-
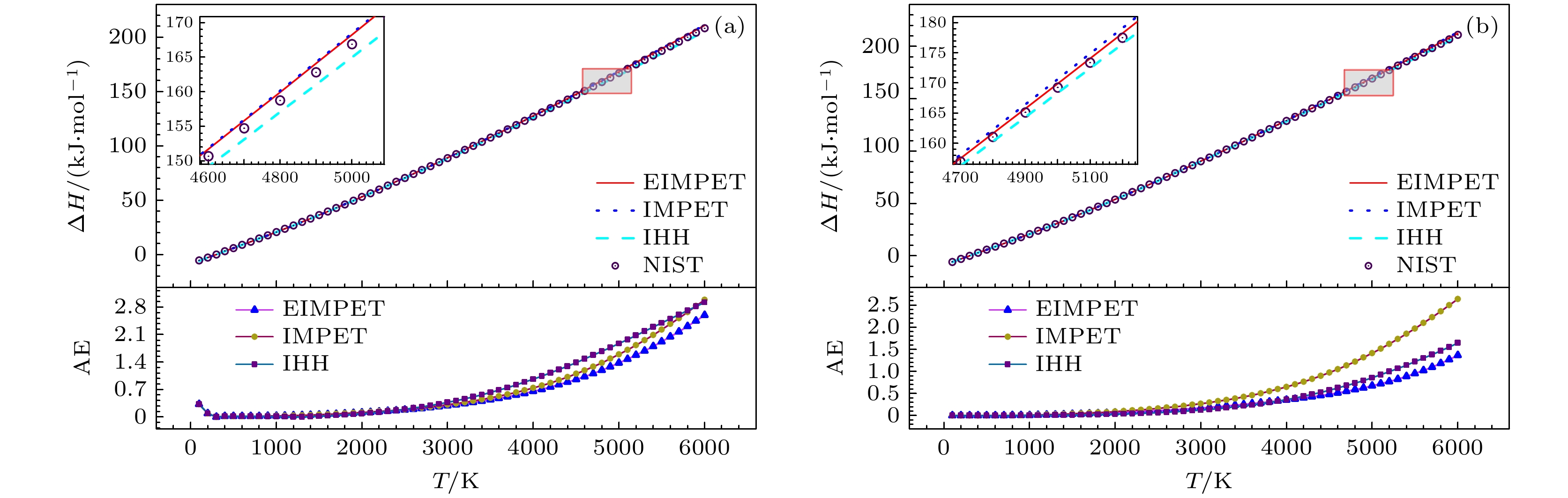
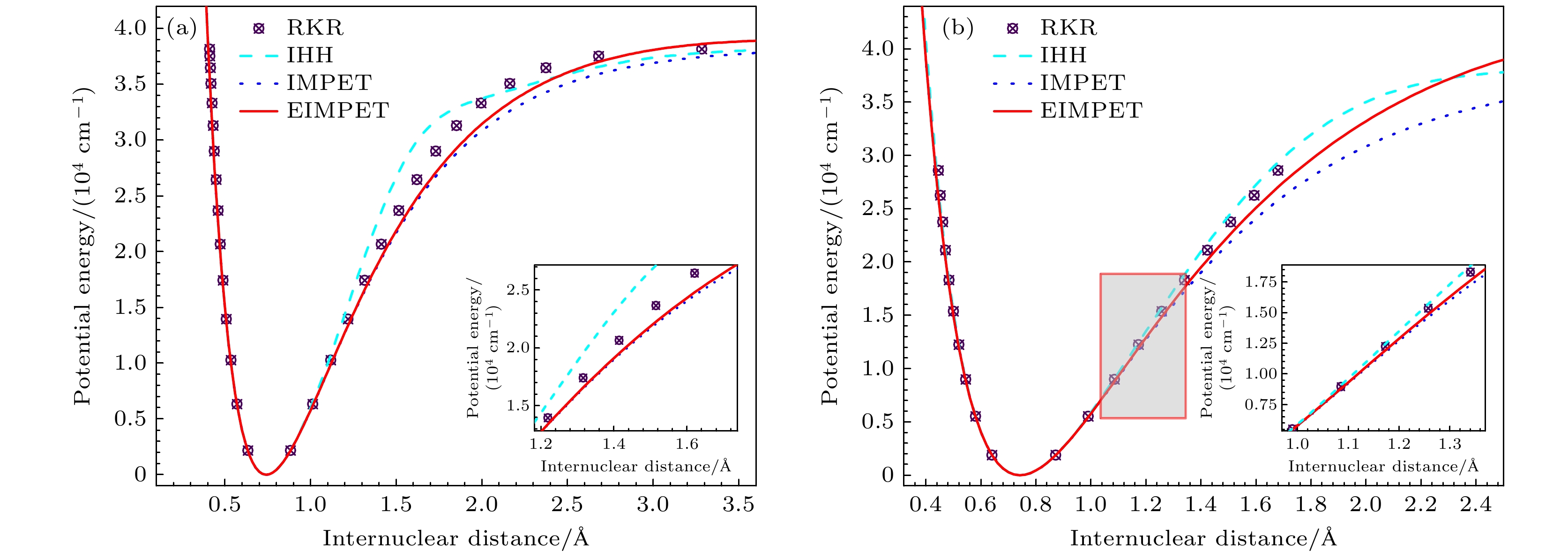
本文通过扩展的改进多参数指数型(the extended improved multiparameter exponential-type, EIMPET)势能模型, 结合实验光谱数据, 研究了H2和HD分子的热力学性质. 首先利用解析势能曲线计算得到分子的振转能级, 其次结合量子统计系综理论计算了分子在100—6000 K温度下的配分函数、摩尔热容、摩尔熵、摩尔焓以及约化摩尔吉布斯自由能. 计算结果与美国国家标准与技术研究所(National Institute of Standards and Technology, NIST)数据库中的数据具有良好的一致性. 本文的理论方法可用于预测某些气态物质的热力学性质.
-
关键词:
- H2分子 /
- HD分子 /
- 扩展的改进多参数指数型势能模型 /
- 热力学性质
H2 molecule and their isotopes represent one of the modern clean energy sources. It is imperative to understand their thermodynamic properties for comprehending their behaviors under various conditions. Thereby promoting their more in-depth applications. In this paper, an extended improved multiparameter exponential-type potential (EIMPET) combined with the quantum statistical ensemble theory is used to investigate and analyze the thermodynamic properties of H2 and HD molecules. Firstly, reliable energy level data for molecules are obtained using the EIMPET potential. Subsequently, the one-dimensional Schrödinger equation is solved with the LEVEL program to determine the rovibrational energy levels of the molecules. Finally, the quantum statistical ensemble theory is integrated to determine the partition functions, molar heat capacity, molar entropy, molar enthalpy, and reduced molar Gibbs free energy of H2 and HD in a temperature range of 100–6000 K. The calculation results indicate that compared with IHH potential and IMPET potential, the EIMPET potential is closer to RKR data. A comparison of the calculated thermodynamic properties of the molecules reveals that the results from the EIMPET potential-based method accord well with those from the NIST database. Specifically, for H2, the root mean square (RMS) errors for $ {C_{\text{m}}}\left( T \right) $, $ {S_{\text{m}}}\left( T \right) $, $ {G_{\text{r}}}\left( T \right) $, and $ \Delta {H_{\text{r}}}\left( T \right) $are 0.6894 J·K–1·mol–1, 0.3824 J·K–1·mol–1, 0.1754 J·K–1·mol–1, and 0.9586 kJ·mol–1, respectively, while for HD, the RMS errors are 0.3431 J·K–1·mol–1, 0.1443 J·K–1·mol–1, 0.0495 J·K–1·mol–1, and 0.4863 kJ·mol–1, respectively. All of these results are superior to those obtained using IMPET potential, and to those obtained using IHH potential as a whole. These findings demonstrate the advantages and practical applications of the EIMPET potential in calculating the thermodynamic properties of diatomic gas molecules, providing a foundation for subsequently studying the thermodynamic properties of triatomic molecules.-
Keywords:
- H2 molecule /
- HD molecule /
- the extended improved multiparameter exponential-type potential energy model /
- thermodynamic properties
[1] Wang C W, Peng X L, Liu J Y, et al. 2022 Int. J. Hydrogen Energy 47 27821
 Google Scholar
Google Scholar
[2] Fan X, Bañados E, Simcoe R A 2023 Annu. Rev. Astron. Astrophys. 61 373
 Google Scholar
Google Scholar
[3] Abramowitz S, Chase M W 1991 Pure Appl. Chem. 63 1449
 Google Scholar
Google Scholar
[4] Grein F 2023 Struct. Chem. 34 317
 Google Scholar
Google Scholar
[5] Yahiatène I, Hennig S, Huser T 2013 Chem. Phys. Lett. 587 1
 Google Scholar
Google Scholar
[6] Angelova M, Frank A 2005 Phys. At. Nucl. 68 1625
 Google Scholar
Google Scholar
[7] Halpern A M 2010 J. Chem. Educ. 87 174
 Google Scholar
Google Scholar
[8] Liu G Y, Sun W G, Liao B T 2015 Indian J. Phys. 89 1109
 Google Scholar
Google Scholar
[9] Jia C S, Zhang L H, Wang C W 2017 Chem. Phys. Lett. 667 211
 Google Scholar
Google Scholar
[10] Ding Q C, Jia C S, Liu J Z, Li J, Du R F, Liu J Y, Peng X L, Wang C W, Tang H X 2022 Chem. Phys. Lett. 803 139844
 Google Scholar
Google Scholar
[11] Jia C S, Wang C W, Zhang L H, Peng X L, Tang H M, Zeng R 2018 Chem. Eng. Sci. 183 26
 Google Scholar
Google Scholar
[12] Ikot A N, Chukwuocha E O, Onyeaju M C, Onate C A, Ita B I, Udoh M E 2018 Pramana-J. Phys. 90 22
 Google Scholar
Google Scholar
[13] Okorie U S, Ikot A N, Chukwuocha E O, Rampho G J 2020 Results Phys. 17 103078
 Google Scholar
Google Scholar
[14] Bakhti H, Diaf A, Hachama M 2020 Comput. Theor. Chem. 1185 112879
 Google Scholar
Google Scholar
[15] Oluwadare O J, Oyewumi K J, Abiola T O 2022 Indian J. Phys. 96 1921
 Google Scholar
Google Scholar
[16] Strekalov M L 2024 Chem. Phys. Impact 8 100444
 Google Scholar
Google Scholar
[17] Coveney P V, Wan S 2016 Phys. Chem. Chem. Phys. 18 30236
 Google Scholar
Google Scholar
[18] Fang Z, Vasiliu M, Peterson K A, Dixon D A 2017 J. Chem. Theory. Comput. 13 1057
 Google Scholar
Google Scholar
[19] Startsev A N 2019 J. Sulfur Chem. 40 435
 Google Scholar
Google Scholar
[20] van Speybroeck V, Gani R, Meier R J 2010 Chem. Soc. Rev. 39 1764
 Google Scholar
Google Scholar
[21] Kang D, Fan Q, Fan Z, Li H, Fu J 2024 Int. J. Quantum Chem. 124 e27373
 Google Scholar
Google Scholar
[22] National Institute of Standards and Technology (NIST), 2017 NIST Chemistry WebBook, NISTS Standard Reference Database Number 69. http://webbook.nist.gov/chemistry/
[23] Xie B J, Jia C S 2020 Int. J. Quantum Chem. 120 e26058
 Google Scholar
Google Scholar
[24] Morse P M 1929 Phys. Rev. 34 57
 Google Scholar
Google Scholar
[25] Desai A M, Mesquita N, Fernandes V 2020 Phys. Scr. 95 085401
 Google Scholar
Google Scholar
[26] Le Roy R J 2017 J. Quant. Spectrosc. Radiat. Transfer 186 167
 Google Scholar
Google Scholar
[27] Ding Q C, Jia C S, Wang C W, Peng X L, Liu J Y, Zhang L H, Jiang R, Zhu S Y, Yuan H, Tang H X 2023 J. Mol. Liq. 371 121088
 Google Scholar
Google Scholar
[28] Hooydonk G V http://hdl.handle.net/1854/LU-1212652 [2024- 12-18]
[29] Tobias I, Vanderslice J T 1961 J. Chem. Phys. 35 1852
 Google Scholar
Google Scholar
[30] Fink E H, Akins D L, Bradley Moore C 1969 Chem. Phys. Lett. 4 283
 Google Scholar
Google Scholar
[31] Wilkinson P G 1968 Can. J. Phys. 46 1225
 Google Scholar
Google Scholar
[32] Tian H, Fan Q, Fan Z, Fu J, Li H, Ma J, Xie F 2022 Int. J. Quantum Chem. 122 e26983
 Google Scholar
Google Scholar
[33] Leachman J W, Jacobsen R T, Penoncello S G, Lemmon E W 2009 J. Phys. Chem. Ref. Data 38 721
 Google Scholar
Google Scholar
-
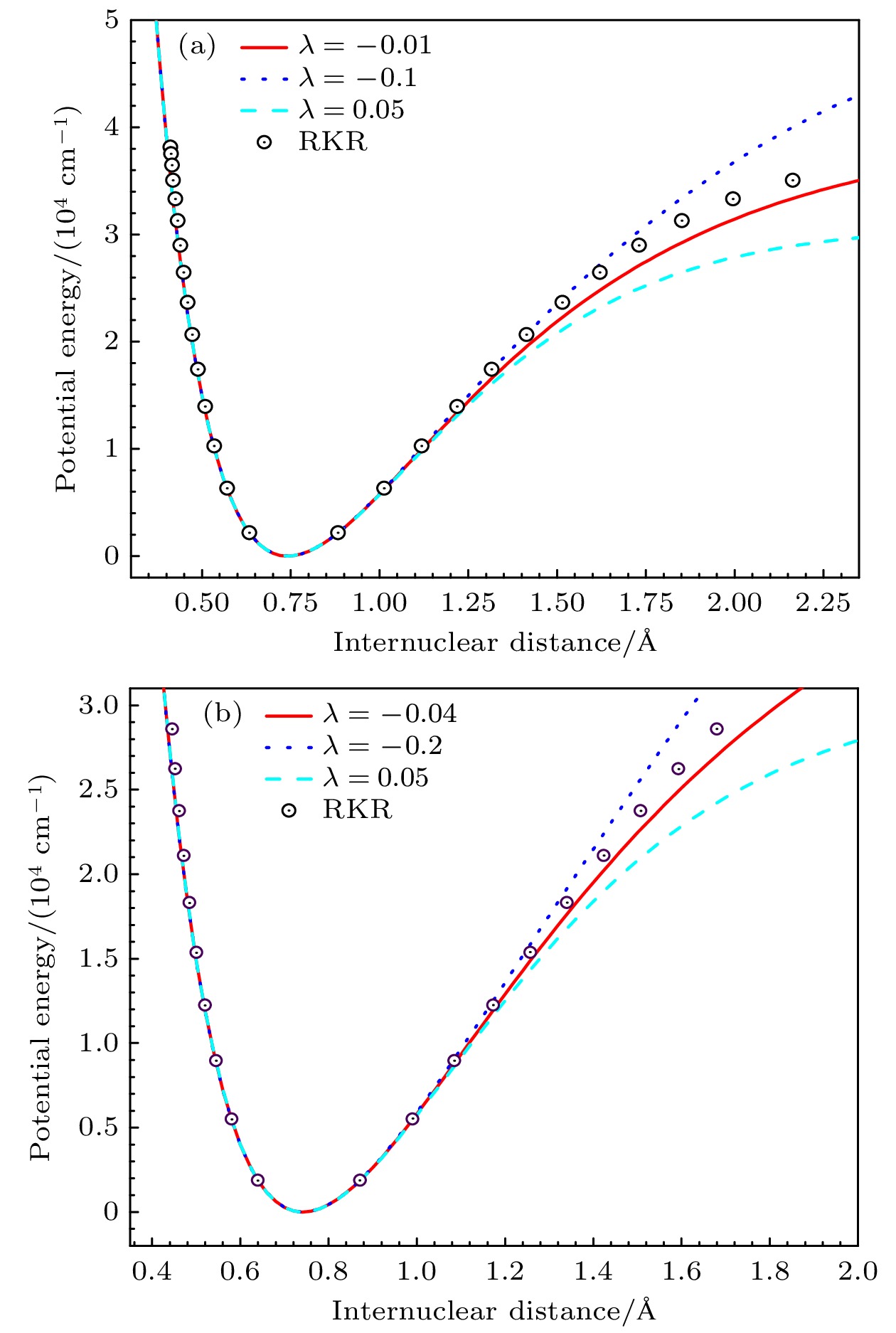
表 1 H2和HD分子电子基态的实验光谱常数和可调参数$ \lambda $的值
Table 1. Experimental spectral constants and adjustable parameter $ \lambda $ of the ground electronic states of H2 and HD molecules.
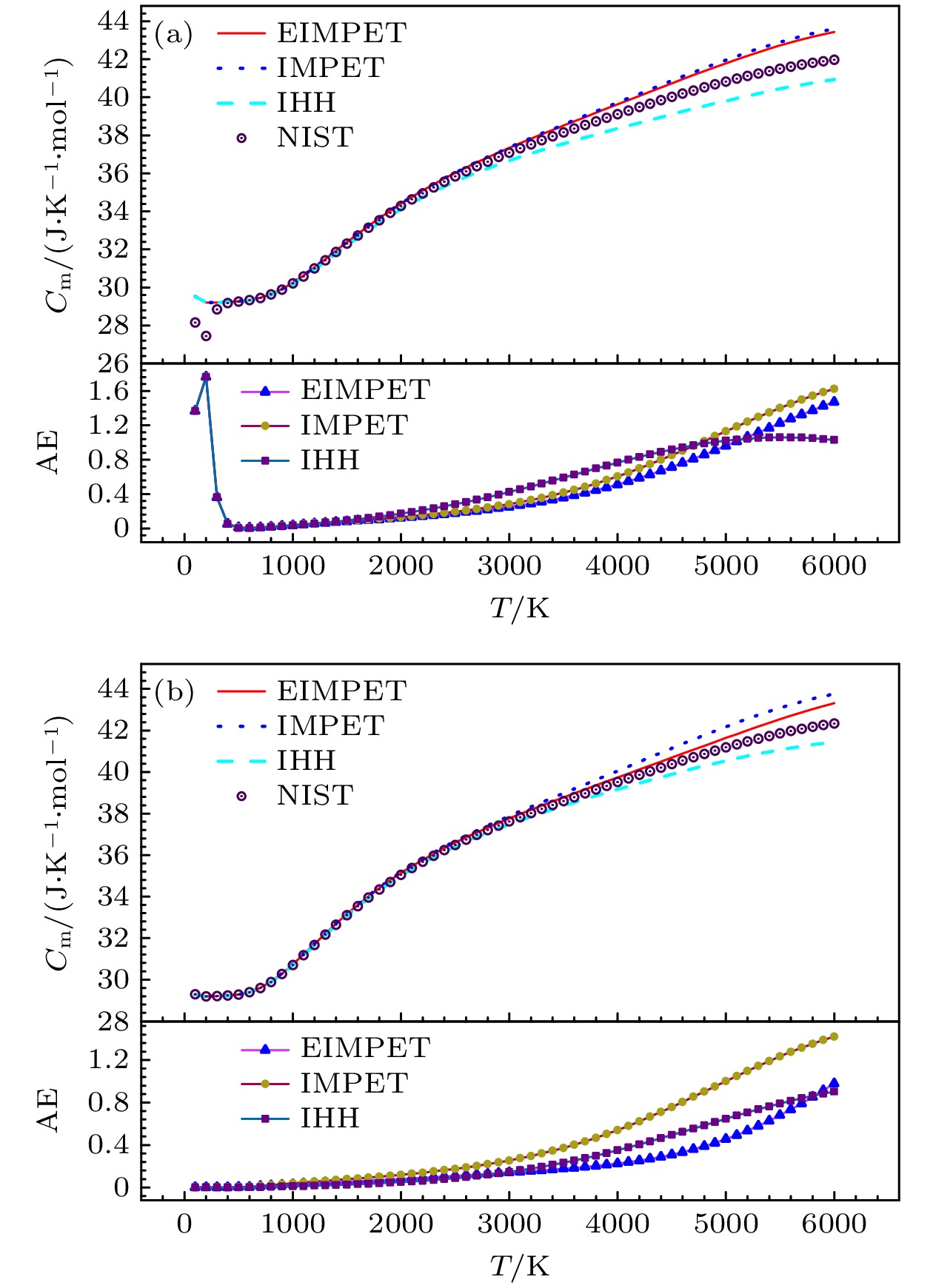
表 2 H2分子不同温度下的摩尔热容以及绝对误差
Table 2. Molar heat capacity of H2 molecule at different temperatures and absolute errors.
T/K $ {C}_{{\mathrm{m}}}^{{\mathrm{N}}{\mathrm{I}}{\mathrm{S}}{\mathrm{T}}} $
/(J·K–1·mol–1)$ {C}_{{\mathrm{m}}}^{{\mathrm{I}}{\mathrm{H}}{\mathrm{H}}} $
/(J·K–1·mol–1)$ {C}_{{\mathrm{m}}}^{{\mathrm{I}}{\mathrm{M}}{\mathrm{P}}{\mathrm{E}}{\mathrm{T}}} $
/(J·K–1·mol–1)$ {C}_{{\mathrm{m}}}^{{\mathrm{E}}{\mathrm{I}}{\mathrm{M}}{\mathrm{P}}{\mathrm{E}}{\mathrm{T}}} $
/(J·K–1·mol–1)$ \Delta {C}_{{\mathrm{m}}}^{{\mathrm{N}}{\mathrm{I}}{\mathrm{S}}{\mathrm{T}}\text{-}{\mathrm{I}}{\mathrm{H}}{\mathrm{H}}} $
/(J·K–1·mol–1)$ \Delta {C}_{{\mathrm{m}}}^{{\mathrm{N}}{\mathrm{I}}{\mathrm{S}}{\mathrm{T}}\text{-}{\mathrm{I}}{\mathrm{M}}{\mathrm{P}}{\mathrm{E}}{\mathrm{T}}} $
/(J·K–1·mol–1)$ \Delta {C}_{{\mathrm{m}}}^{{\mathrm{N}}{\mathrm{I}}{\mathrm{S}}{\mathrm{T}}\text{-}{\mathrm{E}}{\mathrm{I}}{\mathrm{M}}{\mathrm{P}}{\mathrm{E}}{\mathrm{T}}} $
/(J·K–1·mol–1)100 28.154 29.521 29.520 29.520 1.367 1.366 1.366 300 28.849 29.210 29.212 29.212 0.361 0.363 0.363 500 29.260 29.265 29.270 29.270 0.005 0.010 0.010 700 29.441 29.432 29.452 29.452 0.009 0.011 0.011 900 29.881 29.857 29.910 29.908 0.024 0.029 0.027 1100 30.581 30.535 30.629 30.626 0.046 0.048 0.045 1300 31.423 31.354 31.491 31.487 0.069 0.068 0.064 1500 32.298 32.204 32.384 32.378 0.094 0.086 0.080 1700 33.139 33.017 33.243 33.236 0.122 0.104 0.097 1900 33.917 33.763 34.040 34.031 0.154 0.123 0.114 2100 34.624 34.432 34.768 34.757 0.192 0.144 0.133 2300 35.263 35.029 35.430 35.416 0.234 0.167 0.153 2500 35.842 35.560 36.036 36.018 0.282 0.194 0.176 2700 36.370 36.036 36.595 36.573 0.334 0.225 0.203 2900 36.856 36.464 37.119 37.091 0.392 0.263 0.235 3100 37.311 36.856 37.616 37.581 0.455 0.305 0.270 3300 37.740 37.218 38.096 38.051 0.522 0.356 0.311 3500 38.149 37.558 38.565 38.508 0.591 0.416 0.359 3700 38.544 37.882 39.029 38.958 0.662 0.485 0.414 3900 38.928 38.196 39.491 39.405 0.732 0.563 0.477 4100 39.301 38.502 39.952 39.849 0.799 0.651 0.548 4300 39.665 38.803 40.412 40.292 0.862 0.747 0.627 4500 40.017 39.099 40.868 40.732 0.918 0.851 0.715 4700 40.355 39.388 41.316 41.164 0.967 0.961 0.809 4900 40.676 39.671 41.750 41.585 1.005 1.074 0.909 5100 40.976 39.942 42.163 41.988 1.034 1.187 1.012 5300 41.252 40.199 42.548 42.369 1.053 1.296 1.117 5500 41.498 40.439 42.900 42.721 1.059 1.402 1.223 5700 41.712 40.656 43.211 43.038 1.056 1.499 1.326 5900 41.890 40.849 43.476 43.315 1.041 1.586 1.425 6000 41.965 40.934 43.590 43.436 1.031 1.625 1.471 MAE — — — — 0.564 0.587 0.519 *$ \Delta {C}_{{\mathrm{m}}}^{{\mathrm{N}}{\mathrm{I}}{\mathrm{S}}{\mathrm{T}}\text{-}{\mathrm{m}}{\mathrm{o}}{\mathrm{d}}{\mathrm{e}}{\mathrm{l}}}=|{C}_{{\mathrm{m}}}^{{\mathrm{N}}{\mathrm{I}}{\mathrm{S}}{\mathrm{T}}}-{C}_{{\mathrm{m}}}^{{\mathrm{m}}{\mathrm{o}}{\mathrm{d}}{\mathrm{e}}{\mathrm{l}}}| $; MAE: mean absolute error 表 3 H2和HD分子不同势能模型下的预测数据与NIST数据的方均根误差
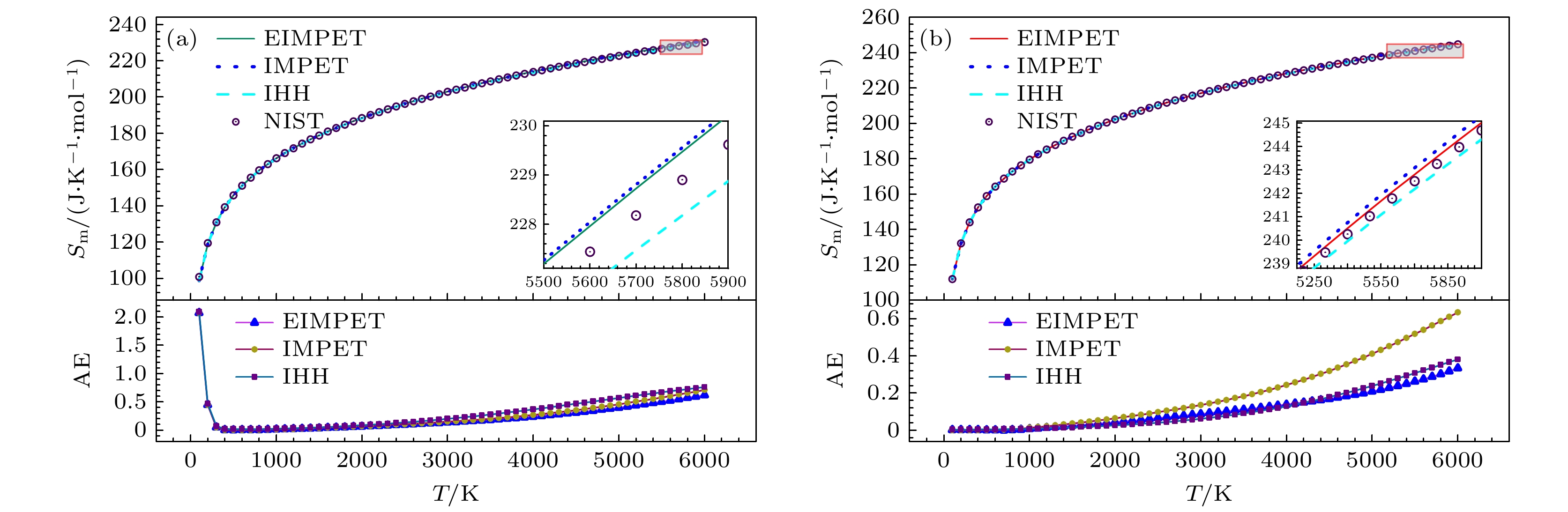
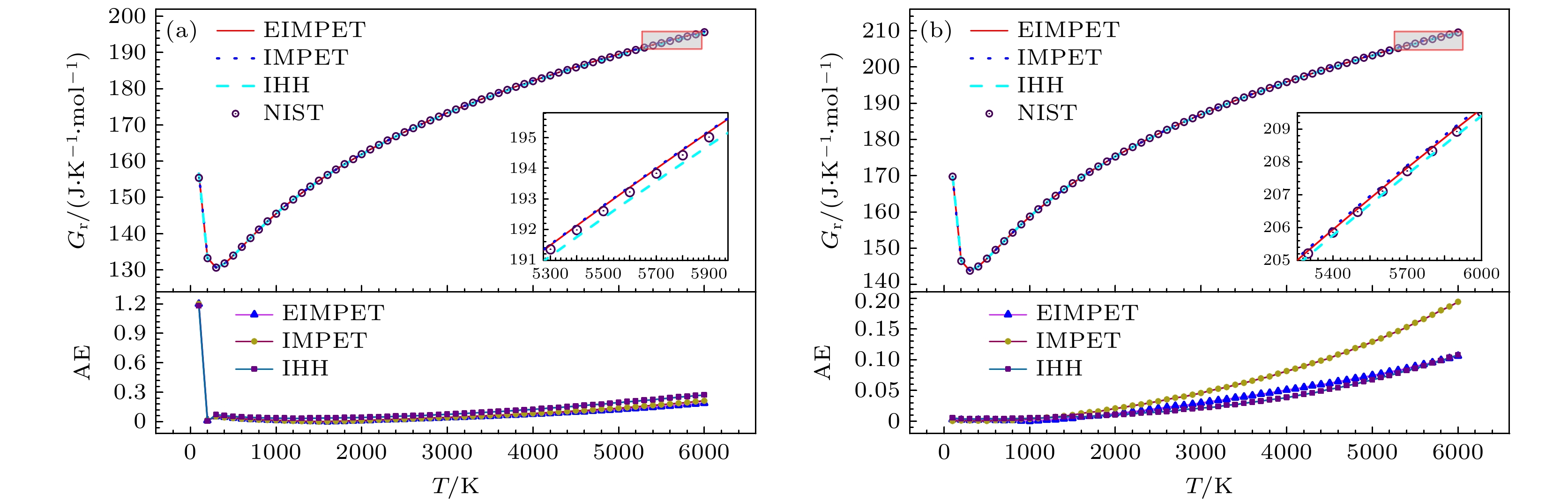
Table 3. Root mean square error of predicted data for H2 and HD molecules under different potential energy models compared to NIST data.
热力学量 H2EIMPET H2IMPET H2IHH HDEIMPET HDIMPET HDIHH Cm/(J·K–1·mol–1) 0.6894 0.7750 0.7019 0.3431 0.6338 0.4055 Sm/(J·K–1·mol–1) 0.3824 0.4096 0.4591 0.1443 0.2732 0.1575 ΔHr/(kJ·mol–1) 0.9586 1.1052 1.2110 0.4863 0.9749 0.5946 Gr/(J·K–1·mol–1) 0.1754 0.1805 0.2005 0.0495 0.0859 0.0452 -
[1] Wang C W, Peng X L, Liu J Y, et al. 2022 Int. J. Hydrogen Energy 47 27821
 Google Scholar
Google Scholar
[2] Fan X, Bañados E, Simcoe R A 2023 Annu. Rev. Astron. Astrophys. 61 373
 Google Scholar
Google Scholar
[3] Abramowitz S, Chase M W 1991 Pure Appl. Chem. 63 1449
 Google Scholar
Google Scholar
[4] Grein F 2023 Struct. Chem. 34 317
 Google Scholar
Google Scholar
[5] Yahiatène I, Hennig S, Huser T 2013 Chem. Phys. Lett. 587 1
 Google Scholar
Google Scholar
[6] Angelova M, Frank A 2005 Phys. At. Nucl. 68 1625
 Google Scholar
Google Scholar
[7] Halpern A M 2010 J. Chem. Educ. 87 174
 Google Scholar
Google Scholar
[8] Liu G Y, Sun W G, Liao B T 2015 Indian J. Phys. 89 1109
 Google Scholar
Google Scholar
[9] Jia C S, Zhang L H, Wang C W 2017 Chem. Phys. Lett. 667 211
 Google Scholar
Google Scholar
[10] Ding Q C, Jia C S, Liu J Z, Li J, Du R F, Liu J Y, Peng X L, Wang C W, Tang H X 2022 Chem. Phys. Lett. 803 139844
 Google Scholar
Google Scholar
[11] Jia C S, Wang C W, Zhang L H, Peng X L, Tang H M, Zeng R 2018 Chem. Eng. Sci. 183 26
 Google Scholar
Google Scholar
[12] Ikot A N, Chukwuocha E O, Onyeaju M C, Onate C A, Ita B I, Udoh M E 2018 Pramana-J. Phys. 90 22
 Google Scholar
Google Scholar
[13] Okorie U S, Ikot A N, Chukwuocha E O, Rampho G J 2020 Results Phys. 17 103078
 Google Scholar
Google Scholar
[14] Bakhti H, Diaf A, Hachama M 2020 Comput. Theor. Chem. 1185 112879
 Google Scholar
Google Scholar
[15] Oluwadare O J, Oyewumi K J, Abiola T O 2022 Indian J. Phys. 96 1921
 Google Scholar
Google Scholar
[16] Strekalov M L 2024 Chem. Phys. Impact 8 100444
 Google Scholar
Google Scholar
[17] Coveney P V, Wan S 2016 Phys. Chem. Chem. Phys. 18 30236
 Google Scholar
Google Scholar
[18] Fang Z, Vasiliu M, Peterson K A, Dixon D A 2017 J. Chem. Theory. Comput. 13 1057
 Google Scholar
Google Scholar
[19] Startsev A N 2019 J. Sulfur Chem. 40 435
 Google Scholar
Google Scholar
[20] van Speybroeck V, Gani R, Meier R J 2010 Chem. Soc. Rev. 39 1764
 Google Scholar
Google Scholar
[21] Kang D, Fan Q, Fan Z, Li H, Fu J 2024 Int. J. Quantum Chem. 124 e27373
 Google Scholar
Google Scholar
[22] National Institute of Standards and Technology (NIST), 2017 NIST Chemistry WebBook, NISTS Standard Reference Database Number 69. http://webbook.nist.gov/chemistry/
[23] Xie B J, Jia C S 2020 Int. J. Quantum Chem. 120 e26058
 Google Scholar
Google Scholar
[24] Morse P M 1929 Phys. Rev. 34 57
 Google Scholar
Google Scholar
[25] Desai A M, Mesquita N, Fernandes V 2020 Phys. Scr. 95 085401
 Google Scholar
Google Scholar
[26] Le Roy R J 2017 J. Quant. Spectrosc. Radiat. Transfer 186 167
 Google Scholar
Google Scholar
[27] Ding Q C, Jia C S, Wang C W, Peng X L, Liu J Y, Zhang L H, Jiang R, Zhu S Y, Yuan H, Tang H X 2023 J. Mol. Liq. 371 121088
 Google Scholar
Google Scholar
[28] Hooydonk G V http://hdl.handle.net/1854/LU-1212652 [2024- 12-18]
[29] Tobias I, Vanderslice J T 1961 J. Chem. Phys. 35 1852
 Google Scholar
Google Scholar
[30] Fink E H, Akins D L, Bradley Moore C 1969 Chem. Phys. Lett. 4 283
 Google Scholar
Google Scholar
[31] Wilkinson P G 1968 Can. J. Phys. 46 1225
 Google Scholar
Google Scholar
[32] Tian H, Fan Q, Fan Z, Fu J, Li H, Ma J, Xie F 2022 Int. J. Quantum Chem. 122 e26983
 Google Scholar
Google Scholar
[33] Leachman J W, Jacobsen R T, Penoncello S G, Lemmon E W 2009 J. Phys. Chem. Ref. Data 38 721
 Google Scholar
Google Scholar
计量
- 文章访问数: 2176
- PDF下载量: 35
- 被引次数: 0













 下载:
下载: