-
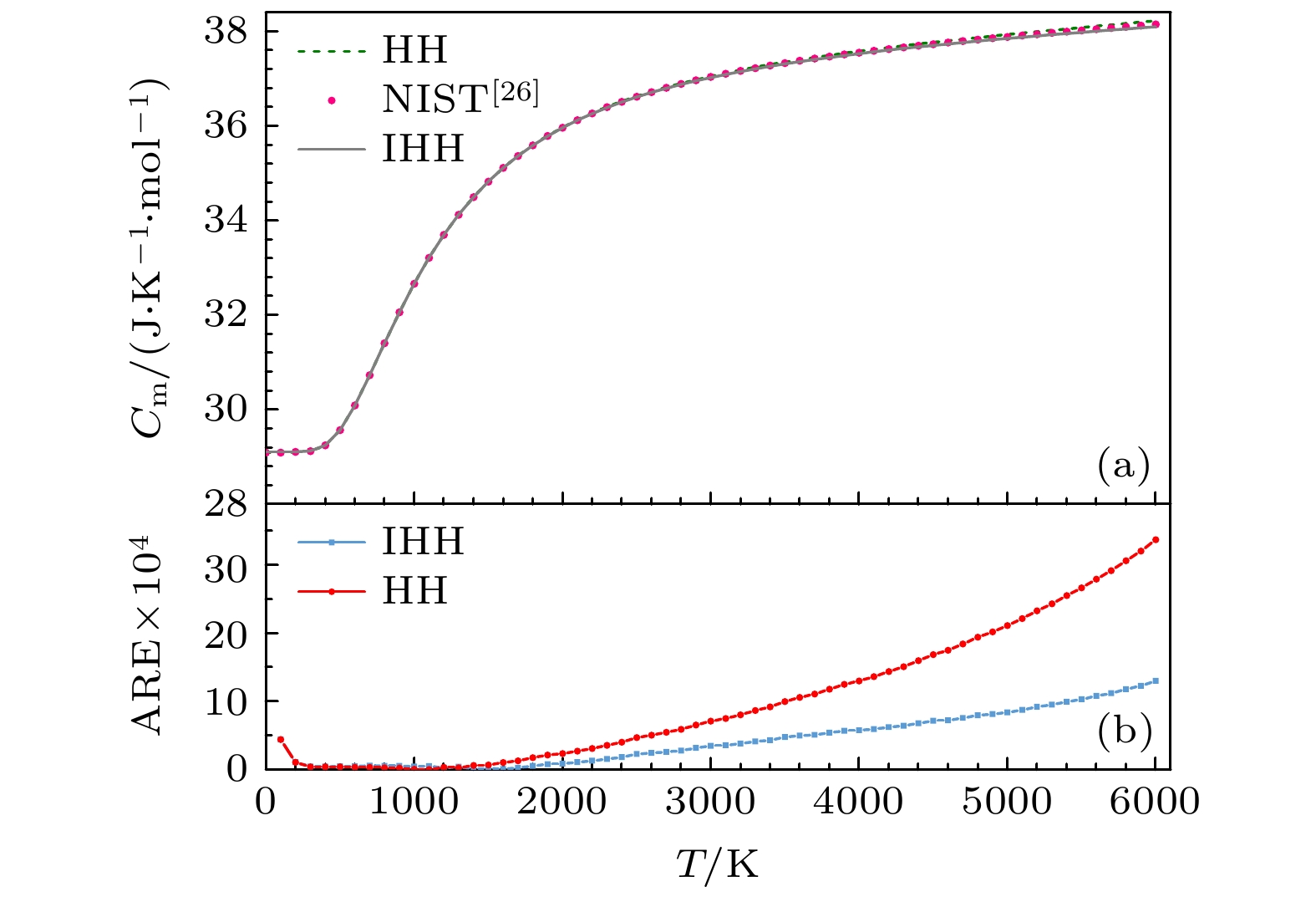
基于课题组前期获得的研究双原子分子某电子态完全势能函数改进后的Hulburt-Hirschfelder (improved Hulburt-Hirschfelder, IHH)势能模型, 结合实验测量得到的光谱常数和Rydberg-Klein-Rees (RKR)光谱数据, 对NO+离子基态包含离解区在内的全程势能曲线进行研究. 利用获得的完全势能曲线数值求解一维薛定谔方程, 得到了体系包含高激发态在内的振转能级, 并以此为基础计算获得体系总的内部配分函数, 最后借助量子统计系综理论计算NO+离子系统在100—6000 K温度内的摩尔热容、摩尔熵、摩尔焓和约化摩尔吉布斯自由能. 对NO+离子系统的研究结果表明, IHH势模型同样也适用于离子系统, 计算的势能曲线与实验数据吻合良好, 其精度优于HH势和MRCI/aug-cc-pV6Z势, 且预测的热力学性质较HH势模型更接近实验值. 本文提出的研究方法为通过双原子离子微观信息获取系统宏观热力学性质提供了一条新的有效途径.
NO+ is one of the most important ions in the atmospheric ionosphere and ionospheric phenomena such as auroras, and is one of the most stable diatomic cations existing in interstellar clouds. It is crucial to understand the thermodynamic properties of NO+ ion for exploring the composition of interstellar gas. To obtain macroscopic thermodynamic properties of diatomic molecules and ions, a practical theoretical method is to determine the partition function associated with a potential model. This approach can be used to calculate various thermodynamic properties of the system based on the microscopic information. In this work, the improved Hulbert-Hirschfelder (IHH) based potential energy model is used to simulate the potential energy curve of NO+ in the ground electronic state. Then, the rovibrational energy levels for the ground electronic state of the NO+ are obtained by numerically solving the radial Schrödinger equation through using the LEVEL program for the IHH potential function. Finally, the total partition function and the thermodynamic properties such as the molar heat capacity, entropy, enthalpy and reduced molar Gibbs free energy of NO+ in a temperature range of 100–6000 K are calculated in the frame of the quantum statistical ensemble theory. The comparison indicates that the potential energy curve calculated based on IHH potential energy function is in better agreement with the experimental data. The root mean square error of IHH potential and experimental Rydberg-Klein-Rees (RKR) potential is 96.9 cm–1, the root mean square error of Hulbert-Hirschfelder (HH) potential is 112.7 cm–1, and the root mean square error of MRCI/aug-cc-pV6Z potential is 133 cm–1. And the macroscopic thermodynamic properties of NO+ predicted by IHH are closer to the experimental values, which shows that the IHH potential model is also applicable to the ion system. A feasible method is presented to obtain the thermodynamic properties of gaseous diatomic ions based on microscopic information by constructing reliable analytical potential energy function associated with quantum statistical ensemble theory. -
Keywords:
- NO+ ion /
- improved Hulburt-Hirschfelder potential /
- partition function /
- thermodynamic properties
[1] Petrou A L 2012 Chem. Geo. 308 50
 Google Scholar
Google Scholar
[2] Ding Q C, Jia C S, Liu J Z, Li J, Du R F, Liu J Y, Peng X L, Wang C W, Tang H X 2022 Chem. Phys. Lett. 803 139844
 Google Scholar
Google Scholar
[3] Peduzzi E, Boissonnet G, Maréchal F 2016 Fuel 181 207
 Google Scholar
Google Scholar
[4] 汪志诚 2013 热力学·统计物理 (第五版) (北京: 高等教育出版社) 第 1 页
Wang Z C 2013 Thermodynamic Statistical Physics (Vol. 5) (Beijing: Higher Education Press) p1 (in Chinese)
[5] Kjelstrup S, Magnanelli E 2020 Trends Food Sci. Technol. 104 84
 Google Scholar
Google Scholar
[6] Horton T E 1970 The Computation of Partition Functions and Thermo-chemistry Data for Atomic, Ionic, Diatomic, and Polyatomic Species Technical Report 32-1425, National Aeronautics and Space Administration (NASA), Jet Propulsion Laboratory, California Institute of Technology, Pasadena, CA (N-70 20647, NASA CR108970)
[7] Bartl J, Bartlova M 1989 Contrib. Plasma Phys. 29 285
 Google Scholar
Google Scholar
[8] Peterson K A, Flowers B A, Francisco J S 2001 J. Chem. Phys. 115 7513
 Google Scholar
Google Scholar
[9] Capitelli M, Colonna G, Giordano D, Maraffa L, Casavola A, Minelli P, Pagano D, Pietanza L D, Taccogna F 2005 Tables of Internal Partition Functions and Thermodynamic Properties of High-Temperature Mars-Atmosphere Species from 50 K to 50000 K (Netherlands: European Space Agency Publications Division) p3
[10] Maltsev M A, Kulikov A N, Morozov IV 2016 J. Phys. Conf. Ser. 774 012023
 Google Scholar
Google Scholar
[11] Qin Z, Zhao J M, Liu L H 2018 J. Quant. Spectrosc. Radiat. Transfer. 210 1
 Google Scholar
Google Scholar
[12] Liang R H, Liu Y M, Li F Y 2021 Contrib. Plasma Phys. 61 e202100036
 Google Scholar
Google Scholar
[13] Eyube E S, Notani P P, Dikko A B 2022 Eur. Phys. J. Plus. 137 329
 Google Scholar
Google Scholar
[14] Maltsev M A, Morozov I V, Osina E L 2019 High. Temp. 57 335
 Google Scholar
Google Scholar
[15] Maltsev M A, Morozov I V, Osina E L 2019 High Temp. 57 37
 Google Scholar
Google Scholar
[16] Maltsev M A, Morozov I V, Osina E L 2020 High Temp. 58 184
 Google Scholar
Google Scholar
[17] Maltsev M A, Aksenova S A, Morozov I V, Minenkov Y, Osina E L 2023 J. Comput. Chem. 44 1189
 Google Scholar
Google Scholar
[18] Tian H R, Fan Q C, Fan Z X, Fu J, Li H D, Ma J, Xie F 2022 Int. J. Quantum Chem. 122 e26983
 Google Scholar
Google Scholar
[19] Partridge H, Langhoff S R, Bauschlicher C W 1990 J. Chem. Phys. 93 7179
 Google Scholar
Google Scholar
[20] Cosby P C, Helm H 1981 J. Chem. Phys. 75 3882
 Google Scholar
Google Scholar
[21] Werner H J, Rosmus P 1982 J. Mol. Spectrosc. 96 362
 Google Scholar
Google Scholar
[22] Sheehan C H, Maurice J P S 2004 J. Geophys. Res. 109 A03302
 Google Scholar
Google Scholar
[23] Liu H, Liu L H, Wei L Q, Chu B X, Qin Z Z, Jin G Z, Tong Z F, Dong L H, Li B 2020 Fuel 272 117738
 Google Scholar
Google Scholar
[24] Smith D, Chippendale T W E, Španěl P 2014 Rapid Commun. Mass Spectrom. 28 1917
 Google Scholar
Google Scholar
[25] Dias M, Gurusinghe R M, Suits A G 2022 J. Phys. Chem. A. 126 5354
 Google Scholar
Google Scholar
[26] Chase M W 1998 Journal of Physical and Chemical Reference Data Monograph (Vol. 9) (New York: National Institute of Standards and Technology Gaithersburg) p1604
[27] Dunham J L 1932 Phys. Rev. 41 721
 Google Scholar
Google Scholar
[28] Ilieva T, Iliev I 2016 J. Mol. Spectrosc. 330 28
 Google Scholar
Google Scholar
[29] Le Roy R J 2017 J. Quant. Spectrosc. Radiat. Transfer. 186 167
 Google Scholar
Google Scholar
[30] Schwabl F 2006 Statistical Mechanics (2nd Ed.) (Berlin Heidelberg: Springer-Verlag) p409
[31] Huber K P, Herzberg G 1950 Molecular Spectra and Molecular Structure: Spectra of Diatomic Molecules (New York: Van Nostrand Reinhold Company) p9
[32] Gordillo-Vázquez F J, Kunc J A 1998 J. Appl. Phys. 84 4693
 Google Scholar
Google Scholar
[33] Holbrook R, Kaledin L, Kunc J A 1993 Phys. Rev. E 47 1285
 Google Scholar
Google Scholar
[34] Holbrook R, Kunc J A 1994 Phys. Plasmas. 1 1075
 Google Scholar
Google Scholar
[35] Zhang J P, Cheng X L, Zhang H, Yang X D 2011 Chinese Phys. B. 20 060401
 Google Scholar
Google Scholar
[36] Reddy R R, Nazeer Ahammed Y, Baba Basha D, Narasimhulu K, Siva Sankar Reddy L, Rama Gopal K 2006 J. Quant. Spectrosc. Radiat. Transfer. 97 344
 Google Scholar
Google Scholar
[37] Barklem P S, Collet R 2016 Astron. Astrophys. 588 A96
 Google Scholar
Google Scholar
-
表 1 NO+离子不同势能及其与RKR实验值[36]的误差值
Table 1. Different potential energy of NO+ ion and its absolute error with the experimental value of RKR[36].
r/Å $ {E_{{\text{RKR}}}} $/cm–1[36] $ {E_{{\text{IHH}}}} $/cm–1 $\left| {\Delta {E_{ {\text{RKR–IHH} } } }} \right|$/cm–1 $ {E_{{\text{HH}}}} $/cm–1 $ \left| {\Delta {E_{{\text{RKR–HH}}}}} \right| $/cm–1 $ {E_{{\text{MRCI}}}} $/cm–1[35] $ \left| {\Delta {E_{{\text{RKR–MRCI}}}}} \right| $/cm–1 0.926 16911.0 17037.50 127 16886.09 25 17113.82 203 0.934 14761.9 14825.64 64 14721.53 40 14833.44 72 0.943 12580.2 12561.20 19 12494.49 86 12417.22 163 0.953 10366.1 10382.87 17 10342.77 23 10343.47 23 0.963 8119.40 8115.441 4 8094.606 25 8111.838 8 0.977 5840.20 5839.586 1 5830.842 9 5833.200 7 0.995 3528.50 3522.741 6 3520.407 8 3526.113 2 1.022 1184.30 1161.440 23 1161.306 23 1142.913 41 1.109 1184.30 1187.712 3 1187.826 4 1160.835 23 1.147 3528.50 3505.745 23 3507.331 21 3502.362 26 1.175 5840.20 5886.843 47 5892.355 52 5887.909 48 1.199 8119.40 8269.826 150 8282.220 163 8289.580 170 1.211 10366.1 10285.10 81 10305.88 60 10179.40 187 1.241 12580.2 12581.93 2 12615.36 35 12503.09 77 1.261 14761.9 14905.62 144 14955.40 194 14955.96 194 1.280 16911.0 17187.37 276 17256.83 346 17219.91 309 RMSD 96.9 112.7 133 表 2 HH, IHH势模型计算得到的NO+离子不同热力学性质的RMSD
Table 2. RMSD of HH and IHH potential models for different thermodynamic properties of NO+ ions.
热力学量 HH IHH $ {C_{\text{m}}} $/(J·mol–1·K–1) 0.053912 0.021733 $ {S_{\text{m}}} $/(J·mol–1·K–1) 0.035233 0.022930 $\Delta H$/(kJ·mol–1) 0.082503 0.034329 $ {G_{\text{r}}} $/(J·mol–1·K–1) 0.021079 0.017711 -
[1] Petrou A L 2012 Chem. Geo. 308 50
 Google Scholar
Google Scholar
[2] Ding Q C, Jia C S, Liu J Z, Li J, Du R F, Liu J Y, Peng X L, Wang C W, Tang H X 2022 Chem. Phys. Lett. 803 139844
 Google Scholar
Google Scholar
[3] Peduzzi E, Boissonnet G, Maréchal F 2016 Fuel 181 207
 Google Scholar
Google Scholar
[4] 汪志诚 2013 热力学·统计物理 (第五版) (北京: 高等教育出版社) 第 1 页
Wang Z C 2013 Thermodynamic Statistical Physics (Vol. 5) (Beijing: Higher Education Press) p1 (in Chinese)
[5] Kjelstrup S, Magnanelli E 2020 Trends Food Sci. Technol. 104 84
 Google Scholar
Google Scholar
[6] Horton T E 1970 The Computation of Partition Functions and Thermo-chemistry Data for Atomic, Ionic, Diatomic, and Polyatomic Species Technical Report 32-1425, National Aeronautics and Space Administration (NASA), Jet Propulsion Laboratory, California Institute of Technology, Pasadena, CA (N-70 20647, NASA CR108970)
[7] Bartl J, Bartlova M 1989 Contrib. Plasma Phys. 29 285
 Google Scholar
Google Scholar
[8] Peterson K A, Flowers B A, Francisco J S 2001 J. Chem. Phys. 115 7513
 Google Scholar
Google Scholar
[9] Capitelli M, Colonna G, Giordano D, Maraffa L, Casavola A, Minelli P, Pagano D, Pietanza L D, Taccogna F 2005 Tables of Internal Partition Functions and Thermodynamic Properties of High-Temperature Mars-Atmosphere Species from 50 K to 50000 K (Netherlands: European Space Agency Publications Division) p3
[10] Maltsev M A, Kulikov A N, Morozov IV 2016 J. Phys. Conf. Ser. 774 012023
 Google Scholar
Google Scholar
[11] Qin Z, Zhao J M, Liu L H 2018 J. Quant. Spectrosc. Radiat. Transfer. 210 1
 Google Scholar
Google Scholar
[12] Liang R H, Liu Y M, Li F Y 2021 Contrib. Plasma Phys. 61 e202100036
 Google Scholar
Google Scholar
[13] Eyube E S, Notani P P, Dikko A B 2022 Eur. Phys. J. Plus. 137 329
 Google Scholar
Google Scholar
[14] Maltsev M A, Morozov I V, Osina E L 2019 High. Temp. 57 335
 Google Scholar
Google Scholar
[15] Maltsev M A, Morozov I V, Osina E L 2019 High Temp. 57 37
 Google Scholar
Google Scholar
[16] Maltsev M A, Morozov I V, Osina E L 2020 High Temp. 58 184
 Google Scholar
Google Scholar
[17] Maltsev M A, Aksenova S A, Morozov I V, Minenkov Y, Osina E L 2023 J. Comput. Chem. 44 1189
 Google Scholar
Google Scholar
[18] Tian H R, Fan Q C, Fan Z X, Fu J, Li H D, Ma J, Xie F 2022 Int. J. Quantum Chem. 122 e26983
 Google Scholar
Google Scholar
[19] Partridge H, Langhoff S R, Bauschlicher C W 1990 J. Chem. Phys. 93 7179
 Google Scholar
Google Scholar
[20] Cosby P C, Helm H 1981 J. Chem. Phys. 75 3882
 Google Scholar
Google Scholar
[21] Werner H J, Rosmus P 1982 J. Mol. Spectrosc. 96 362
 Google Scholar
Google Scholar
[22] Sheehan C H, Maurice J P S 2004 J. Geophys. Res. 109 A03302
 Google Scholar
Google Scholar
[23] Liu H, Liu L H, Wei L Q, Chu B X, Qin Z Z, Jin G Z, Tong Z F, Dong L H, Li B 2020 Fuel 272 117738
 Google Scholar
Google Scholar
[24] Smith D, Chippendale T W E, Španěl P 2014 Rapid Commun. Mass Spectrom. 28 1917
 Google Scholar
Google Scholar
[25] Dias M, Gurusinghe R M, Suits A G 2022 J. Phys. Chem. A. 126 5354
 Google Scholar
Google Scholar
[26] Chase M W 1998 Journal of Physical and Chemical Reference Data Monograph (Vol. 9) (New York: National Institute of Standards and Technology Gaithersburg) p1604
[27] Dunham J L 1932 Phys. Rev. 41 721
 Google Scholar
Google Scholar
[28] Ilieva T, Iliev I 2016 J. Mol. Spectrosc. 330 28
 Google Scholar
Google Scholar
[29] Le Roy R J 2017 J. Quant. Spectrosc. Radiat. Transfer. 186 167
 Google Scholar
Google Scholar
[30] Schwabl F 2006 Statistical Mechanics (2nd Ed.) (Berlin Heidelberg: Springer-Verlag) p409
[31] Huber K P, Herzberg G 1950 Molecular Spectra and Molecular Structure: Spectra of Diatomic Molecules (New York: Van Nostrand Reinhold Company) p9
[32] Gordillo-Vázquez F J, Kunc J A 1998 J. Appl. Phys. 84 4693
 Google Scholar
Google Scholar
[33] Holbrook R, Kaledin L, Kunc J A 1993 Phys. Rev. E 47 1285
 Google Scholar
Google Scholar
[34] Holbrook R, Kunc J A 1994 Phys. Plasmas. 1 1075
 Google Scholar
Google Scholar
[35] Zhang J P, Cheng X L, Zhang H, Yang X D 2011 Chinese Phys. B. 20 060401
 Google Scholar
Google Scholar
[36] Reddy R R, Nazeer Ahammed Y, Baba Basha D, Narasimhulu K, Siva Sankar Reddy L, Rama Gopal K 2006 J. Quant. Spectrosc. Radiat. Transfer. 97 344
 Google Scholar
Google Scholar
[37] Barklem P S, Collet R 2016 Astron. Astrophys. 588 A96
 Google Scholar
Google Scholar
计量
- 文章访问数: 5833
- PDF下载量: 111
- 被引次数: 0














 下载:
下载: