-
气体分子与壁面之间的相互作用是影响稀薄气体流动状态的主要因素, 但是由于其物理上的复杂性和微观性, 这一过程的机理并没有得到充分揭示. 本文利用分子束法对Ar分子在金属Pt表面的碰撞过程进行了分子动力学模拟, 并探究了入射速度、角度和壁面粗糙度对动量、能量转化机制的影响. 结果表明, 当气体分子以
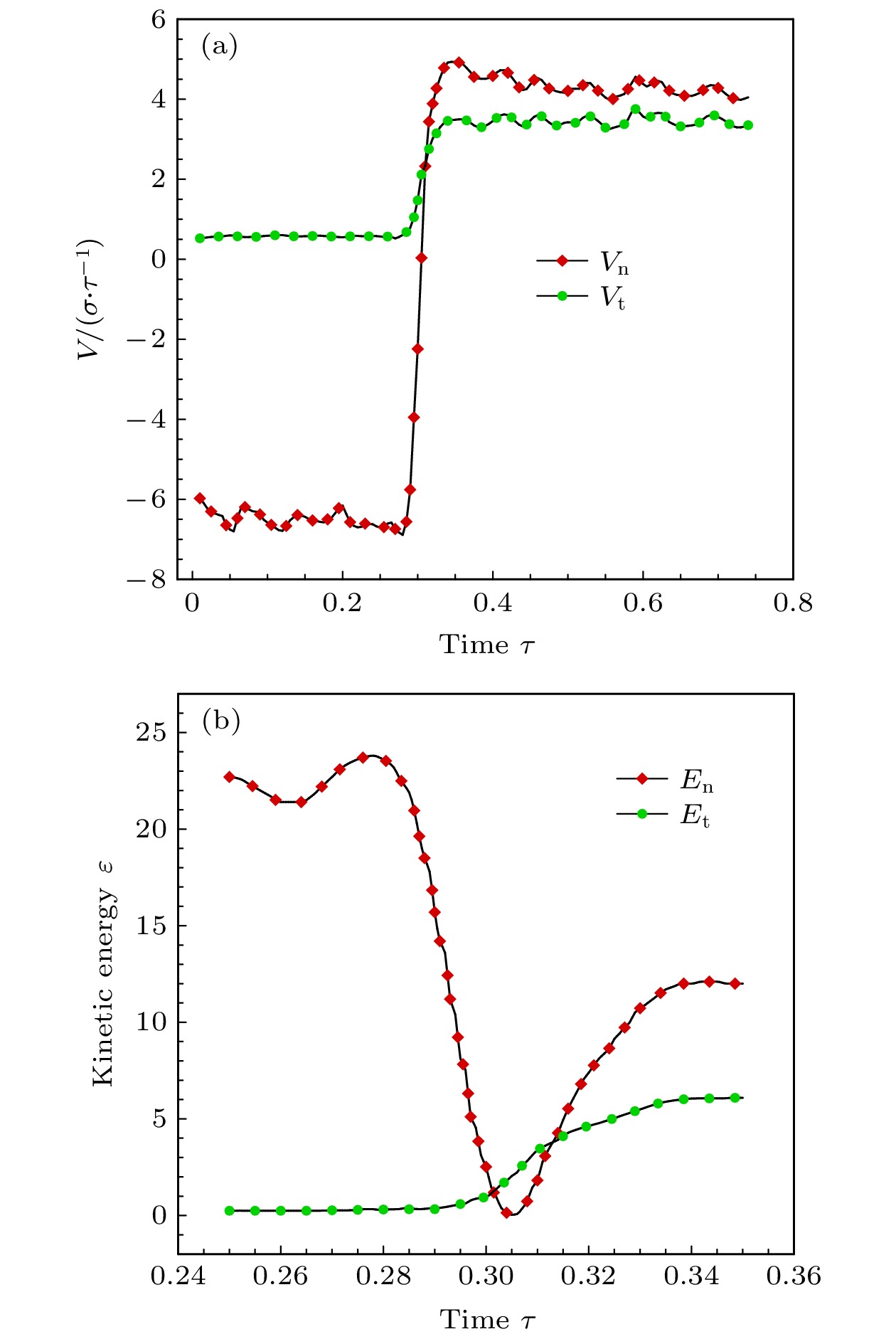
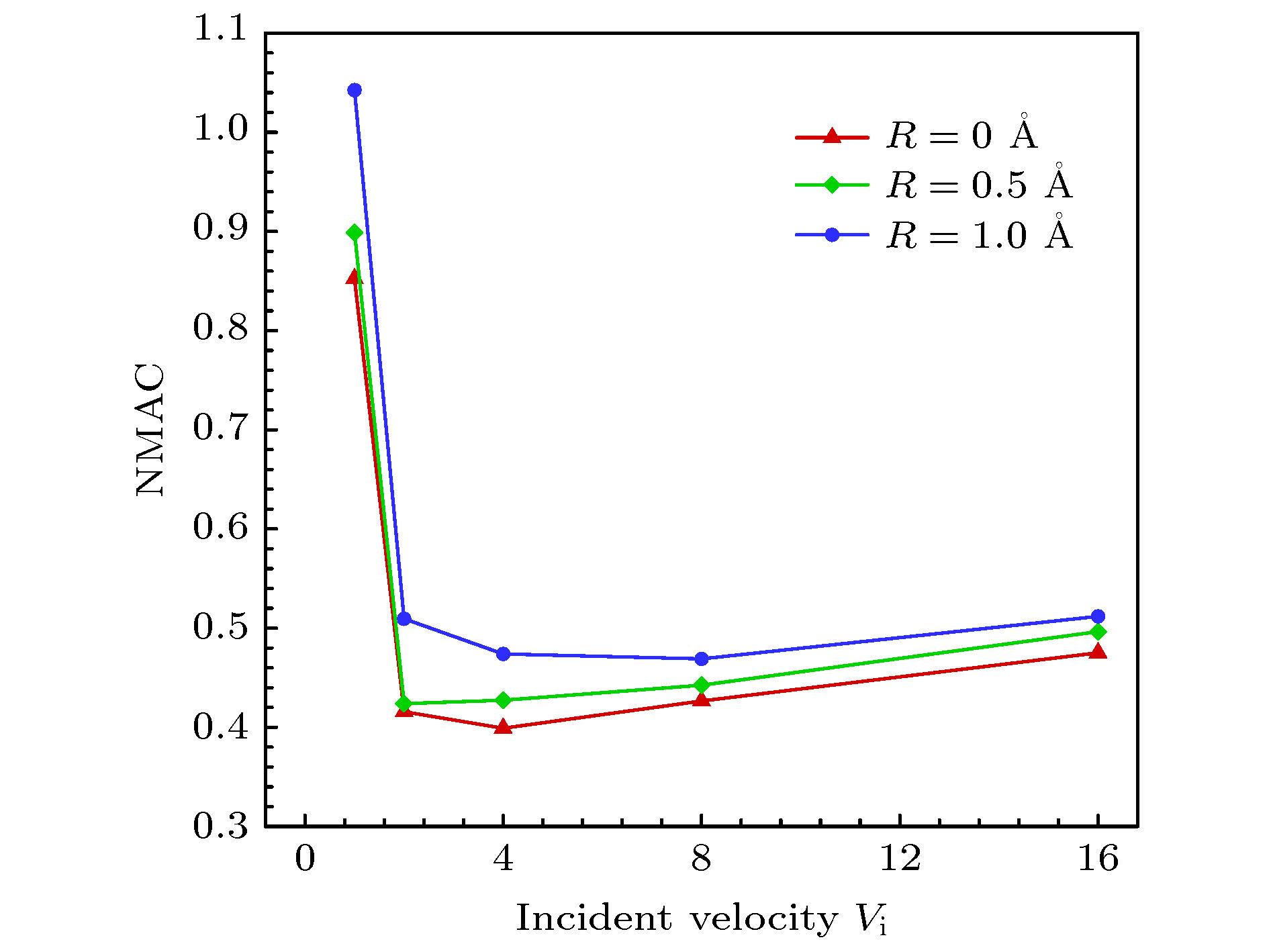
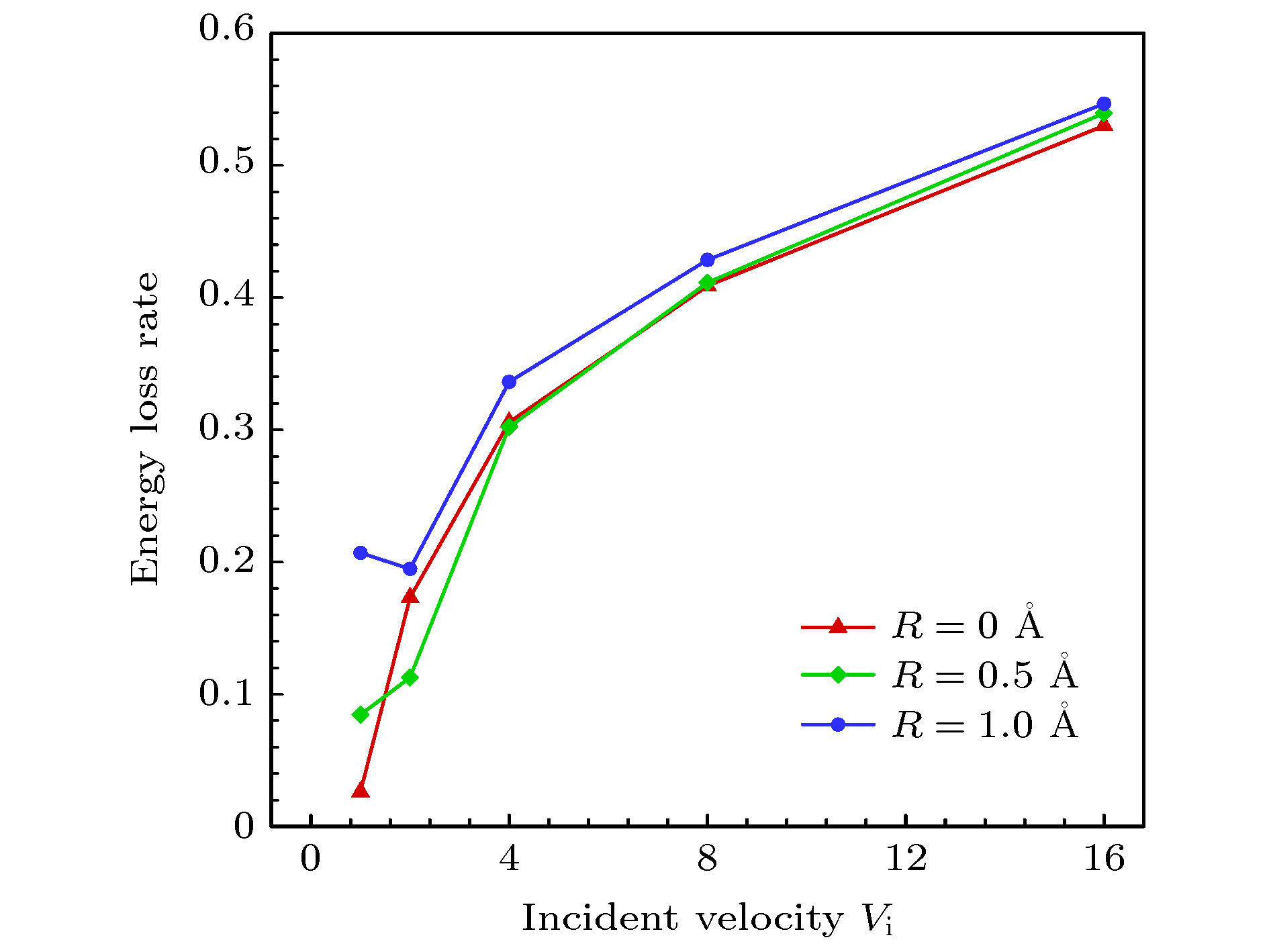
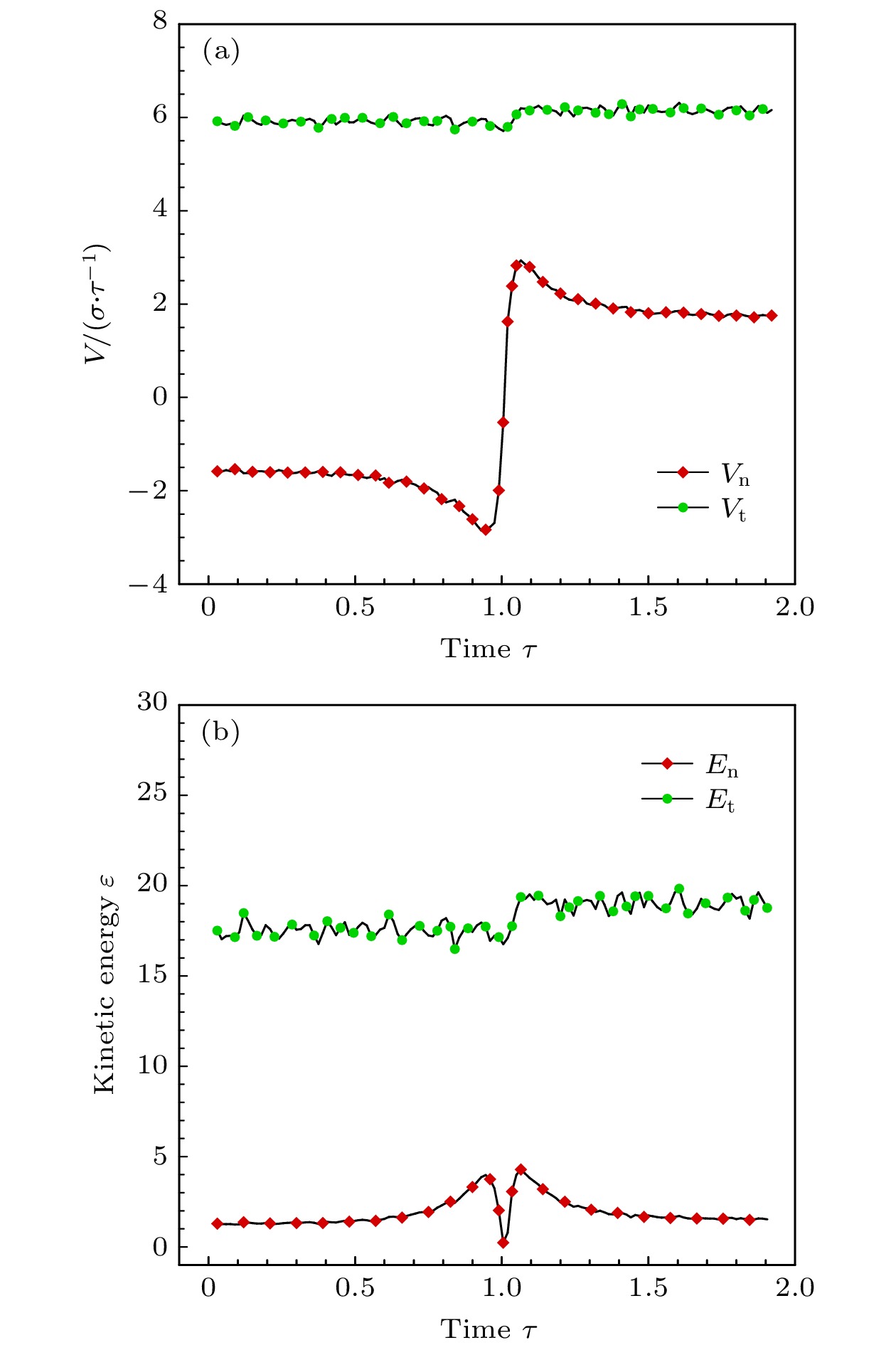
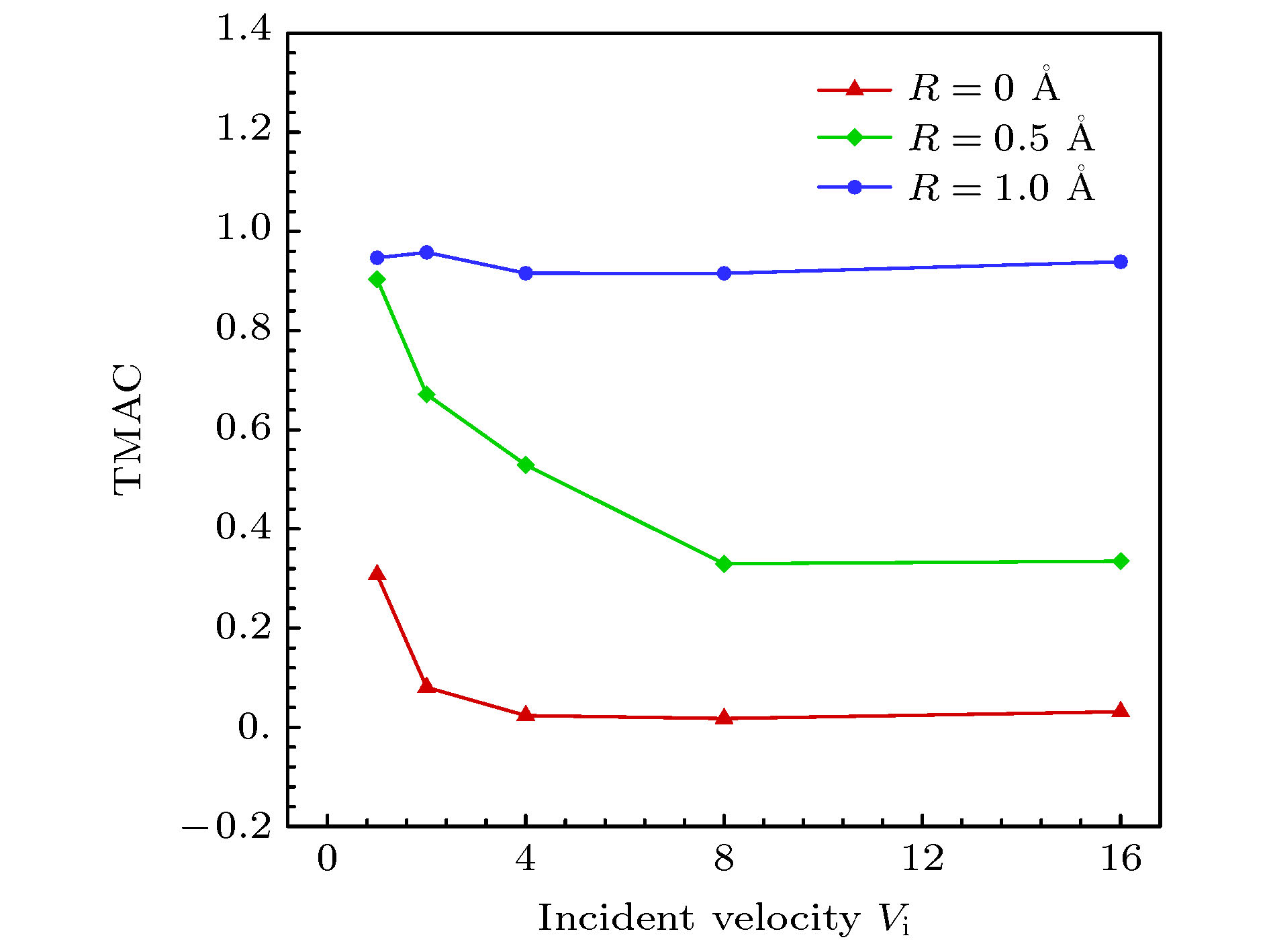
$ 5^{\circ}$ 的极角入射时, 分子的法向速度分量占主导因素, 在与壁面发生碰撞之后, 分子的切向和法向动量都会损失, 法向动能会向切向转移, 并且当分子速度不低于2.0时, 切向和法向动能的比值会稳定在一个很小的区间, 而粗糙度对动量和能量转化的影响不明显. 与小角度入射时不同, 当气体分子以$ 75^{\circ} $ 的极角与金属表面碰撞时, 粗糙度的影响就不能再被忽略了. 大极角入射的气体分子在光滑壁面散射之后, 其运动规律基本符合Maxwell所假设的镜面反射, 动量和能量分量的变化都不明显. 而粗糙度的引入则会促进气体分子切向动量和能量向法向转移, 并且会使分子总能量的损失更加显著.With the rapid development of micro-nano technology and aerospace technology, researches related to rarefied gas flows have received more and more attention. For micro-/nanoscale systems and spacecraft in a rarefied environment, the reduction in the frequency of intermolecular collisions in the flow field makes the interaction between gas molecules and the solid surface develop into a major factor affecting the flow state. However, the mechanism of gas-surface interaction in rarefied flow has not been fully revealed due to its microscopic nature and physical complexity, and the existing simulation methods cannot accurately reflect the effect of this process on the flow state. In this paper, molecular beam method is adopted to simulate the scattering process of argon molecules on platinum surface, and the impacts of incident velocity, angle and wall roughness on the momentum and energy conversion mechanism are explored. By simulating the molecular scattering process under the two incident angles of$ 5^{\circ} $ and$ 75^{\circ}$ , the following conclusions are obtained. When colliding with the wall at an angle close to vertical, both components of the momentum of the gas molecules are lost. The normal energy transfers to the tangential direction, and when the molecular velocity is not less than 2.0, the transfer rate is not significantly affected by the incident energy of the molecule and the surface roughness. The total energy loss of gas molecules after scattering becomes significant with the increase of incident velocity, and it is not sensitive to changes of surface roughness. When the gas molecules are incident at$ 75^{\circ} $ , the roughness of the surface has a significant impact on the conversion mechanism of molecular momentum and energy. After colliding with a smooth wall, the momentum and energy values of the gas molecules remain basically unchanged, only the direction of the momentum is reversed. The motion state of molecules is close to the mirror reflection, and the conversion between momentum and energy components is not obvious. The introduction of roughness enhances the degree of accommodation between gas molecules and metal surface, and promotes the transfer of molecular tangential momentum and kinetic energy to the normal direction. When incident at a large polar angle, as opposed to the small-angle cases, the total energy loss of molecules is not sensitive to changes of incident velocity, it goes up significantly with the surface roughness increasing. The research in this article not only explores the gas-surface interaction mechanism, but also provides a useful reference for the high-fidelity simulation of rare gas flow and the development of appropriate gas-surface interaction models.[1] Liang T, Li Q, Ye W 2017 J. Comput. Phys. 352 105
 Google Scholar
Google Scholar
[2] 曹炳阳, 陈民, 过增元 2006 物理学报 55 5305
 Google Scholar
Google Scholar
Cao B Y, Chen M, Guo Z Y 2006 Acta Phys. Sin. 55 5305
 Google Scholar
Google Scholar
[3] Sharipov F, Kalempa D 2005 Phys. Fluids 10 16
 Google Scholar
Google Scholar
[4] Burnett D 1935 Proc. Lond. Math. Soc. 1 1
 Google Scholar
Google Scholar
[5] Shavaliyev M S 1993 J. Appl. Math. Mech. 57 573
 Google Scholar
Google Scholar
[6] Bird G A 1994 Molecular Gas Dynamics and the Direct Simulation of Gas Flows (Oxford: Oxford University Press) p199
[7] Yuan Y D, Rahman S 2016 Phys. A 463 25
 Google Scholar
Google Scholar
[8] Lockerby D A, Reese J M, Emerson D R, Barber R W 2004 Phys. Rev. E 70 017303
 Google Scholar
Google Scholar
[9] Barber R W, Emerson D R 2006 Heat Transfer Eng. 27 3
 Google Scholar
Google Scholar
[10] Lockerby D A, Reese J M, Gallis M A 2005 AIAA J. 43 1391
 Google Scholar
Google Scholar
[11] Reese J M, Zheng Y, Lockerby D A 2007 J. Comput. Theor. Nanosci. 4 807
 Google Scholar
Google Scholar
[12] Bruno D, Cacciatore M, Longo S, Rutigliano M 2000 Chem. Phys. Lett. 320 245
 Google Scholar
Google Scholar
[13] Padilla, J F, Bo yd, Iain D 2009 J. Thermophys. Heat Transfer 23 96
 Google Scholar
Google Scholar
[14] Liang T, Li Q, Ye W 2013 Phys. Rev. E 88 013009
 Google Scholar
Google Scholar
[15] Woronowicz M S, Rault D F 1994 J. Spacecraft Rockets 31 532
 Google Scholar
Google Scholar
[16] Myong R S 2004 J. Comput. Phys. 195 655
 Google Scholar
Google Scholar
[17] Alder B J, Wainwright T E 1957 J. Chem. Phys. 27 1208
 Google Scholar
Google Scholar
[18] Kovalev V, Yakunchikov A, Li F 2011 Acta Astronaut. 69 744
 Google Scholar
Google Scholar
[19] 张忠强, 刘汉伦, 范晋伟, 丁建宁, 程广贵 2019 物理学报 68 170202
 Google Scholar
Google Scholar
Zhang Z Q, Liu H L, Fan J W, Ding J N, Cheng G G 2019 Acta Phys. Sin. 68 170202
 Google Scholar
Google Scholar
[20] Chirita V, Pailthorpe B A, Collins R E 1997 Nucl. Instrum. Meth. B 129 465
 Google Scholar
Google Scholar
[21] Yamanishi N, Matsumoto Y, Shobatake K 1999 Phys. Fluids 11 3540
 Google Scholar
Google Scholar
[22] 曹炳阳 2005 博士学位论文 (北京: 清华大学)
Cao B Y 2005 Ph. D. Dissertation (Beijing: Tsinghua University) (in Chinese)
[23] Finger G W, Kapat J S, Bhattacharya A 2007 J. Fluids Eng-Trans. ASME 129 31
 Google Scholar
Google Scholar
[24] Ozhgibesov M S, Leu T, Cheng C 2012 J. Mol. Graph. Model. 38 375
 Google Scholar
Google Scholar
[25] Ozhgibesov M S, Leu T, Cheng C 2013 J. Mol. Graph. Model. 45 45
 Google Scholar
Google Scholar
[26] Leu T, Cheng C, Ozhgibesov M S 2011 J. Mol. Graph. Model. 31 35
 Google Scholar
Google Scholar
[27] Bao F, Mao Z, Qiu L 2014 Int. J. Numer. Methods Heat Fluid Flow 24 1338
 Google Scholar
Google Scholar
[28] 宋诚谦 2014 博士学位论文 (合肥: 中国科技大学)
Song C Q 2014 Ph. D. Dissertation (Hefei: University of Science and Technology of China) (in Chinese)
[29] Cao B Y, Sun J, Chen M, Guo Z Y 2009 Int. J. Mol. Sci. 10 11
 Google Scholar
Google Scholar
[30] Sun J, Li Z 2008 Mol. Phys. 106 2325
 Google Scholar
Google Scholar
[31] Sun J, Li Z 2009 Mol. Simul. 35 228
 Google Scholar
Google Scholar
[32] 张冉, 常青, 李桦 2018 物理学报 67 223401
 Google Scholar
Google Scholar
Zhang R, Chang Q, Li H 2018 Acta Phys. Sin. 67 223401
 Google Scholar
Google Scholar
[33] Maruyama S 2000 Adv. Numer. Heat Transfer 2 189
 Google Scholar
Google Scholar
[34] Wu L, Bogy D B 2002 J. Tribol.-Trans. ASME 124 562
 Google Scholar
Google Scholar
[35] Swope W C, Andersen H C, Berens P H, Wilson K R 1982 J. Chem. Phys. 76 637
 Google Scholar
Google Scholar
[36] Dongari N, Zhang Y H, Reese J M 2012 AIP Conf. Proc. 1501 895
 Google Scholar
Google Scholar
[37] 张烨, 张冉, 赖剑奇, 李桦 2019 物理学报 68 124702
 Google Scholar
Google Scholar
Zhang Y, Zhang R, Lai J Q, Li H 2019 Acta Phys. Sin. 68 124702
 Google Scholar
Google Scholar
[38] Chirita V, Pailthorpe B A, Collins R E 1993 J. Phys. D: Appl. Phys. 26 133
 Google Scholar
Google Scholar
[39] Maxwell J C 1878 Proc. R. Soc. London 27 304
 Google Scholar
Google Scholar
-
表 1 入射极角为
$ 5^{\circ}$ 时, 不同速度的气体分子在不同表面上散射后的平均速度和能量Table 1. When the incident polar angle is
$ 5^{\circ}$ , the mean velocity and energy of gas molecules that with different velocities after scattering on different surfaces.${V_{\rm{i}}}$/$\sqrt {2{k_{\rm{B}}}T} $ States ${V_{\rm{t}}}$/($\sigma \cdot {\tau^{-1}}$) ${V_{\rm{n}}}$/($\sigma \cdot {\tau^{-1}}$) ${E_{{\rm{t}}}}$/$\varepsilon $ ${E_{{\rm{n}}}}$/$\varepsilon $ E/$\varepsilon $ 1.0 Incident 3.0566 –0.8191 4.6720 0.3355 5.0083 Re-R = 0 Å 2.1165 1.8812 3.1534 2.0912 5.2478 Re-R = 0.5 Å 0.2948 1.8122 2.2248 1.9670 4.1892 Re-R = 1.0 Å 0.1625 1.7772 1.9206 1.9334 3.8632 2.0 Incident 6.1133 –1.6382 18.692 1.3420 20.034 Re-R = 0 Å 5.6211 2.2725 16.724 3.1156 19.829 Re-R = 0.5 Å 2.0090 2.9777 9.8840 5.6781 15.547 Re-R = 1.0 Å 0.2579 3.3517 7.4644 6.7519 14.216 4.0 Incident 12.226 –3.2766 74.766 5.3679 80.135 Re-R = 0.0 Å 11.937 3.0014 72.802 5.1488 78.079 Re-R = 0.5 Å 5.7585 5.6367 34.796 18.909 62.501 Re-R = 1.0 Å 1.0305 6.6301 26.878 25.619 52.504 8.0 Incident 24.452 –6.5532 299.06 21.472 320.54 Re-R = 0 Å 24.021 6.0509 293.14 19.210 312.38 Re-R = 0.5 Å 16.386 9.6325 209.77 49.747 259.52 Re-R = 1.0 Å 2.0656 12.770 117.64 95.714 213.20 16.0 Incident 48.906 –13.106 1196.3 85.880 1282.1 Re-R = 0 Å 47.378 12.256 1143.2 79.050 1220.8 Re-R = 0.5 Å 32.525 16.818 832.86 177.59 1009.5 Re-R = 1.0 Å 2.9900 23.156 474.64 318.22 790.68 表 2 入射极角为
$ 75^{\circ}$ 时, 不同速度的气体分子在不同表面上散射后的平均速度和能量Table 2. When the incident polar angle is
$ 75^{\circ} $ , the mean velocity and energy of gas molecules that with different velocities after scattering on different surfaces.${V_{\rm{i}}}$/$\sqrt {2{k_{\rm{B}}}T} $ States ${V_{\rm{t}}}$/($\sigma \cdot {\tau^{-1} }$) ${V_{\rm{n}}}$/($\sigma \cdot {\tau^{-1}}$) ${E_{{\rm{t}}}}$/$\varepsilon$ ${E_{{\rm{n}}}}$/$\varepsilon $ E/$\varepsilon $ 1.0 Incident 3.0566 –0.8191 4.6720 0.3355 5.0083 Re-R = 0 Å 2.1165 1.8812 3.1534 2.0912 5.2478 Re-R = 0.5 Å 0.2948 1.8122 2.2248 1.9670 4.1892 Re-R = 1.0 Å 0.1625 1.7772 1.9206 1.9334 3.8632 2.0 Incident 6.1133 –1.6382 18.692 1.3420 20.034 Re-R = 0 Å 5.6211 2.2725 16.724 3.1156 19.829 Re-R = 0.5 Å 2.0090 2.9777 9.8840 5.6781 15.547 Re-R = 1.0 Å 0.2579 3.3517 7.4644 6.7519 14.216 4.0 Incident 12.226 –3.2766 74.766 5.3679 80.135 Re-R = 0.0 Å 11.937 3.0014 72.802 5.1488 78.079 Re-R = 0.5 Å 5.7585 5.6367 34.796 18.909 62.501 Re-R = 1.0 Å 1.0305 6.6301 26.878 25.619 52.504 8.0 Incident 24.452 –6.5532 299.06 21.472 320.54 Re-R = 0 Å 24.021 6.0509 293.14 19.210 312.38 Re-R = 0.5 Å 16.386 9.6325 209.77 49.747 259.52 Re-R = 1.0 Å 2.0656 12.770 117.64 95.714 213.20 16.0 Incident 48.906 –13.106 1196.3 85.880 1282.1 Re-R = 0 Å 47.378 12.256 1143.2 79.050 1220.8 Re-R = 0.5 Å 32.525 16.818 832.86 177.59 1009.5 Re-R = 1.0 Å 2.9900 23.156 474.64 318.22 790.68 -
[1] Liang T, Li Q, Ye W 2017 J. Comput. Phys. 352 105
 Google Scholar
Google Scholar
[2] 曹炳阳, 陈民, 过增元 2006 物理学报 55 5305
 Google Scholar
Google Scholar
Cao B Y, Chen M, Guo Z Y 2006 Acta Phys. Sin. 55 5305
 Google Scholar
Google Scholar
[3] Sharipov F, Kalempa D 2005 Phys. Fluids 10 16
 Google Scholar
Google Scholar
[4] Burnett D 1935 Proc. Lond. Math. Soc. 1 1
 Google Scholar
Google Scholar
[5] Shavaliyev M S 1993 J. Appl. Math. Mech. 57 573
 Google Scholar
Google Scholar
[6] Bird G A 1994 Molecular Gas Dynamics and the Direct Simulation of Gas Flows (Oxford: Oxford University Press) p199
[7] Yuan Y D, Rahman S 2016 Phys. A 463 25
 Google Scholar
Google Scholar
[8] Lockerby D A, Reese J M, Emerson D R, Barber R W 2004 Phys. Rev. E 70 017303
 Google Scholar
Google Scholar
[9] Barber R W, Emerson D R 2006 Heat Transfer Eng. 27 3
 Google Scholar
Google Scholar
[10] Lockerby D A, Reese J M, Gallis M A 2005 AIAA J. 43 1391
 Google Scholar
Google Scholar
[11] Reese J M, Zheng Y, Lockerby D A 2007 J. Comput. Theor. Nanosci. 4 807
 Google Scholar
Google Scholar
[12] Bruno D, Cacciatore M, Longo S, Rutigliano M 2000 Chem. Phys. Lett. 320 245
 Google Scholar
Google Scholar
[13] Padilla, J F, Bo yd, Iain D 2009 J. Thermophys. Heat Transfer 23 96
 Google Scholar
Google Scholar
[14] Liang T, Li Q, Ye W 2013 Phys. Rev. E 88 013009
 Google Scholar
Google Scholar
[15] Woronowicz M S, Rault D F 1994 J. Spacecraft Rockets 31 532
 Google Scholar
Google Scholar
[16] Myong R S 2004 J. Comput. Phys. 195 655
 Google Scholar
Google Scholar
[17] Alder B J, Wainwright T E 1957 J. Chem. Phys. 27 1208
 Google Scholar
Google Scholar
[18] Kovalev V, Yakunchikov A, Li F 2011 Acta Astronaut. 69 744
 Google Scholar
Google Scholar
[19] 张忠强, 刘汉伦, 范晋伟, 丁建宁, 程广贵 2019 物理学报 68 170202
 Google Scholar
Google Scholar
Zhang Z Q, Liu H L, Fan J W, Ding J N, Cheng G G 2019 Acta Phys. Sin. 68 170202
 Google Scholar
Google Scholar
[20] Chirita V, Pailthorpe B A, Collins R E 1997 Nucl. Instrum. Meth. B 129 465
 Google Scholar
Google Scholar
[21] Yamanishi N, Matsumoto Y, Shobatake K 1999 Phys. Fluids 11 3540
 Google Scholar
Google Scholar
[22] 曹炳阳 2005 博士学位论文 (北京: 清华大学)
Cao B Y 2005 Ph. D. Dissertation (Beijing: Tsinghua University) (in Chinese)
[23] Finger G W, Kapat J S, Bhattacharya A 2007 J. Fluids Eng-Trans. ASME 129 31
 Google Scholar
Google Scholar
[24] Ozhgibesov M S, Leu T, Cheng C 2012 J. Mol. Graph. Model. 38 375
 Google Scholar
Google Scholar
[25] Ozhgibesov M S, Leu T, Cheng C 2013 J. Mol. Graph. Model. 45 45
 Google Scholar
Google Scholar
[26] Leu T, Cheng C, Ozhgibesov M S 2011 J. Mol. Graph. Model. 31 35
 Google Scholar
Google Scholar
[27] Bao F, Mao Z, Qiu L 2014 Int. J. Numer. Methods Heat Fluid Flow 24 1338
 Google Scholar
Google Scholar
[28] 宋诚谦 2014 博士学位论文 (合肥: 中国科技大学)
Song C Q 2014 Ph. D. Dissertation (Hefei: University of Science and Technology of China) (in Chinese)
[29] Cao B Y, Sun J, Chen M, Guo Z Y 2009 Int. J. Mol. Sci. 10 11
 Google Scholar
Google Scholar
[30] Sun J, Li Z 2008 Mol. Phys. 106 2325
 Google Scholar
Google Scholar
[31] Sun J, Li Z 2009 Mol. Simul. 35 228
 Google Scholar
Google Scholar
[32] 张冉, 常青, 李桦 2018 物理学报 67 223401
 Google Scholar
Google Scholar
Zhang R, Chang Q, Li H 2018 Acta Phys. Sin. 67 223401
 Google Scholar
Google Scholar
[33] Maruyama S 2000 Adv. Numer. Heat Transfer 2 189
 Google Scholar
Google Scholar
[34] Wu L, Bogy D B 2002 J. Tribol.-Trans. ASME 124 562
 Google Scholar
Google Scholar
[35] Swope W C, Andersen H C, Berens P H, Wilson K R 1982 J. Chem. Phys. 76 637
 Google Scholar
Google Scholar
[36] Dongari N, Zhang Y H, Reese J M 2012 AIP Conf. Proc. 1501 895
 Google Scholar
Google Scholar
[37] 张烨, 张冉, 赖剑奇, 李桦 2019 物理学报 68 124702
 Google Scholar
Google Scholar
Zhang Y, Zhang R, Lai J Q, Li H 2019 Acta Phys. Sin. 68 124702
 Google Scholar
Google Scholar
[38] Chirita V, Pailthorpe B A, Collins R E 1993 J. Phys. D: Appl. Phys. 26 133
 Google Scholar
Google Scholar
[39] Maxwell J C 1878 Proc. R. Soc. London 27 304
 Google Scholar
Google Scholar
计量
- 文章访问数: 8680
- PDF下载量: 113
- 被引次数: 0



















 下载:
下载: