-
析氧反应 (OER) 在锌空气电池、燃料电池和电解水等能源储存和转换设备中都有至关重要的作用. 然而OER过程涉及四电子转移, 导致反应动力学缓慢. 尽管贵金属氧化物被认为是最先进的OER电催化剂, 但昂贵的价格以及稀缺性限制了其商业应用. 因此, 本工作结合水热和水浴法制备了NaCu5S3@NixFe-LDH (x = 1, 2, 3, 4) 纳米片阵列复合电催化剂. 对样品的结构进行了表征, 结果显示NaCu5S3和Ni2Fe-LDH充分混合, 形成紧密结合的界面, 有利于电荷的快速转移, 这将增强两相界面处的电子调控作用, 改变其局域结构特性, 促进OER电催化性能. 电化学测试结果显示, 当电流密度为20 mA·cm–2时, NaCu5S3@Ni2Fe-LDH在1.0 M KOH电解液中的氧析出过电位仅为227 mV, 电催化性能优于原始的NaCu5S3 (271 mV) 和Ni2Fe-LDH (275 mV), 并且具有长达72 h的稳定性. 此外, NaCu5S3@Ni2Fe-LDH复合电催化剂具有较小的电荷转移电阻, 较大的双层电容值 (10.0 mF·cm–2) 和电化学活性表面积 (250 cm2), 有利于OER的进行. 本工作为设计廉价、高效且稳定的OER复合电催化剂提供了切实可行的实践路径.The oxygen evolution reaction (OER) plays a critical role in energy storage and conversion devices such as zinc-air batteries, fuel cells, and electrolysis water. However, the OER process involves a four-electron transfer, leading to slow reaction kinetics. Therefore, it is necessary to explore an efficient, inexpensive, and durable electrocatalysts to accelerate the OER process. Noble metal oxides are considered the most advanced OER electrocatalysts, but their high price and scarcity limit their commercial applications. Thus, researchers have started exploring other low-cost materials as alternatives. Nanocomposite materials have emerged as a promising alternative to expensive and scarce noble metal oxide electrocatalysts for OER. Therefore, this work synthesizes novel nanocomposite materials, NaCu5S3@NixFe-LDH (x = 1, 2, 3, 4) nanosheet array via hydrothermal and water bath methods. The structure and morphology of each product are characterized, indicating a tightly integrated interface between NaCu5S3 and Ni2Fe-LDH, which facilitates rapid charge transfer and enhancement of electron regulation at the interface. This changes the local structure characteristics and promotes the OER catalytic performance. Electrochemical characterization results show that in a 1.0 M KOH electrolyte, the overpotential of NaCu5S3@Ni2Fe-LDH for OER at a current density of 20 mA/cm2 is only 227 mV, significantly lower than that of the original NaCu5S3 (271 mV) and Ni2Fe-LDH (275 mV), with stability duration reaching 72 h. Electrochemical results also reveal that with the increase of overpotential, NaCu5S3@Ni2Fe-LDH shows a significant oxidation peak between 1.35–1.45 (V vs. RHE), which leads to the activation of Ni2+ to Ni3+ high oxidation state. The high oxidation state of Ni will promote the OER. The NaCu5S3@Ni2Fe-LDH composite electrocatalyst exhibits lower charge transfer resistance, higher double layer capacitance value (10.0 mF/cm2), and electrochemical active surface area (250 cm2), which are also beneficial to promoting OER. This study highlights the potential of nanocomposite materials as cost-effective alternatives to noble metal oxide electrocatalysts for OER. The NaCu5S3@Ni2Fe-LDH composite electrocatalyst exhibits excellent OER performance with a low overpotential, high stability, and favorable electrochemical properties. This research provides a valuable insight into the design and development of efficient and sustainable electrocatalysts for energy conversion and storage applications.
-
Keywords:
- oxygen evolution reaction /
- electrocatalysis /
- composite electrocatalyst /
- nanosheet array
[1] Zhang Z H, Wang C L, Ma X L, Liu F, Xiao H, Zhang J, Lin Z, Hao Z P 2021 Small 17 2103785
 Google Scholar
Google Scholar
[2] Zhao X H, Pattengale B, Fan D H, Zou Z H, Zhao Y Q, Du J, Huang J E, Xu C L 2018 ACS Energy Lett. 3 2520
 Google Scholar
Google Scholar
[3] Zhao X, Zheng X R, Lu Q, Li Y, Xiao F P, Tang B, Wang S X, Yu D Y W, Rogach A L 2023 EcoMat. 5 e12293
 Google Scholar
Google Scholar
[4] Song J J, Wei C, Huang Z F, Liu C T, Zeng L, Wang X, Xu Z C 2020 J. Chem. Soc. Rev. 49 2196
 Google Scholar
Google Scholar
[5] Gao J J, Xu C Q, Hung S F, Liu W, Cai W Z, Zeng Z P, Jia C M, Chen H M, Xiao H, Li J, Huang Y Q, Liu B 2019 J. Am. Chem. Soc. 141 3014
 Google Scholar
Google Scholar
[6] 孙涛, 袁健美 2023 物理学报 72 028901
 Google Scholar
Google Scholar
Sun T, Yuan J M 2023 Acta Phys. Sin. 72 028901
 Google Scholar
Google Scholar
[7] 汤衍浩 2023 物理学报 72 027802
 Google Scholar
Google Scholar
Tang Y H 2023 Acta Phys. Sin. 72 027802
 Google Scholar
Google Scholar
[8] She Z W, Kibsgaard J, Dickens C F, Chorkendorff I B, Norskov J K, Jaramillo T F 2017 Science 355 4998
 Google Scholar
Google Scholar
[9] Guo Y N, Park T, Yi J W, Henzie J, Kim J, Wang Z L, Jiang B, Bando Y, Sugahara Y, Tang J, Yamauchi Y 2019 Adv. Mater. 31 1807134
 Google Scholar
Google Scholar
[10] Chia X, Eng A Y S, Ambrosi A, Tan S M, Pumera M 2015 Chem. Rev. 115 11941
 Google Scholar
Google Scholar
[11] Zheng Y, Jiao Y, Jaroniec M, Qiao S Z 2015 Angew. Chem. Int. Ed. 54 52
 Google Scholar
Google Scholar
[12] 李雨芃, 汤秀章, 陈欣南, 高春宇, 陈雁南, 范澄军, 吕建友 2023 物理学报 72 029501
 Google Scholar
Google Scholar
Li Y P, Tang X Z, Chen X N, Gao C Y, Chen Y N, Fan C J, Lü J Y 2023 Acta Phys. Sin. 72 029501
 Google Scholar
Google Scholar
[13] Deng S J, Shen Y B, Xie D, Lu Y F, Yu X L, Yang L, Wang X L, Xia X H, Tu J P 2019 J. Energy Chem. 39 61
 Google Scholar
Google Scholar
[14] Liu G M, Schulmeyer T, Brötz J, Klein A, Jaegermann W 2003 Thin Solid Films 431 477
 Google Scholar
Google Scholar
[15] 邓晨华, 于忠海, 王宇涛, 孔森, 周超, 杨森 2023 物理学报 72 027501
 Google Scholar
Google Scholar
Deng C H, Yu Z H, Wang Y T, Kong S, Zhou C, Yang S 2023 Acta Phys. Sin. 72 027501
 Google Scholar
Google Scholar
[16] Zhao J, Zhang J J, Li Z Y, Bu X H 2020 Small 16 2003916
 Google Scholar
Google Scholar
[17] Lü L, Yang Z X, Chen K, Wang C D, Xiong Y J 2019 Adv. Energy Mater. 9 1803358
 Google Scholar
Google Scholar
[18] Huang Z N, Liao X P, Zhang W B, Hu J L, Gao Q S 2022 ACS Catal. 12 13951
 Google Scholar
Google Scholar
[19] Lin X J, Cao S F, Chen H Y, Chen X D, Wang Z J, Zhou S N, Xu H, Liu S Y, Wei S X, Lu X Q 2022 Chem. Engine. J. 433 133524
 Google Scholar
Google Scholar
[20] Song S Z, Mu L H, Jiang Y, Sun J, Zhang Y, Shi G S, Sun H N 2022 ACS Appl. Mater. Inter. 14 47560
 Google Scholar
Google Scholar
[21] Li D, Qin Y Y, Liu J, Zhao H Y, Sun Z J, Chen G B, Wu D Y, Su Y Q, Ding S J, Xiao C H V 2022 Adv. Funct. Mater. 32 2107056
 Google Scholar
Google Scholar
[22] Lv L, He X B, Wang J S, Ruan Y J, Yang S X, Yuan H, Zhang T R 2021 Appl. Catal. B 298 120531
 Google Scholar
Google Scholar
[23] Gu Z X, Yang N, Han P, Kuang M, Mei B B, Jiang Z, Zhong J, Li L, Zheng G F 2019 Small Methods 3 1800449
 Google Scholar
Google Scholar
[24] Du C F, Dinh K N, Liang Q H, Zheng Y, Luo Y B, Zhang J L, Yan Q Y 2018 Adv. Energy Mater. 8 1801127
 Google Scholar
Google Scholar
[25] Dinh K N, Sun Y X, Pei Z X, Yuan Z W, Suwardi A, Huang Q W, Liao X Z, Wang Z G, Chen Y, Yan Q Y 2020 Small 16 1905885
 Google Scholar
Google Scholar
[26] Liu M J, Min K A, Han B C, Lee L Y S 2021 Adv. Energy Mater. 11 2101281
 Google Scholar
Google Scholar
[27] Li A, Zhang Z, Feng J, Lü F, Li Y, Wang R, Lu M, Gupta R B, Xi P, Zhang S 2018 J. Am. Chem. Soc. 140 17624
 Google Scholar
Google Scholar
[28] Xie Q X, Ren D, Bai L C, Ge R L, Zhou W H, Bai L, Xie W, Wang J H, Grätzel M, Luo J S 2023 Chin. J. Catalysis 44 127
 Google Scholar
Google Scholar
[29] Li S, Chen B B, Wang Y, Ye M Y, Aken P A V, Cheng C, Thomas A 2021 Nat. Mater. 20 1240
 Google Scholar
Google Scholar
[30] Wan K, Luo J S, Zhou C, Zhang T, Arbiol J, Lu X H, Mao B W, Zhang X, Fransaer J 2019 Adv. Funct. Mater. 29 1900315
 Google Scholar
Google Scholar
[31] Bai Y K, Wu Y, Zhou X C, Ye Y F, Nie K Q, Wang J, Xie M, Zhang Z X, Liu Z J, Cheng T, Gao C B 2022 Nat. Commun. 13 6094
 Google Scholar
Google Scholar
[32] Zhu J L, Qian J M, Peng X B, Xia B R, Gao D Q 2023 Nano-Micro Lett. 15 30
 Google Scholar
Google Scholar
[33] Chakraborty B, Kalra S, Beltrán-Suito R, Das C, Hellmann T, Menezes W P, Driess M 2020 Chem. Asian J. 15 852
 Google Scholar
Google Scholar
[34] Liang H J, Shuang W, Zhang Y T, Chao S J, Han H J, Wang X B, Zhang H, Yang L 2018 Chem. Electro. Chem. 5 494
 Google Scholar
Google Scholar
[35] Li Y M, Zhang X Y, Zhuo S Y, Liu S L, Han A X, Li L G, Tian Y 2021 Appl. Surf. Sci. 555 149441
 Google Scholar
Google Scholar
[36] He L B, Zhou D, Lin Y, Ge R X, Hou X D, Sun X P, Zheng C B 2018 ACS Catal. 8 3859
 Google Scholar
Google Scholar
[37] Chinnadurai D, Rajendiran R, Kandasamy P 2022 J. Colloid Inter. Sci. 606 101
 Google Scholar
Google Scholar
[38] Tan L, Yu J T, Wang C, Wang H F, Liu X, Gao H T, Xin L T, Liu D Z, Hou W G, Zhan T R 2022 Adv. Funct. Mater. 32 2200951
[39] Sun S, Zhou X, Cong B, Hong W, Chen G 2020 ACS Catal. 10 9086
 Google Scholar
Google Scholar
[40] Zhang J, Wang T, Pohl D, Rellinghaus B, Dong R H, Liu S H, Zhuang X D, Feng X L 2016 Angew. Chem. Int. Ed. 55 6702
 Google Scholar
Google Scholar
[41] Zhao Z L, Wu H X, He H L, Xu X L, Jin Y D 2014 Adv. Funct. Mater. 24 4698
 Google Scholar
Google Scholar
[42] Li Y, Chen G, Zhu Y, Hu Z, Chan T, She S, Dai J, Zhou W, Shao Z 2021 Adv. Funct. Mater. 31 2103569
 Google Scholar
Google Scholar
-
图 2 (a)—(e) NaCu5S3, NaCu5S3@NiFe-LDH, NaCu5S3@Ni2Fe-LDH, NaCu5S3@Ni3Fe-LDH和NaCu5S3@Ni4Fe-LDH的SEM图像; (f)—(i) NaCu5S3和NaCu5S3@Ni2Fe-LDH的TEM和HRTEM图像; (j), (k) NaCu5S3和NaCu5S3@Ni2Fe-LDH的EDS能谱图
Fig. 2. (a)–(e) SEM image of the NaCu5S3, NaCu5S3@NiFe-LDH, NaCu5S3@Ni2Fe-LDH, NaCu5S3@Ni3Fe-LDH and NaCu5S3@Ni4Fe-LDH; (f)–(i) TEM and HRTEM images of the NaCu5S3 and NaCu5S3@Ni2Fe-LDH; (j), (k) EDS images for the NaCu5S3 and NaCu5S3@Ni2Fe-LDH.
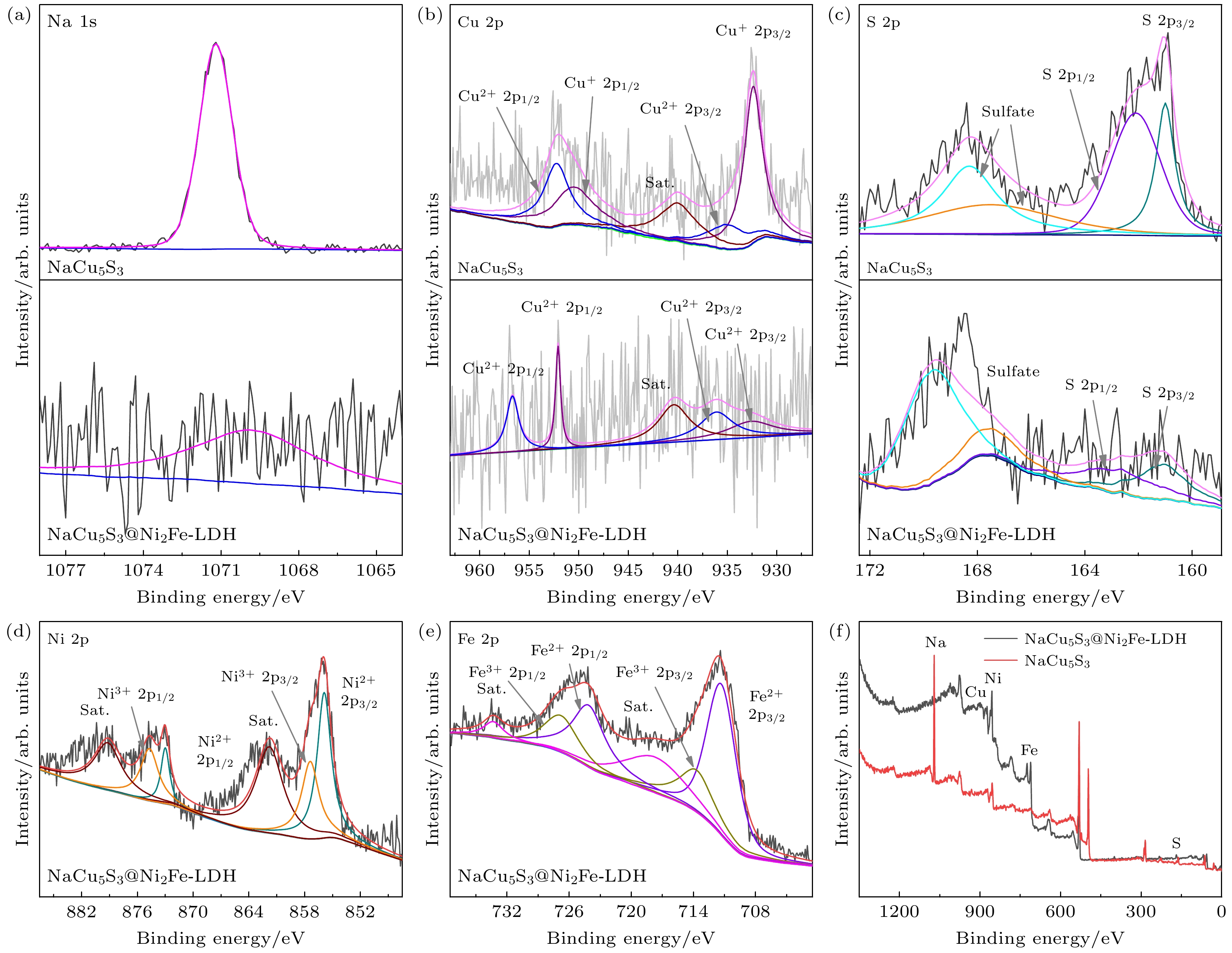
图 3 (a)—(c) NaCu5S3和NaCu5S3@Ni2Fe-LDH Na 1s, Cu 2p和S 2p XPS能谱; (d), (e) NaCu5S3@Ni2Fe-LDH Ni 2p和Fe 2p XPS能谱; (f) NaCu5S3和NaCu5S3@Ni2Fe-LDH XPS能谱
Fig. 3. (a)–(c) Na 1s, Cu 2p and S 2p XPS spectra of NaCu5S3 and NaCu5S3@Ni2Fe-LDH; (d), (e) Ni 2p and Fe 2p XPS spectra of the NaCu5S3@Ni2Fe-LDH; (f) XPS spectra of NaCu5S3 and NaCu5S3@Ni2Fe-LDH.
图 4 NaCu5S3@NiFe-LDH, NaCu5S3@Ni2Fe-LDH, NaCu5S3@Ni3Fe-LDH, NaCu5S3@Ni4Fe-LDH, NaCu5S3和Ni2Fe-LDH (a) LSV极化曲线; (b) Tafel斜率; (c) EIS; (d) Cdl; (e) ECSA; (f) NaCu5S3@Ni2Fe-LDH CP曲线
Fig. 4. NaCu5S3@NiFe-LDH, NaCu5S3@Ni2Fe-LDH, NaCu5S3@Ni3Fe-LDH, NaCu5S3@Ni4Fe-LDH, NaCu5S3 and Ni2Fe-LDH: (a) LSV polarization curves; (b) Tafel slope; (c) EIS; (d) Cdl; (e) ECSA; (f) CP curve of NaCu5S3@Ni2Fe-LDH.
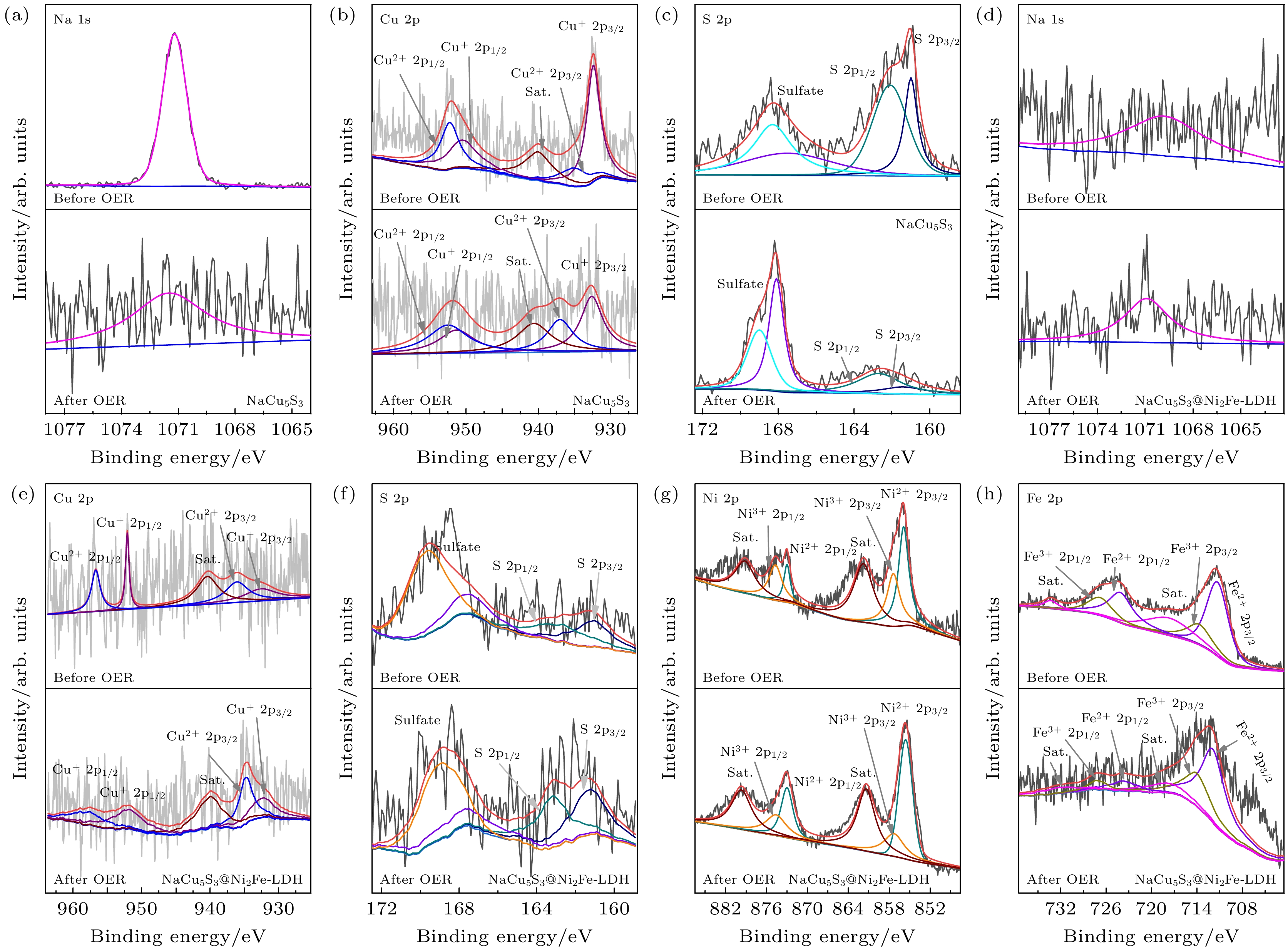
图 5 (a)—(c) NaCu5S3 OER前后的Na 1s, Cu 2p和S 2p XPS能谱; (d)—(h) NaCu5S3@Ni2Fe-LDH OER前后的Na 1s, Cu 2p, S 2p, Ni 2p和Fe 2p XPS能谱
Fig. 5. (a)–(c) Na 1s, Cu 2p and S 2p XPS spectra of the NaCu5S3 before and after OER; (d)–(h) Na 1s, Cu 2p, S 2p, Ni 2p and Fe 2p XPS spectra of the NaCu5S3@Ni2Fe-LDH before and after OER.
表 1 1 M KOH电解液中催化剂的OER活性比较
Table 1. Comparison of OER activity of catalysts in 1 M KOH electrolytes.
Catalyst Electrolyte Overpotential/mV Current density
/(mA·cm–2)Ref. NaCu5S3@NiFe-LDH 1 M KOH 254 20 This work NaCu5S3@Ni2Fe-LDH 1 M KOH 227 20 This work NaCu5S3@Ni3Fe-LDH 1 M KOH 248 20 This work NaCu5S3@Ni4Fe-LDH 1 M KOH 259 20 This work NaCu5S3 1 M KOH 271 20 This work Cu9S5/NF 1 M KOH 298 10 [33] CuS-FSM 1 M KOH 408 10 [34] CoO@Cu2S 1 M KOH 277 10 [35] Cu2S/CF 1 M KOH 336 20 [36] CuNiS 1 M KOH 337 10 [37] Cu-NiS2 1 M KOH 232 10 [25] Cu2S/TiO2/Cu2S 1 M KOH 284 10 [13] -
[1] Zhang Z H, Wang C L, Ma X L, Liu F, Xiao H, Zhang J, Lin Z, Hao Z P 2021 Small 17 2103785
 Google Scholar
Google Scholar
[2] Zhao X H, Pattengale B, Fan D H, Zou Z H, Zhao Y Q, Du J, Huang J E, Xu C L 2018 ACS Energy Lett. 3 2520
 Google Scholar
Google Scholar
[3] Zhao X, Zheng X R, Lu Q, Li Y, Xiao F P, Tang B, Wang S X, Yu D Y W, Rogach A L 2023 EcoMat. 5 e12293
 Google Scholar
Google Scholar
[4] Song J J, Wei C, Huang Z F, Liu C T, Zeng L, Wang X, Xu Z C 2020 J. Chem. Soc. Rev. 49 2196
 Google Scholar
Google Scholar
[5] Gao J J, Xu C Q, Hung S F, Liu W, Cai W Z, Zeng Z P, Jia C M, Chen H M, Xiao H, Li J, Huang Y Q, Liu B 2019 J. Am. Chem. Soc. 141 3014
 Google Scholar
Google Scholar
[6] 孙涛, 袁健美 2023 物理学报 72 028901
 Google Scholar
Google Scholar
Sun T, Yuan J M 2023 Acta Phys. Sin. 72 028901
 Google Scholar
Google Scholar
[7] 汤衍浩 2023 物理学报 72 027802
 Google Scholar
Google Scholar
Tang Y H 2023 Acta Phys. Sin. 72 027802
 Google Scholar
Google Scholar
[8] She Z W, Kibsgaard J, Dickens C F, Chorkendorff I B, Norskov J K, Jaramillo T F 2017 Science 355 4998
 Google Scholar
Google Scholar
[9] Guo Y N, Park T, Yi J W, Henzie J, Kim J, Wang Z L, Jiang B, Bando Y, Sugahara Y, Tang J, Yamauchi Y 2019 Adv. Mater. 31 1807134
 Google Scholar
Google Scholar
[10] Chia X, Eng A Y S, Ambrosi A, Tan S M, Pumera M 2015 Chem. Rev. 115 11941
 Google Scholar
Google Scholar
[11] Zheng Y, Jiao Y, Jaroniec M, Qiao S Z 2015 Angew. Chem. Int. Ed. 54 52
 Google Scholar
Google Scholar
[12] 李雨芃, 汤秀章, 陈欣南, 高春宇, 陈雁南, 范澄军, 吕建友 2023 物理学报 72 029501
 Google Scholar
Google Scholar
Li Y P, Tang X Z, Chen X N, Gao C Y, Chen Y N, Fan C J, Lü J Y 2023 Acta Phys. Sin. 72 029501
 Google Scholar
Google Scholar
[13] Deng S J, Shen Y B, Xie D, Lu Y F, Yu X L, Yang L, Wang X L, Xia X H, Tu J P 2019 J. Energy Chem. 39 61
 Google Scholar
Google Scholar
[14] Liu G M, Schulmeyer T, Brötz J, Klein A, Jaegermann W 2003 Thin Solid Films 431 477
 Google Scholar
Google Scholar
[15] 邓晨华, 于忠海, 王宇涛, 孔森, 周超, 杨森 2023 物理学报 72 027501
 Google Scholar
Google Scholar
Deng C H, Yu Z H, Wang Y T, Kong S, Zhou C, Yang S 2023 Acta Phys. Sin. 72 027501
 Google Scholar
Google Scholar
[16] Zhao J, Zhang J J, Li Z Y, Bu X H 2020 Small 16 2003916
 Google Scholar
Google Scholar
[17] Lü L, Yang Z X, Chen K, Wang C D, Xiong Y J 2019 Adv. Energy Mater. 9 1803358
 Google Scholar
Google Scholar
[18] Huang Z N, Liao X P, Zhang W B, Hu J L, Gao Q S 2022 ACS Catal. 12 13951
 Google Scholar
Google Scholar
[19] Lin X J, Cao S F, Chen H Y, Chen X D, Wang Z J, Zhou S N, Xu H, Liu S Y, Wei S X, Lu X Q 2022 Chem. Engine. J. 433 133524
 Google Scholar
Google Scholar
[20] Song S Z, Mu L H, Jiang Y, Sun J, Zhang Y, Shi G S, Sun H N 2022 ACS Appl. Mater. Inter. 14 47560
 Google Scholar
Google Scholar
[21] Li D, Qin Y Y, Liu J, Zhao H Y, Sun Z J, Chen G B, Wu D Y, Su Y Q, Ding S J, Xiao C H V 2022 Adv. Funct. Mater. 32 2107056
 Google Scholar
Google Scholar
[22] Lv L, He X B, Wang J S, Ruan Y J, Yang S X, Yuan H, Zhang T R 2021 Appl. Catal. B 298 120531
 Google Scholar
Google Scholar
[23] Gu Z X, Yang N, Han P, Kuang M, Mei B B, Jiang Z, Zhong J, Li L, Zheng G F 2019 Small Methods 3 1800449
 Google Scholar
Google Scholar
[24] Du C F, Dinh K N, Liang Q H, Zheng Y, Luo Y B, Zhang J L, Yan Q Y 2018 Adv. Energy Mater. 8 1801127
 Google Scholar
Google Scholar
[25] Dinh K N, Sun Y X, Pei Z X, Yuan Z W, Suwardi A, Huang Q W, Liao X Z, Wang Z G, Chen Y, Yan Q Y 2020 Small 16 1905885
 Google Scholar
Google Scholar
[26] Liu M J, Min K A, Han B C, Lee L Y S 2021 Adv. Energy Mater. 11 2101281
 Google Scholar
Google Scholar
[27] Li A, Zhang Z, Feng J, Lü F, Li Y, Wang R, Lu M, Gupta R B, Xi P, Zhang S 2018 J. Am. Chem. Soc. 140 17624
 Google Scholar
Google Scholar
[28] Xie Q X, Ren D, Bai L C, Ge R L, Zhou W H, Bai L, Xie W, Wang J H, Grätzel M, Luo J S 2023 Chin. J. Catalysis 44 127
 Google Scholar
Google Scholar
[29] Li S, Chen B B, Wang Y, Ye M Y, Aken P A V, Cheng C, Thomas A 2021 Nat. Mater. 20 1240
 Google Scholar
Google Scholar
[30] Wan K, Luo J S, Zhou C, Zhang T, Arbiol J, Lu X H, Mao B W, Zhang X, Fransaer J 2019 Adv. Funct. Mater. 29 1900315
 Google Scholar
Google Scholar
[31] Bai Y K, Wu Y, Zhou X C, Ye Y F, Nie K Q, Wang J, Xie M, Zhang Z X, Liu Z J, Cheng T, Gao C B 2022 Nat. Commun. 13 6094
 Google Scholar
Google Scholar
[32] Zhu J L, Qian J M, Peng X B, Xia B R, Gao D Q 2023 Nano-Micro Lett. 15 30
 Google Scholar
Google Scholar
[33] Chakraborty B, Kalra S, Beltrán-Suito R, Das C, Hellmann T, Menezes W P, Driess M 2020 Chem. Asian J. 15 852
 Google Scholar
Google Scholar
[34] Liang H J, Shuang W, Zhang Y T, Chao S J, Han H J, Wang X B, Zhang H, Yang L 2018 Chem. Electro. Chem. 5 494
 Google Scholar
Google Scholar
[35] Li Y M, Zhang X Y, Zhuo S Y, Liu S L, Han A X, Li L G, Tian Y 2021 Appl. Surf. Sci. 555 149441
 Google Scholar
Google Scholar
[36] He L B, Zhou D, Lin Y, Ge R X, Hou X D, Sun X P, Zheng C B 2018 ACS Catal. 8 3859
 Google Scholar
Google Scholar
[37] Chinnadurai D, Rajendiran R, Kandasamy P 2022 J. Colloid Inter. Sci. 606 101
 Google Scholar
Google Scholar
[38] Tan L, Yu J T, Wang C, Wang H F, Liu X, Gao H T, Xin L T, Liu D Z, Hou W G, Zhan T R 2022 Adv. Funct. Mater. 32 2200951
[39] Sun S, Zhou X, Cong B, Hong W, Chen G 2020 ACS Catal. 10 9086
 Google Scholar
Google Scholar
[40] Zhang J, Wang T, Pohl D, Rellinghaus B, Dong R H, Liu S H, Zhuang X D, Feng X L 2016 Angew. Chem. Int. Ed. 55 6702
 Google Scholar
Google Scholar
[41] Zhao Z L, Wu H X, He H L, Xu X L, Jin Y D 2014 Adv. Funct. Mater. 24 4698
 Google Scholar
Google Scholar
[42] Li Y, Chen G, Zhu Y, Hu Z, Chan T, She S, Dai J, Zhou W, Shao Z 2021 Adv. Funct. Mater. 31 2103569
 Google Scholar
Google Scholar
计量
- 文章访问数: 7611
- PDF下载量: 163
- 被引次数: 0
















 下载:
下载: