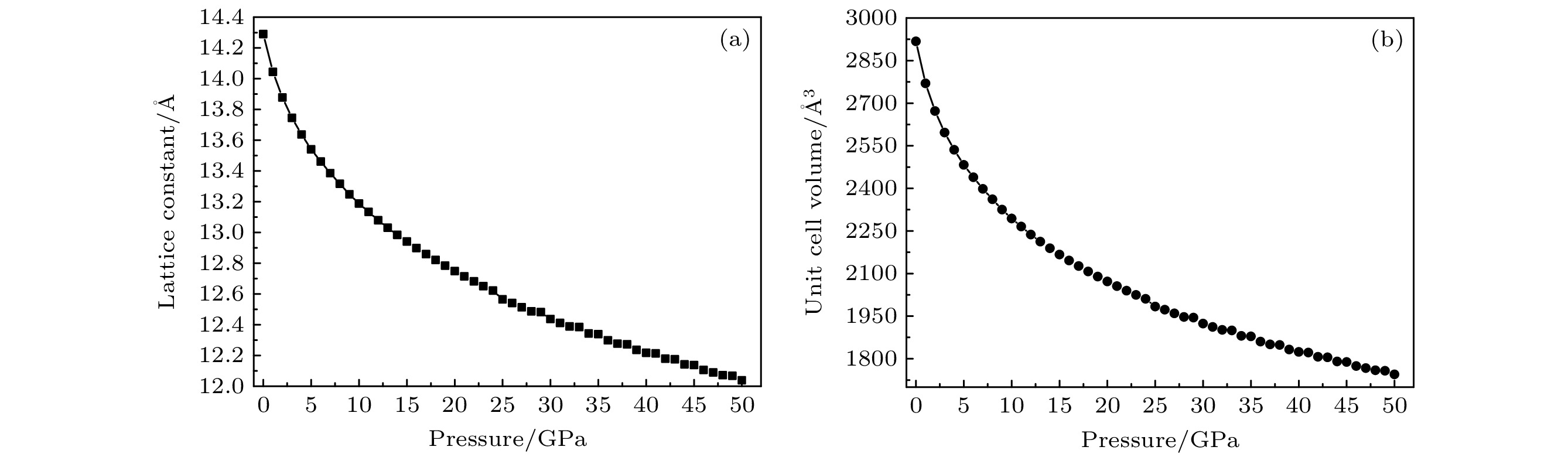
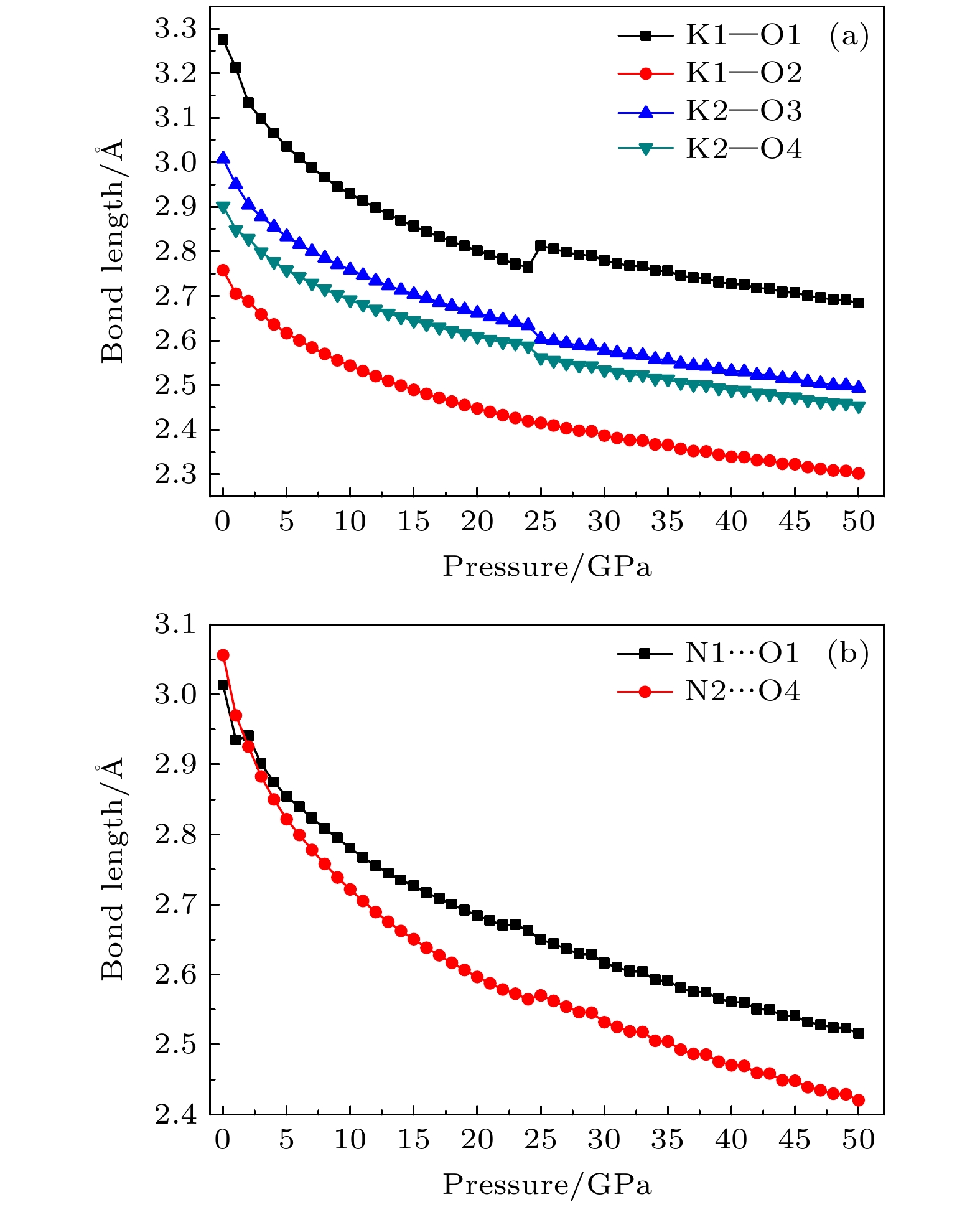
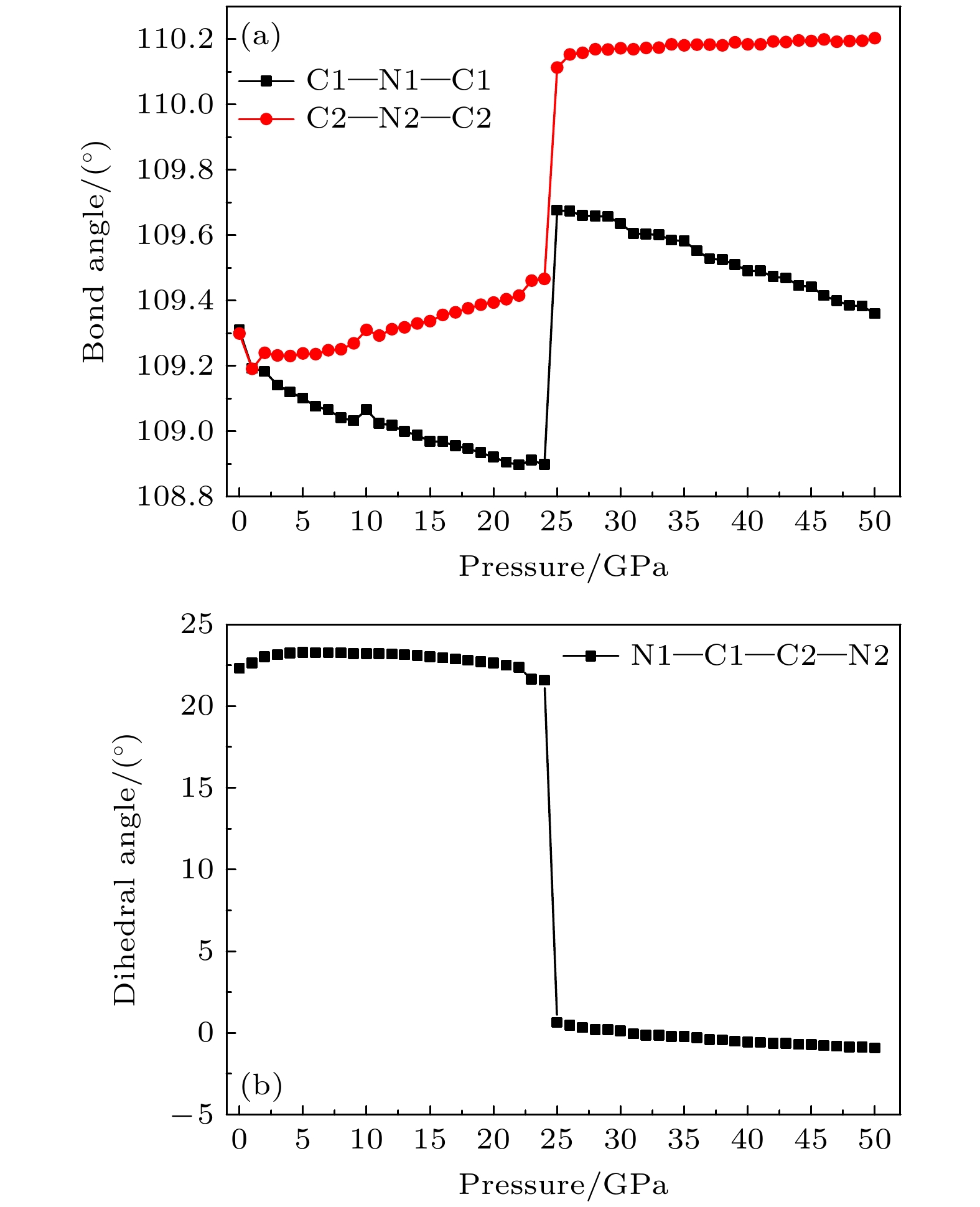
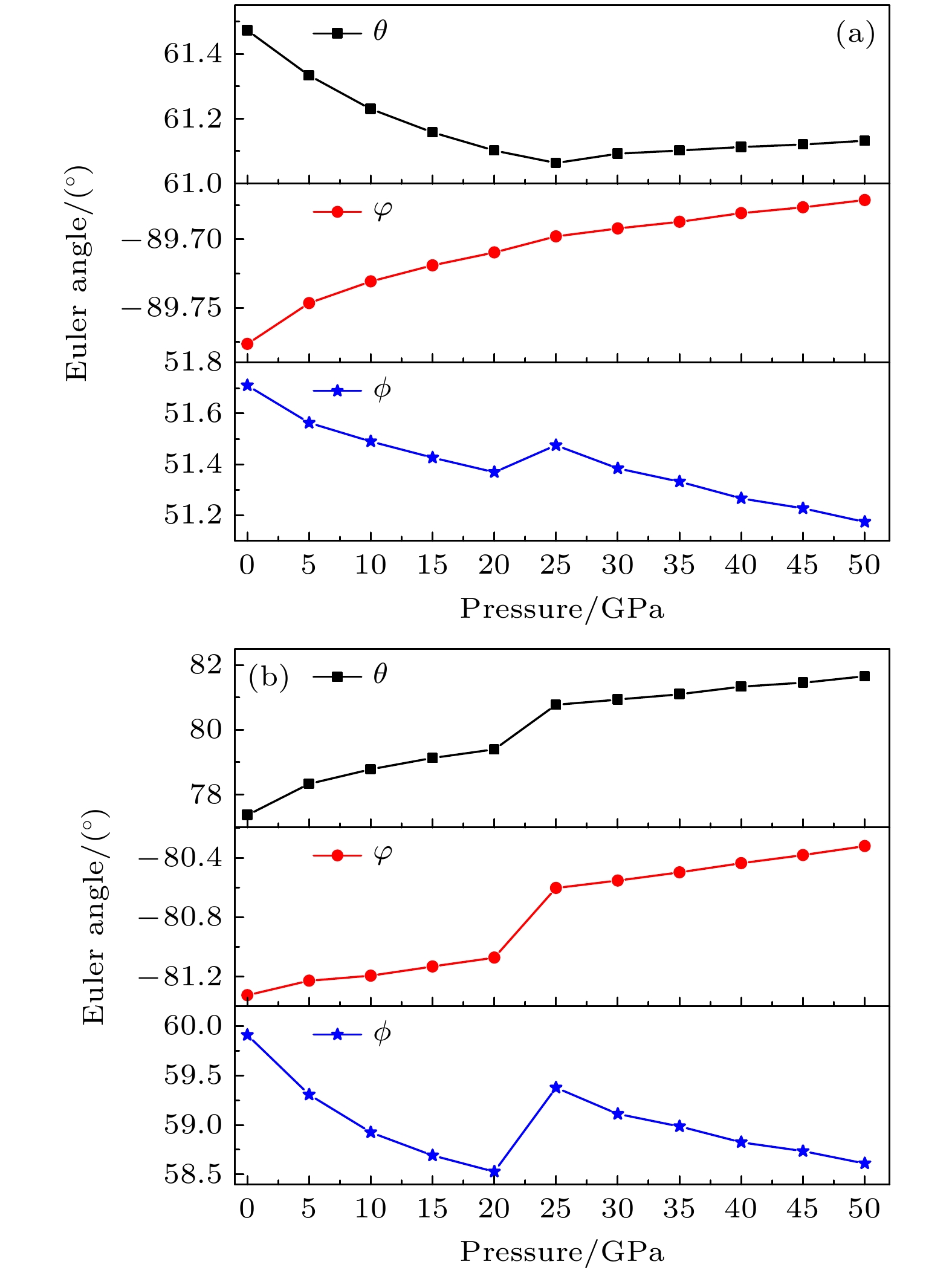
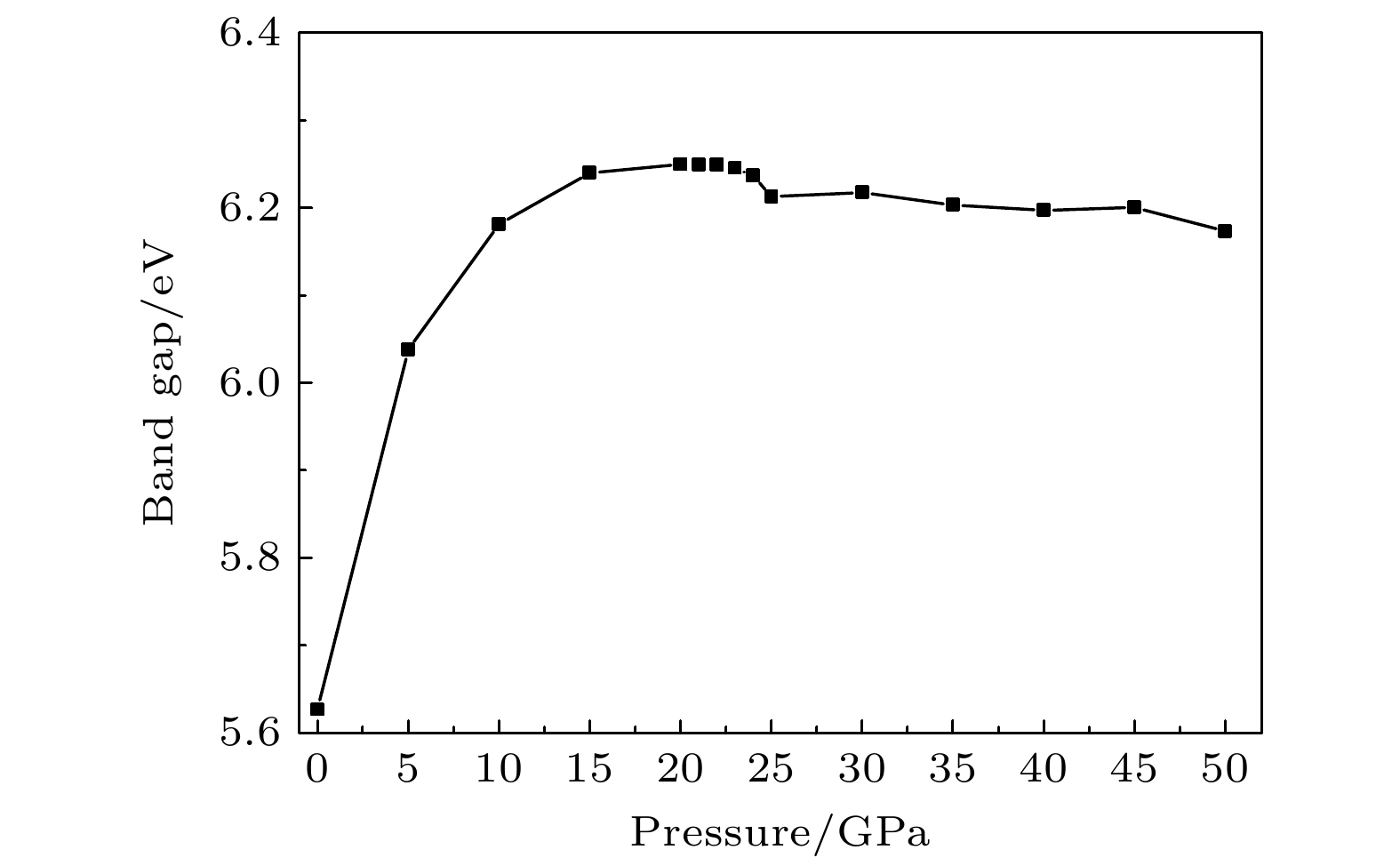
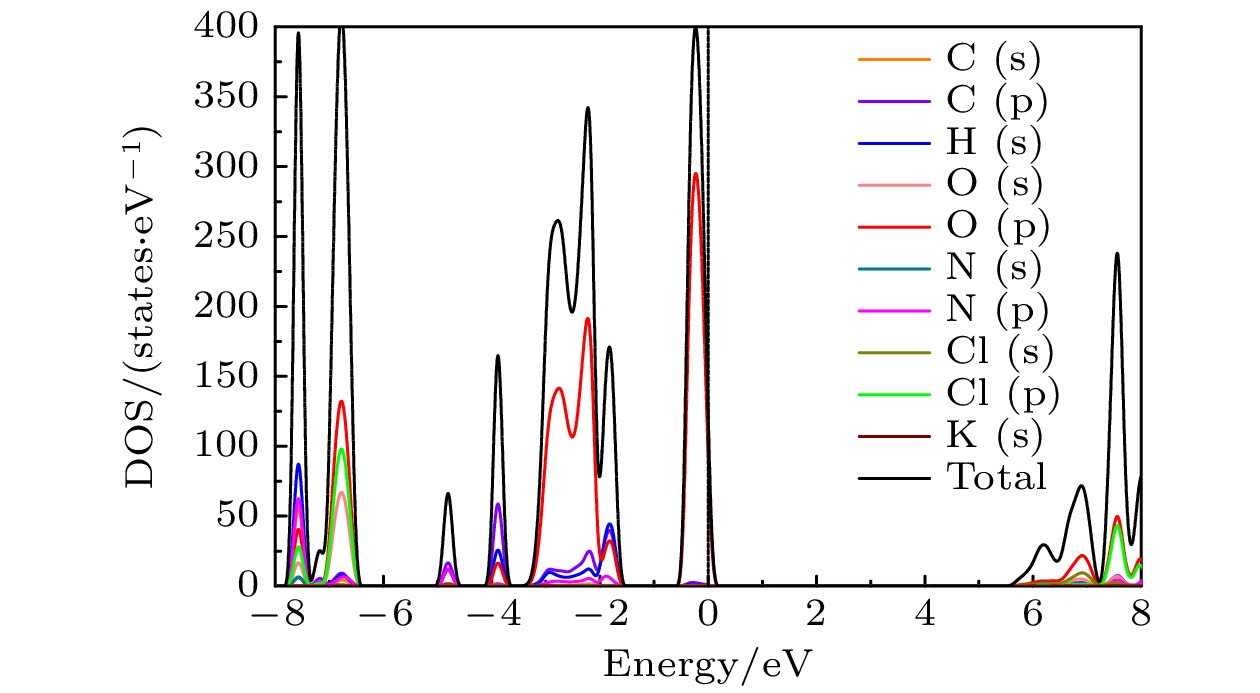
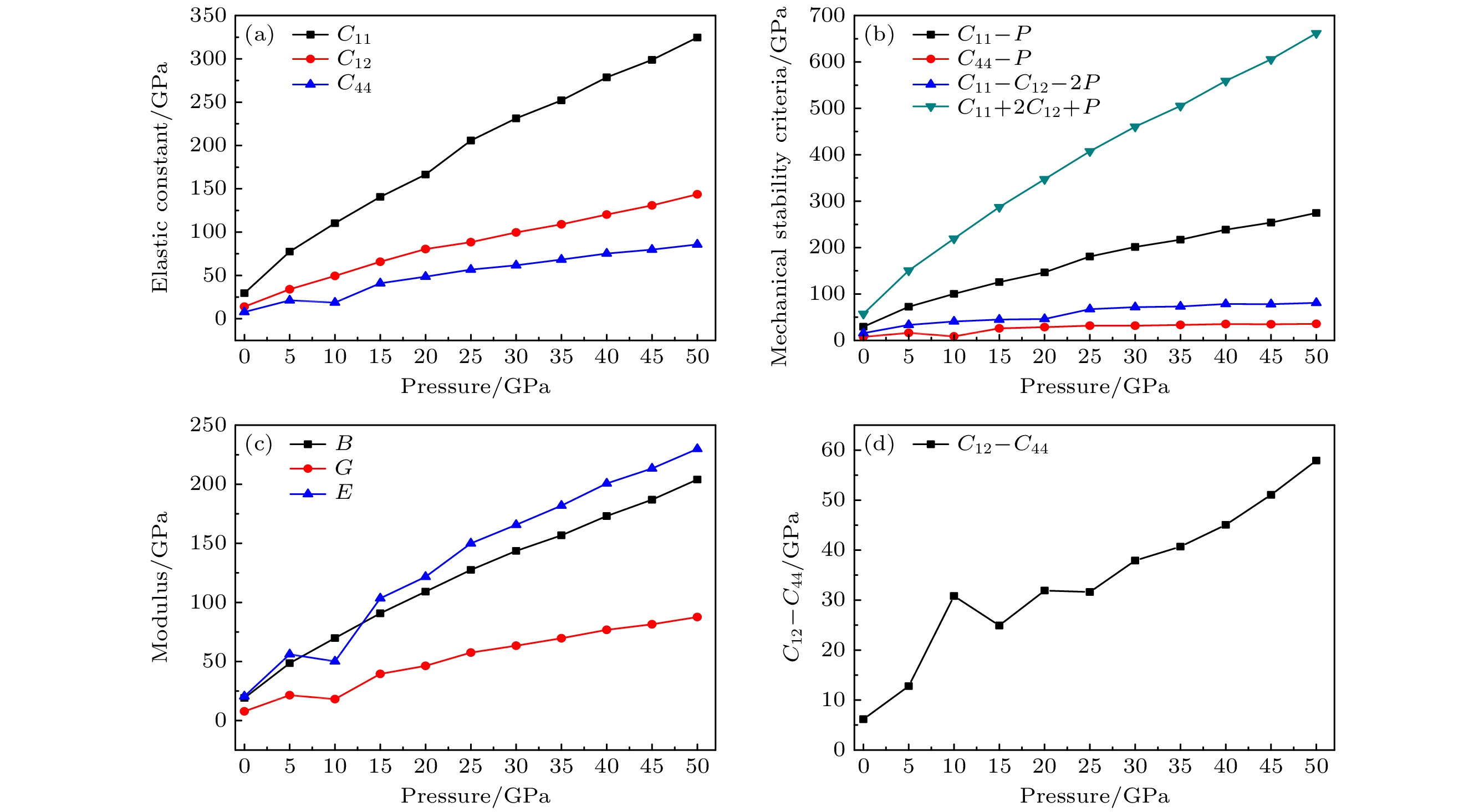
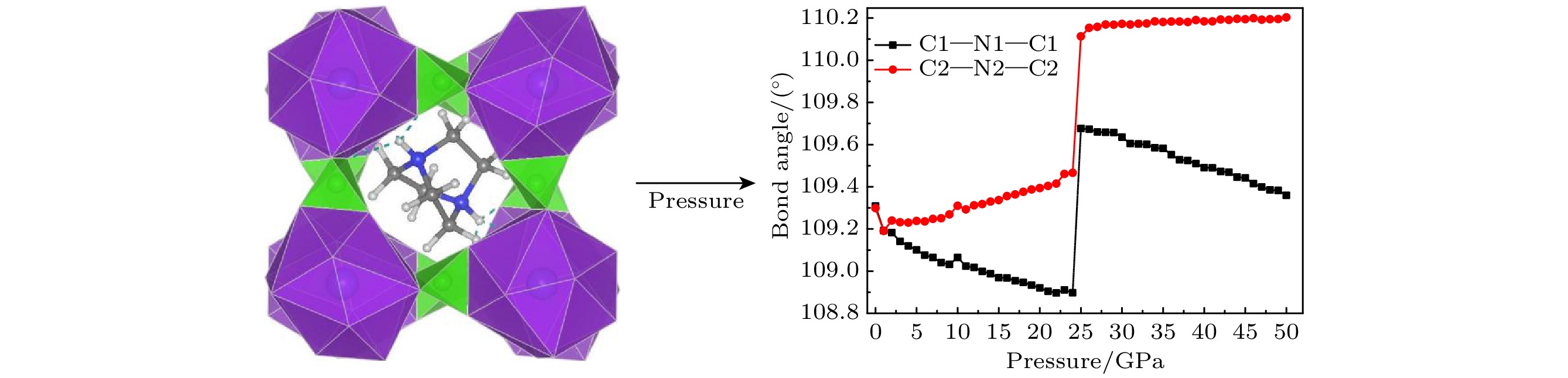
The crystal structure, molecular structure, electronic structure and mechanical properties of molecular perovskite high-energetic material (H2dabco)[K(ClO4)3] (DAP-2) under hydrostatic pressure ranging from 0 to 50 GPa are calculated and studied based on density functional theory. And the influences of pressure on its stability and impact sensitivity of DAP-2 are investigated. As the external pressure gradually increases, both the lattice parameters and the volume of DAP-2 crystal exhibit a monotonic decreasing trend. In the entire pressure range, the unit cell volume shrinks by up to 40.20%. By using the Birch Munnaghan equation of state to fit P-V relation, the bulk modulus B0 and its first-order derivative B0’ with respect to pressure are obtained to be 23.4 GPa and 4.9 GPa, respectively. The observations of the characteristic bond length and bond angle within the crystal indicate that the cage-like structure of organic cation H2dabco2+ undergoes distortion at 25 GPa. Further analysis of the average fractional coordinates of the center-of-mass and Euler angles for H2dabco2+ and KO12 polyhedron shows that within a pressure range from 0 to 50 GPa, both the average fractional coordinates of the center-of-mass and the Euler angles exhibit fluctuations at 25 GPa, but the overall amplitude of these fluctuations is very small. Based on this finding, it is speculated that the space group symmetry of the crystal may remain unchanged in the entire pressure range. In terms of electronic structure, with the increase of pressure, the band gap value increases rapidly and reaches a maximum value at about 20 GPa, followed by a slow decreasing trend. Based on the first-principles band gap criterion and the variation of the band gap under different pressures, it is demonstrated that below 20 GPa, the impact sensitivity of DAP-2 gradually decreases with pressure increasing; however, when the pressure exceeds 20 GPa, the impact sensitivity exhibits a slow increasing trend. In addition, the elastic constants Cij, Young’s modulus (E), bulk modulus (B), shear modulus (G), and Cauchy pressure (C12 – C44) all increase with pressure rising, indicating that the rigidity and ductility of the crystal under pressure are significantly strengthened. According to the mechanical stability criterion, the crystal maintains the mechanical stability throughout the pressure range.
-
Keywords:
- energetic perovskite /
- density functional theory /
- crystal structure /
- electronic structure /
- mechanical properties
[1] Agrawal J P, Hodgson R 2007 Organic Chemistry of Explosives (New York: Wiley
[2] Agrawal J P 2005 Propel. Explos. Pyrot. 30 316
 Google Scholar
Google Scholar
[3] Yu Q, Yin P, Zhang J H, He C L, Imler G H, Parrish D A, Shreeve J M 2017 J. Am. Chem. Soc. 139 8816
 Google Scholar
Google Scholar
[4] Kumar D, Imler G H, Parrish D A 2017 J. Mater. Chem. A 5 16767
 Google Scholar
Google Scholar
[5] Bennion J C, Siddiqi Z R, Matzger A J 2017 Chem. Commun. 53 6065
 Google Scholar
Google Scholar
[6] Zhang J H, Dharavath S, Mitchell L A, Parrish D A, Shreeve J M 2016 J. Am. Chem. Soc. 138 7500
 Google Scholar
Google Scholar
[7] He C, Shreeve J M 2016 Angew. Chem. 128 782
 Google Scholar
Google Scholar
[8] Liu W, Liu W L, Pang S P 2017 RSC Adv. 7 3617
 Google Scholar
Google Scholar
[9] Xu J G, Sun C, Zhang M J, Liu B W, Li X Z, Lu J, Wang S H, Zheng F K, Guo G C 2017 Chem. Mater. 29 9725
 Google Scholar
Google Scholar
[10] Sun C G, Zhang C, Jiang C, Yang C, Du Y, Zhao Y, Hu B C, Zheng Z S, Christe K O 2018 Nat. Commun. 9 1269
 Google Scholar
Google Scholar
[11] Wang S, Wang Q Y, Feng X, Wang B, Yang L 2017 Adv. Mater. 29 1701898
 Google Scholar
Google Scholar
[12] Shen C, Liu Y, Zhu Z Q, Xu Y G, Lu M 2017 Chem. Commun. 53 7489
 Google Scholar
Google Scholar
[13] Lin J D, Li Y H, Xu J G, Zheng F K, Guo G C, Lv R X, He W C, Huang Z N, Liu J F 2018 J. Solid State Chem. 265 42
 Google Scholar
Google Scholar
[14] Nielsen A T, Chafin A P, Christian S L, Moore D W, Nadler M P, Nissan R A, Vanderah D J, Gilardi R D, George C F, Flippen-Anderson J L 1998 Tetrahedron 54 11793
 Google Scholar
Google Scholar
[15] Liao W Q, Zhao D W, Tang Y Y, Zhang Y, Li P F, Shi P P, Chen X G, You Y M, Xiong R G 2019 Science 363 1206
 Google Scholar
Google Scholar
[16] Ye H Y, Tang Y Y, Li P F, Liao W Q, Gao J X, Hua X N, Cai H, Shi P P, You Y M, Xiong R G 2018 Science 361 151
 Google Scholar
Google Scholar
[17] 徐豪杰, 韩世国, 孙志华, 罗军华 2021 化学学报 79 23
 Google Scholar
Google Scholar
Luo J H, Sun Z H, Han S G, Xu H J 2021 Acta Chim. Sin. 79 23
 Google Scholar
Google Scholar
[18] He Y P, Galli G 2014 Chem. Mater. 26 5394
 Google Scholar
Google Scholar
[19] Xing G, Mathews N, Lim S S, Yantara N, Liu X F, Sabba D, Grätzel M, Mhaisalkar S, Sum T C 2014 Nat. Mater. 13 476
 Google Scholar
Google Scholar
[20] Guo Y L, Liu C, Tanaka H, Nakamura E 2015 J. Phys. Chem. Lett. 6 535
 Google Scholar
Google Scholar
[21] Chen S L, Yang Z R, Wang B J, Shang Y, Sun L Y, He C T, Zhou H L, Zhang W X, Chen X M 2018 Sci. China Mater. 61 1123
 Google Scholar
Google Scholar
[22] Chen S L, Shang Y, He C T, Sun L Y, Ye Z M, Zhang W X, Chen X M 2018 CrystEngComm 20 7458
 Google Scholar
Google Scholar
[23] Shang Y, Huang R K, Chen S L, He C T, Yu Z H, Ye Z M, Zhang W X, Chen X M, Design 2020 Cryst. Growth Des. 20 1891
 Google Scholar
Google Scholar
[24] Shang Y, Yu Z H, Huang R K, Chen S L, Liu D X, Chen X X, Zhang W X, Chen X M 2020 Eng. PRC. 6 1013
[25] Shang Y, Chen S L, Yu Z H, Huang R K, He C T, Ye Z M, Zhang W X, Chen X M 2022 Inorg. Chem. 61 4143
 Google Scholar
Google Scholar
[26] Feng Y, Zhang J, Cao W, Zhang J, Shreeve J n M 2023 Nat. Commun. 14 7765
 Google Scholar
Google Scholar
[27] Chen S, Yi Z, Jia C, Li Y, Chen H, Zhu S, Zhang L 2023 Small 19 2302631
 Google Scholar
Google Scholar
[28] Zhou J, Ding L, Bi F, Wang B, Zhang J 2018 J. Anal. Appl. Pyrolysis 129 189
 Google Scholar
Google Scholar
[29] An T, He W, Chen S W, Zuo B L, Qi X F, Zhao F Q, Luo Y J, Yan Q L 2018 J. Phys. Chem. C 122 26956
 Google Scholar
Google Scholar
[30] Jia Q, Deng P, Li X X, Hu L S, Cao X 2020 Vacuum 175 109257
 Google Scholar
Google Scholar
[31] Deng P, Wang H, Yang X, Ren H, Jiao Q J 2020 J. Alloy. Compd. 827 154257
 Google Scholar
Google Scholar
[32] Zhou J, Ding L, Zhao F Q, Wang B, Zhang J L 2020 Chin. Chem. Lett. 31 554
 Google Scholar
Google Scholar
[33] Li X X, Hu S Q, Cao X, Hu L S, Deng P, Xie Z B 2020 J. Energ. Mater. 38 162
 Google Scholar
Google Scholar
[34] Jia Q, Bai X, Zhu S, Cao X, Deng P, Hu L 2019 J. Energ. Mater. 38 377
[35] Kresse G, Hafner J 1993 Phys. Rev. B 47 558
 Google Scholar
Google Scholar
[36] Kresse G, Hafner J 1994 Phys. Rev. B 49 14251
 Google Scholar
Google Scholar
[37] Kresse G, Furthmüller J 1996 Phys. Rev. B 54 11169
 Google Scholar
Google Scholar
[38] Kresse G, Furthmüller J 1996 Comp. Mater. Sci. 6 15
 Google Scholar
Google Scholar
[39] Kresse G, Joubert D 1999 Phys. Rev. B 59 1758
[40] Perdew J P, Burke K, Ernzerhof M 1996 Phys. Rev. Lett. 77 3865
 Google Scholar
Google Scholar
[41] Ganose A M, Savory C N, Scanlon D O 2015 J. Phys. Chem. Lett. 6 4594
 Google Scholar
Google Scholar
[42] P J, Ruzsinszky A, Csonka G I, Vydrov O A, Scuseria G E, Constantin L A, Zhou X l, Burke K 2008 Phys. Rev. Lett. 100 136406
 Google Scholar
Google Scholar
[43] Le Page Y, Saxe P 2002 Phys. Rev. B 65 104104
 Google Scholar
Google Scholar
[44] Alyoubi R Y, Raffah B M, Hamioud F, Mubarak A A 2021 Mod. Phys. Lett. B 35 2150056
[45] Murnaghan F D 1944 P. Natl. Acad. Sci. USA 30 244
 Google Scholar
Google Scholar
[46] Feng G Q, Jiang X X, Wei W J, Gong P F, Kang L, Li Z H, Li Y C, Li X D, Wu X S, Lin Z S 2016 Dalton T. 45 4303
 Google Scholar
Google Scholar
[47] Agrawal P M, Rice B M, Zheng L Q, Velardez G F, Thompson D L 2006 J. Phys. Chem. B 110 5721
 Google Scholar
Google Scholar
[48] Agrawal P M, Rice B M, Zheng L Q, Thompson D L 2006 J. Phys. Chem. B 110 26185
 Google Scholar
Google Scholar
[49] Xiao H M, Li Y F 1995 Sci. China Ser. B 5 538
[50] Zhu W H, Xiao J J, Ji G F, Zhao F, Xiao H M 2007 J. Phys. Chem. B 111 12715
 Google Scholar
Google Scholar
[51] Xu X J, Zhu W H, Xiao H M 2007 J. Phys. Chem. B 111 2090
 Google Scholar
Google Scholar
[52] Zhu W H, Xiao H M 2008 J. Comput. Chem. 29 176
 Google Scholar
Google Scholar
[53] Zhu W H, Xiao H M 2010 Struct. Chem. 21 657
 Google Scholar
Google Scholar
[54] Fan J Y, Su Y, Zheng Z Y, Zhang Q Y, Zhao J J 2019 J. Raman Spectrosc. 50 889
 Google Scholar
Google Scholar
[55] Wu Q, Zhu W, Xiao H 2014 Struct. Chem. 26 477
[56] Xiang F, Wu Q, Zhu W H, Xiao H M 2014 Struct. Chem. 25 1625
 Google Scholar
Google Scholar
[57] Wang W P, Liu F S, Liu Q J, Wang Y G, Liu Z T 2016 Comp. Mater. Sci. 121 225
 Google Scholar
Google Scholar
[58] Feng J 2014 APL Mater. 2 081801
 Google Scholar
Google Scholar
[59] 袁文翎, 姚碧霞, 李喜, 胡顺波, 任伟 2024 物理学报 73 086104
 Google Scholar
Google Scholar
Yuan W L, Yao B X, Li X, Hu S B, Ren W 2024 Acta Phys. Sin. 73 086104
 Google Scholar
Google Scholar
[60] Liu Q J, Ran Z, Liu F S, Liu Z T 2015 J. Alloys Compd. 631 192
 Google Scholar
Google Scholar
[61] Chen S, Sun Y, Duan Y H, Huang B, Peng M J 2015 J. Alloys Compd. 630 202
 Google Scholar
Google Scholar
[62] Pettifor D G 1992 Mater. Sci. Technol. 8 345
 Google Scholar
Google Scholar
[63] Jund P, Viennois R, Tao X M, Niedziolka K, Tédenac J C 2012 Phys. Rev. B 85 224105
 Google Scholar
Google Scholar
-
图 1 (a) DAP-2单胞的多面体模型示意图; (b) DAP-2单胞的球棍模型示意图; (c) 有机阳离子H2dabco2+结构, K, O, Cl, C, N和H原子分别用蓝紫色、粉色、绿色、深灰色、蓝色和浅灰色表示, 而N—H…O键用青色虚线表示, 对称性代码: A: –z+1, –x+1, –y+1; B: –y+1/2, z –1/2, x; C: x –1/2, y, –z+3/2; D: z –1/2, –x+1/2, –y+1; E: x, y, z; F: –y+1/2, –z+1, x+1/2
Figure 1. (a) Schematic diagram of the polyhedral model for the unit cell of DAP-2; (b) schematic diagram of the ball-and-stick model for the unit cell of DAP-2; (c) structure of the organic cation H2dabco2+. The atoms of K, O, Cl, C, N and H are represented by blue purple, pink, green, dark gray, blue, and light gray, respectively, while N—H···O bonds are represented by cyan dashed lines. Symmetry code: A: –z+1, –x+1, –y+1; B: –y+1/2, z –1/2, x; C: x –1/2, y, –z+3/2; D: z –1/2, –x+1/2, –y+1; E: x, y, z; F: –y+1/2, –z+1, x+1/2.
表 1 DAP-2晶胞参数的计算值与实验值
Table 1. The calculated and experimental values of crystal cell parameters for DAP-2.
Method a/Å Δa/% α/(°) V/Å3 ΔV/% Experiment[21] 14.291 — 90 2918.689 — PBE 14.530 +1.67 90 3067.650 +5.10 PBEsol 14.288 –0.02 90 2917.954 –0.03 PBE+D3 14.282 –0.06 90 2913.178 –0.19 -
[1] Agrawal J P, Hodgson R 2007 Organic Chemistry of Explosives (New York: Wiley
[2] Agrawal J P 2005 Propel. Explos. Pyrot. 30 316
 Google Scholar
Google Scholar
[3] Yu Q, Yin P, Zhang J H, He C L, Imler G H, Parrish D A, Shreeve J M 2017 J. Am. Chem. Soc. 139 8816
 Google Scholar
Google Scholar
[4] Kumar D, Imler G H, Parrish D A 2017 J. Mater. Chem. A 5 16767
 Google Scholar
Google Scholar
[5] Bennion J C, Siddiqi Z R, Matzger A J 2017 Chem. Commun. 53 6065
 Google Scholar
Google Scholar
[6] Zhang J H, Dharavath S, Mitchell L A, Parrish D A, Shreeve J M 2016 J. Am. Chem. Soc. 138 7500
 Google Scholar
Google Scholar
[7] He C, Shreeve J M 2016 Angew. Chem. 128 782
 Google Scholar
Google Scholar
[8] Liu W, Liu W L, Pang S P 2017 RSC Adv. 7 3617
 Google Scholar
Google Scholar
[9] Xu J G, Sun C, Zhang M J, Liu B W, Li X Z, Lu J, Wang S H, Zheng F K, Guo G C 2017 Chem. Mater. 29 9725
 Google Scholar
Google Scholar
[10] Sun C G, Zhang C, Jiang C, Yang C, Du Y, Zhao Y, Hu B C, Zheng Z S, Christe K O 2018 Nat. Commun. 9 1269
 Google Scholar
Google Scholar
[11] Wang S, Wang Q Y, Feng X, Wang B, Yang L 2017 Adv. Mater. 29 1701898
 Google Scholar
Google Scholar
[12] Shen C, Liu Y, Zhu Z Q, Xu Y G, Lu M 2017 Chem. Commun. 53 7489
 Google Scholar
Google Scholar
[13] Lin J D, Li Y H, Xu J G, Zheng F K, Guo G C, Lv R X, He W C, Huang Z N, Liu J F 2018 J. Solid State Chem. 265 42
 Google Scholar
Google Scholar
[14] Nielsen A T, Chafin A P, Christian S L, Moore D W, Nadler M P, Nissan R A, Vanderah D J, Gilardi R D, George C F, Flippen-Anderson J L 1998 Tetrahedron 54 11793
 Google Scholar
Google Scholar
[15] Liao W Q, Zhao D W, Tang Y Y, Zhang Y, Li P F, Shi P P, Chen X G, You Y M, Xiong R G 2019 Science 363 1206
 Google Scholar
Google Scholar
[16] Ye H Y, Tang Y Y, Li P F, Liao W Q, Gao J X, Hua X N, Cai H, Shi P P, You Y M, Xiong R G 2018 Science 361 151
 Google Scholar
Google Scholar
[17] 徐豪杰, 韩世国, 孙志华, 罗军华 2021 化学学报 79 23
 Google Scholar
Google Scholar
Luo J H, Sun Z H, Han S G, Xu H J 2021 Acta Chim. Sin. 79 23
 Google Scholar
Google Scholar
[18] He Y P, Galli G 2014 Chem. Mater. 26 5394
 Google Scholar
Google Scholar
[19] Xing G, Mathews N, Lim S S, Yantara N, Liu X F, Sabba D, Grätzel M, Mhaisalkar S, Sum T C 2014 Nat. Mater. 13 476
 Google Scholar
Google Scholar
[20] Guo Y L, Liu C, Tanaka H, Nakamura E 2015 J. Phys. Chem. Lett. 6 535
 Google Scholar
Google Scholar
[21] Chen S L, Yang Z R, Wang B J, Shang Y, Sun L Y, He C T, Zhou H L, Zhang W X, Chen X M 2018 Sci. China Mater. 61 1123
 Google Scholar
Google Scholar
[22] Chen S L, Shang Y, He C T, Sun L Y, Ye Z M, Zhang W X, Chen X M 2018 CrystEngComm 20 7458
 Google Scholar
Google Scholar
[23] Shang Y, Huang R K, Chen S L, He C T, Yu Z H, Ye Z M, Zhang W X, Chen X M, Design 2020 Cryst. Growth Des. 20 1891
 Google Scholar
Google Scholar
[24] Shang Y, Yu Z H, Huang R K, Chen S L, Liu D X, Chen X X, Zhang W X, Chen X M 2020 Eng. PRC. 6 1013
[25] Shang Y, Chen S L, Yu Z H, Huang R K, He C T, Ye Z M, Zhang W X, Chen X M 2022 Inorg. Chem. 61 4143
 Google Scholar
Google Scholar
[26] Feng Y, Zhang J, Cao W, Zhang J, Shreeve J n M 2023 Nat. Commun. 14 7765
 Google Scholar
Google Scholar
[27] Chen S, Yi Z, Jia C, Li Y, Chen H, Zhu S, Zhang L 2023 Small 19 2302631
 Google Scholar
Google Scholar
[28] Zhou J, Ding L, Bi F, Wang B, Zhang J 2018 J. Anal. Appl. Pyrolysis 129 189
 Google Scholar
Google Scholar
[29] An T, He W, Chen S W, Zuo B L, Qi X F, Zhao F Q, Luo Y J, Yan Q L 2018 J. Phys. Chem. C 122 26956
 Google Scholar
Google Scholar
[30] Jia Q, Deng P, Li X X, Hu L S, Cao X 2020 Vacuum 175 109257
 Google Scholar
Google Scholar
[31] Deng P, Wang H, Yang X, Ren H, Jiao Q J 2020 J. Alloy. Compd. 827 154257
 Google Scholar
Google Scholar
[32] Zhou J, Ding L, Zhao F Q, Wang B, Zhang J L 2020 Chin. Chem. Lett. 31 554
 Google Scholar
Google Scholar
[33] Li X X, Hu S Q, Cao X, Hu L S, Deng P, Xie Z B 2020 J. Energ. Mater. 38 162
 Google Scholar
Google Scholar
[34] Jia Q, Bai X, Zhu S, Cao X, Deng P, Hu L 2019 J. Energ. Mater. 38 377
[35] Kresse G, Hafner J 1993 Phys. Rev. B 47 558
 Google Scholar
Google Scholar
[36] Kresse G, Hafner J 1994 Phys. Rev. B 49 14251
 Google Scholar
Google Scholar
[37] Kresse G, Furthmüller J 1996 Phys. Rev. B 54 11169
 Google Scholar
Google Scholar
[38] Kresse G, Furthmüller J 1996 Comp. Mater. Sci. 6 15
 Google Scholar
Google Scholar
[39] Kresse G, Joubert D 1999 Phys. Rev. B 59 1758
[40] Perdew J P, Burke K, Ernzerhof M 1996 Phys. Rev. Lett. 77 3865
 Google Scholar
Google Scholar
[41] Ganose A M, Savory C N, Scanlon D O 2015 J. Phys. Chem. Lett. 6 4594
 Google Scholar
Google Scholar
[42] P J, Ruzsinszky A, Csonka G I, Vydrov O A, Scuseria G E, Constantin L A, Zhou X l, Burke K 2008 Phys. Rev. Lett. 100 136406
 Google Scholar
Google Scholar
[43] Le Page Y, Saxe P 2002 Phys. Rev. B 65 104104
 Google Scholar
Google Scholar
[44] Alyoubi R Y, Raffah B M, Hamioud F, Mubarak A A 2021 Mod. Phys. Lett. B 35 2150056
[45] Murnaghan F D 1944 P. Natl. Acad. Sci. USA 30 244
 Google Scholar
Google Scholar
[46] Feng G Q, Jiang X X, Wei W J, Gong P F, Kang L, Li Z H, Li Y C, Li X D, Wu X S, Lin Z S 2016 Dalton T. 45 4303
 Google Scholar
Google Scholar
[47] Agrawal P M, Rice B M, Zheng L Q, Velardez G F, Thompson D L 2006 J. Phys. Chem. B 110 5721
 Google Scholar
Google Scholar
[48] Agrawal P M, Rice B M, Zheng L Q, Thompson D L 2006 J. Phys. Chem. B 110 26185
 Google Scholar
Google Scholar
[49] Xiao H M, Li Y F 1995 Sci. China Ser. B 5 538
[50] Zhu W H, Xiao J J, Ji G F, Zhao F, Xiao H M 2007 J. Phys. Chem. B 111 12715
 Google Scholar
Google Scholar
[51] Xu X J, Zhu W H, Xiao H M 2007 J. Phys. Chem. B 111 2090
 Google Scholar
Google Scholar
[52] Zhu W H, Xiao H M 2008 J. Comput. Chem. 29 176
 Google Scholar
Google Scholar
[53] Zhu W H, Xiao H M 2010 Struct. Chem. 21 657
 Google Scholar
Google Scholar
[54] Fan J Y, Su Y, Zheng Z Y, Zhang Q Y, Zhao J J 2019 J. Raman Spectrosc. 50 889
 Google Scholar
Google Scholar
[55] Wu Q, Zhu W, Xiao H 2014 Struct. Chem. 26 477
[56] Xiang F, Wu Q, Zhu W H, Xiao H M 2014 Struct. Chem. 25 1625
 Google Scholar
Google Scholar
[57] Wang W P, Liu F S, Liu Q J, Wang Y G, Liu Z T 2016 Comp. Mater. Sci. 121 225
 Google Scholar
Google Scholar
[58] Feng J 2014 APL Mater. 2 081801
 Google Scholar
Google Scholar
[59] 袁文翎, 姚碧霞, 李喜, 胡顺波, 任伟 2024 物理学报 73 086104
 Google Scholar
Google Scholar
Yuan W L, Yao B X, Li X, Hu S B, Ren W 2024 Acta Phys. Sin. 73 086104
 Google Scholar
Google Scholar
[60] Liu Q J, Ran Z, Liu F S, Liu Z T 2015 J. Alloys Compd. 631 192
 Google Scholar
Google Scholar
[61] Chen S, Sun Y, Duan Y H, Huang B, Peng M J 2015 J. Alloys Compd. 630 202
 Google Scholar
Google Scholar
[62] Pettifor D G 1992 Mater. Sci. Technol. 8 345
 Google Scholar
Google Scholar
[63] Jund P, Viennois R, Tao X M, Niedziolka K, Tédenac J C 2012 Phys. Rev. B 85 224105
 Google Scholar
Google Scholar
Catalog
Metrics
- Abstract views: 3858
- PDF Downloads: 126
- Cited By: 0















 DownLoad:
DownLoad: