-
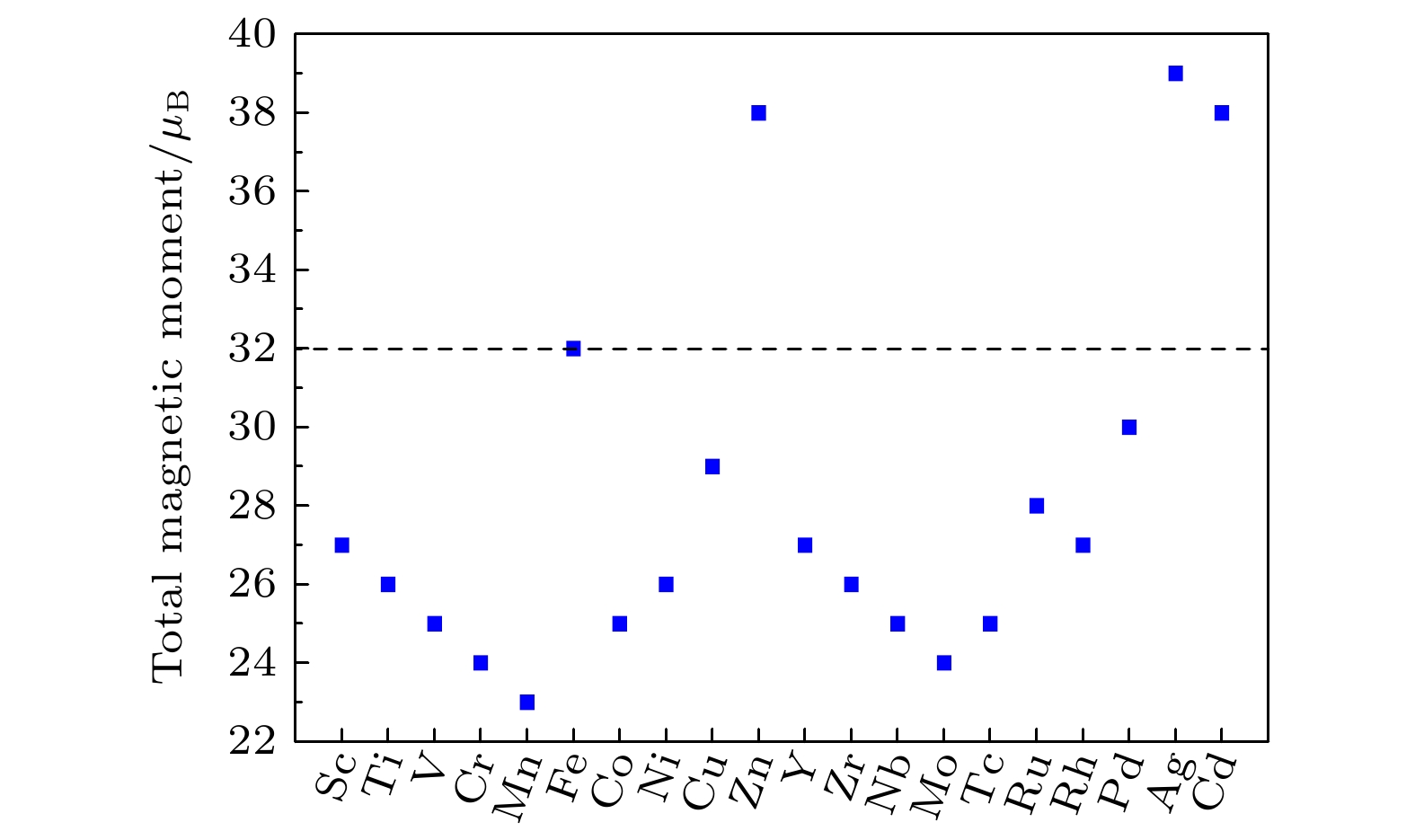
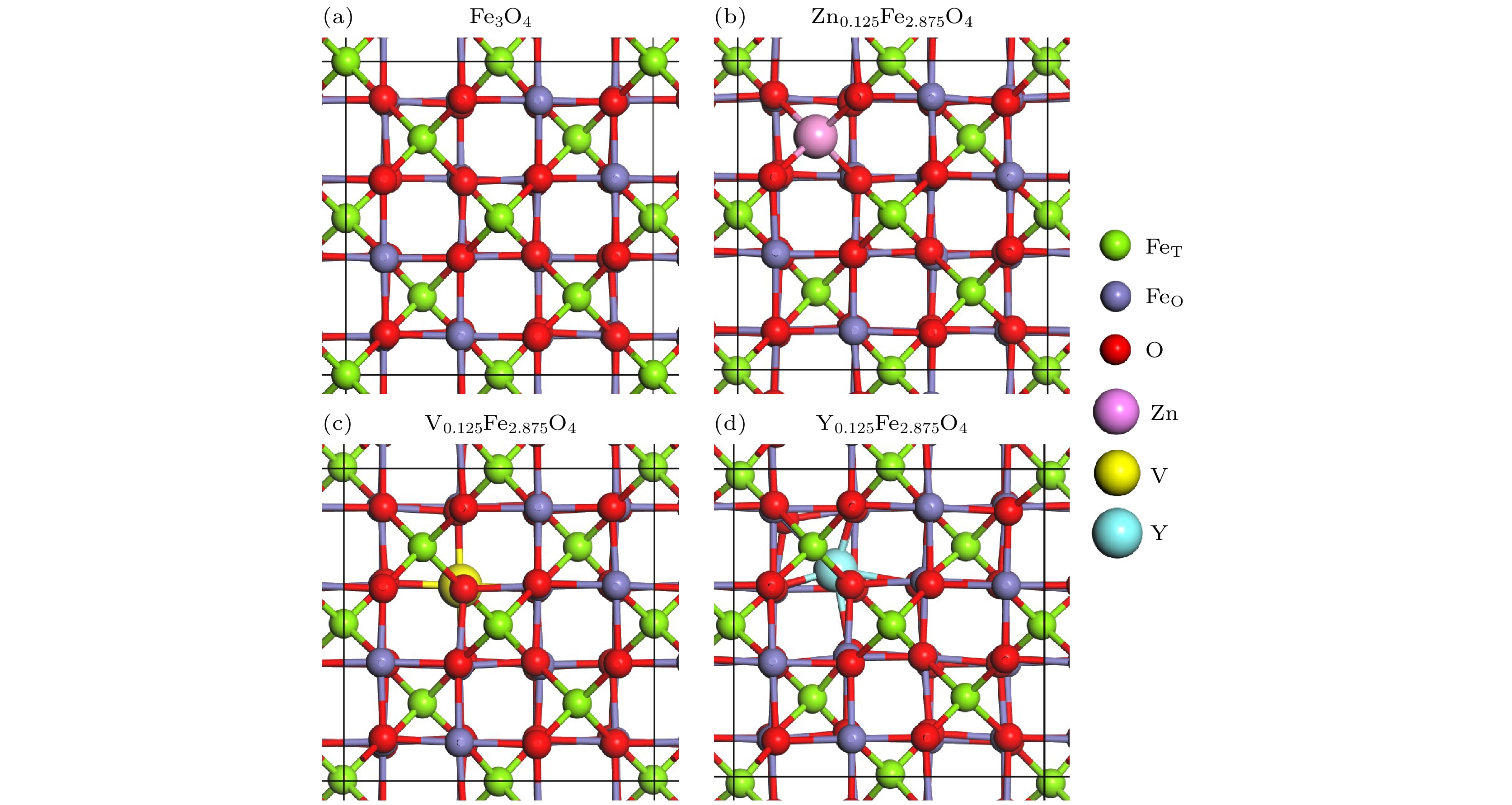
Magnetic Fe3O4 nanoparticles show promising applications in nanomedicine. The saturation magnetization (MS) and magnetic anisotropy are critical for the applications of Fe3O4 nanoparticles in drug delivery and magnetic hyperthermia. Here, by density functional computation, the doping effects of 3d and 4d transition metal elements (including Sc, Ti, V, Cr, Mn, Fe, Co, Ni, Cu, Zn, Y, Zr, Nb, Mo, Tc, Ru, Rh, Pd, Ag and Cd) on the magnetic properties of Fe3O4 are investigated in-depth. A conventional cell of Fe3O4, containing 24 Fe atoms and 32 O atoms, has been used to investigate the doping of group III elements. One 3d or 4d atom is doped in one conventional cell of Fe3O4, resulting in the formation of X0.125Fe2.875O4 where X represents the dopant. The results show that the doping of most 3d and 4d transition metal elements will reduce the total magnetic moment, while the doping of Ag, Zn and Cd in Fe3O4 will increase the total magnetic moment by 19%–22%. However, it is hard to dope Ag into Fe3O4 according to the positive formation energy. Therefore, Zn and Cd are good candidates to improve the MS of Fe3O4. The doping of Zn and Cd has also an influence on the magnetic anisotropy of Fe3O4. For Zn0.125Fe2.875O4, the magnetic anisotropy energy is about 0.25 meV per cell, which is slightly larger than that of intrinsic Fe3O4 (0.2 meV per cell). Interestingly, the doping of Cd (Cd0.125Fe2.875O4) will greatly increase the magnetic anisotropy energy to 0.8 meV per cell, which is significant for the specific absorption rate in the magnetic hyperthermia application. In addition, the doping of Zn and Cd will not induce any defect states in the band gap according to the density of states. Zn0.125Fe2.875O4 and Cd0.125Fe2.875O4 are both semiconducting and both the top of valence band and the bottom of conduction band originate from octahedral Fe. This is because the impurity states are very deep in energy. Our research results show that doping Cd is a feasible way to improve the performance of Fe3O4 as a material for drug delivery and magnetic hyperthermia.
-
Keywords:
- saturation magnetization /
- magnetic anisotropy /
- density functional theory /
- Fe3O4
[1] Perez J M, Josephson L, O'Loughlin T, Högemann D, Weissleder R 2002 Nat. Biotechnol. 20 816
 Google Scholar
Google Scholar
[2] Liu J, Sun Z, Deng Y, Zou Y, Li C, Guo X, Xiong L, Gao Y, Li F, Zhao D 2009 Angew. Chem. Int. Ed. 48 5875
 Google Scholar
Google Scholar
[3] Wu W, Wu Z, Yu T, Jiang C, Kim W S 2015 Sci. Technol. Adv. Mater. 16 023501
 Google Scholar
Google Scholar
[4] Martinkova P, Brtnicky M, Kynicky J, Pohanka M 2018 Adv. Healthc. Mater. 7 1700932
 Google Scholar
Google Scholar
[5] Pankhurst Q A, Thanh N T K, Jones S K, Dobson J 2009 J. Phys. D: Appl. Phys. 42 224001
 Google Scholar
Google Scholar
[6] Gupta A K, Gupta M 2005 Biomaterials 26 3995
 Google Scholar
Google Scholar
[7] Sun C, Lee J S H, Zhang M 2008 Adv. Drug. Deliv. Rev. 60 1252
 Google Scholar
Google Scholar
[8] Pankhurst Q A, Connolly J, Jones S K, Dobson J 2003 J. Phys. D: Appl. Phys. 36 R167
 Google Scholar
Google Scholar
[9] Laurent S, Forge D, Port M, Roch A, Robic C, Vander Elst L, Muller R N 2008 Chem. Rev. 108 2064
 Google Scholar
Google Scholar
[10] Colombo M, Carregal-Romero S, Casula M F, Gutiérrez L, Morales M P, Böhm I B, Heverhagen J T, Prosperi D, Parak W J 2012 Chem. Soc. Rev. 41 4306
 Google Scholar
Google Scholar
[11] Mitchell M J, Billingsley M M, Haley R M, Wechsler M E, Peppas N A, Langer R 2021 Nat. Rev. Drug Discov. 20 101
 Google Scholar
Google Scholar
[12] Dilnawaz F, Singh A, Mohanty C, Sahoo S K 2010 Biomaterials 31 3694
 Google Scholar
Google Scholar
[13] Wang Y, Zhao R B, Wang S B, Liu Z M, Tang R K 2016 Biomaterials 75 71
 Google Scholar
Google Scholar
[14] Liao S H, Liu C H, Bastakoti B P, Suzuki N, Chang Y, Yamauchi Y, Lin F H, Wu K C 2015 Int. J. Nanomed. 10 3315
[15] Rajan A, Sharma M, Sahu N K 2020 Sci. Rep. 10 15045
 Google Scholar
Google Scholar
[16] Sun S, Zeng H 2002 J. Am. Chem. Soc. 124 8204
 Google Scholar
Google Scholar
[17] Hou Y, Yu J, Gao S 2003 J. Mater. Chem. 13 1983
 Google Scholar
Google Scholar
[18] Sun S, Zeng H, Robinson D B, Raoux S, Rice P M, Wang S X, Li G 2004 J. Am. Chem. Soc. 126 273
 Google Scholar
Google Scholar
[19] Park J, An K, Hwang Y, Park J G, Noh H J, Kim J Y, Park J H, Hwang N M, Hyeon T 2004 Nat. Mater. 3 891
 Google Scholar
Google Scholar
[20] Tian Y, Yu B B, Li X, Li K 2011 J. Mater. Chem. 21 2476
 Google Scholar
Google Scholar
[21] Kovalenko M V, Bodnarchuk M I, Lechner R T, Hesser G, Schäffler F, Heiss W 2007 J. Am. Chem. Soc. 129 6352
 Google Scholar
Google Scholar
[22] Yang H, Ogawa T, Hasegawa D, Takahashi M 2008 J. Appl. Phys. 103 07D526
 Google Scholar
Google Scholar
[23] Kim D, Lee N, Park M, Kim B H, An K, Hyeon T 2009 J. Am. Chem. Soc. 131 454
 Google Scholar
Google Scholar
[24] Zhao L, Duan L 2010 Eur. J. Inorg. Chem. 2010 5635
 Google Scholar
Google Scholar
[25] Zhang L H, Wu J J, Liao H B, Hou Y L, Gao S 2009 Chem. Commun. 29 4378
[26] Li X, Liu D, Song S, Wang X, Ge X, Zhang H 2011 CrystEngComm 13 6017
[27] Cheng X L, Jiang J S, Jiang D M, Zhao Z J 2014 J. Phys. Chem. C 118 12588
 Google Scholar
Google Scholar
[28] Zheng R K, Gu H, Xu B, Fung K K, Zhang X X, Ringer S P 2006 Adv. Mater. 18 2418
 Google Scholar
Google Scholar
[29] Zhao L J, Zhang H J, Xing Y, Song S Y, Yu S Y, Shi W D, Guo X M, Yang J H, Lei Y Q, Cao F 2008 Chem. Mat. 20 198
 Google Scholar
Google Scholar
[30] Woo K, Hong J, Choi S, Lee H W, Ahn J P, Kim C S, Lee S W 2004 Chem. Mat. 16 2814
 Google Scholar
Google Scholar
[31] Li Q, Kartikowati C W, Horie S, Ogi T, Iwaki T, Okuyama K 2017 Sci. Rep. 7 9894
 Google Scholar
Google Scholar
[32] Liu J, Bin Y, Matsuo M 2012 J. Phys. Chem. C 116 134
 Google Scholar
Google Scholar
[33] Ahghari M R, Amiri-khamakani Z, Maleki A 2023 Sci. Rep. 13 1007
 Google Scholar
Google Scholar
[34] Qi Z L, Joshi T P, Liu R P, Liu H J, Qu J H 2017 J. Hazard. Mater. 329 193
 Google Scholar
Google Scholar
[35] Kresse G, Furthmüller J 1996 Phys. Rev. B 54 11169
 Google Scholar
Google Scholar
[36] Kresse G, Joubert D 1999 Phys. Rev. B 59 1758
[37] Perdew J P, Burke K, Ernzerhof M 1996 Phys. Rev. Lett. 77 3865
 Google Scholar
Google Scholar
[38] Liu H, Di Valentin C 2017 J. Phys. Chem. C 121 25736
 Google Scholar
Google Scholar
[39] Dovesi R, Orlando R, Erba A, Zicovich-Wilson C M, Civalleri B, Casassa S, Maschio L, Ferrabone M, De La Pierre M, D'Arco P, Noël Y, Causà M, Rérat M, Kirtman B 2014 Int. J. Quantum Chem. 114 1287
 Google Scholar
Google Scholar
[40] Krukau A V, Vydrov O A, Izmaylov A F, Scuseria G E 2006 J. Chem. Phys. 125 224106
 Google Scholar
Google Scholar
[41] Liu H, Di Valentin C 2018 Nanoscale 10 11021
[42] Liu H, Di Valentin C 2019 Phys. Rev. Lett. 123 186101
 Google Scholar
Google Scholar
[43] Hay P J, Wadt W R 1985 J. Chem. Phys. 82 299
 Google Scholar
Google Scholar
[44] Durand P, Barthelat J C 1975 Theor. Chim. Acta 38 283
 Google Scholar
Google Scholar
-
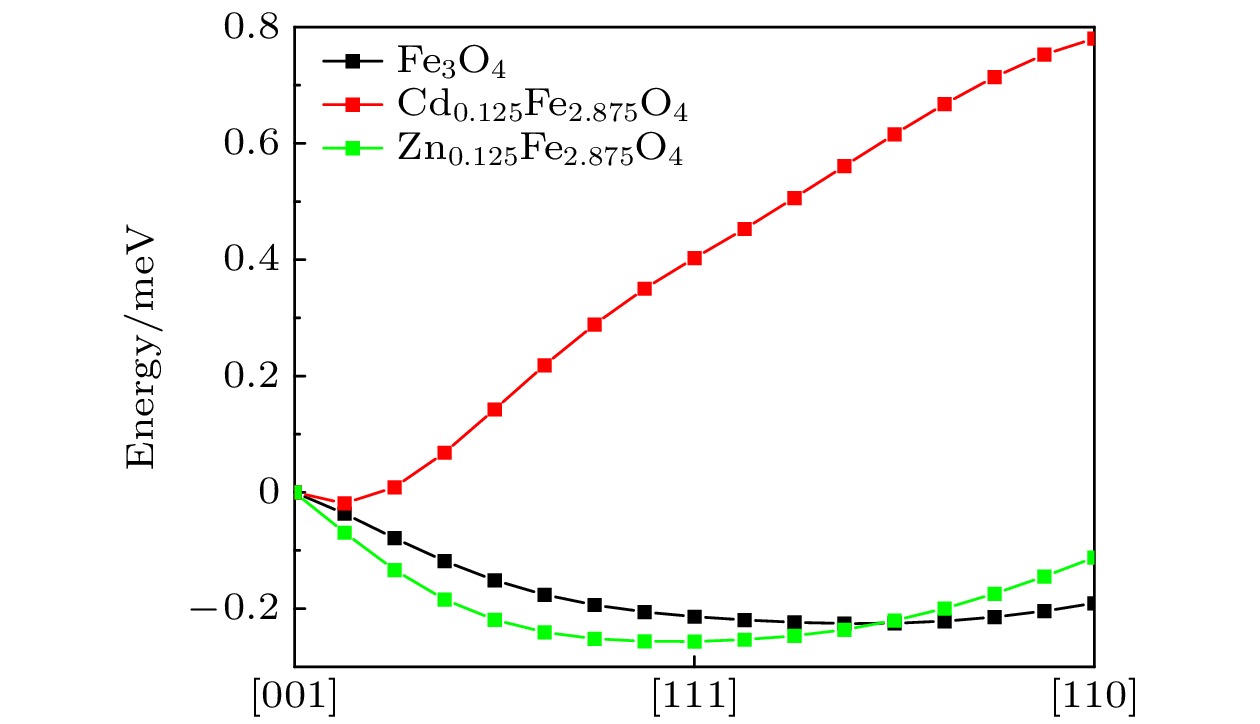
图 3 本征和掺杂的 Fe3O4 的磁各向异性, 图中横坐标为不同的磁化方向, 磁化轴从[001]方向开始沿着(1, –1, 0)面旋转到[111]方向, 中间均匀取7个数据点, 再从[111]方向沿着(1, –1, 0)面旋转到[110]方向, 中间也均匀取7个数据点(数据来源于VASP的PBE + U计算)
Fig. 3. Magnetic anisotropy of Fe3O4 with and without doping. The abscissas in the figure represent different magnetization directions. The magnetization axis starts from the [001] direction and rotates along the (1, –1, 0) plane to the [111] direction. Seven data points are evenly taken in the middle, and then the magnetization axis rotates from the [111] direction along the (1, –1, 0) plane to the [110] direction, and 7data points are evenly taken in the middle (data is from PBE + U calculation with VASP).
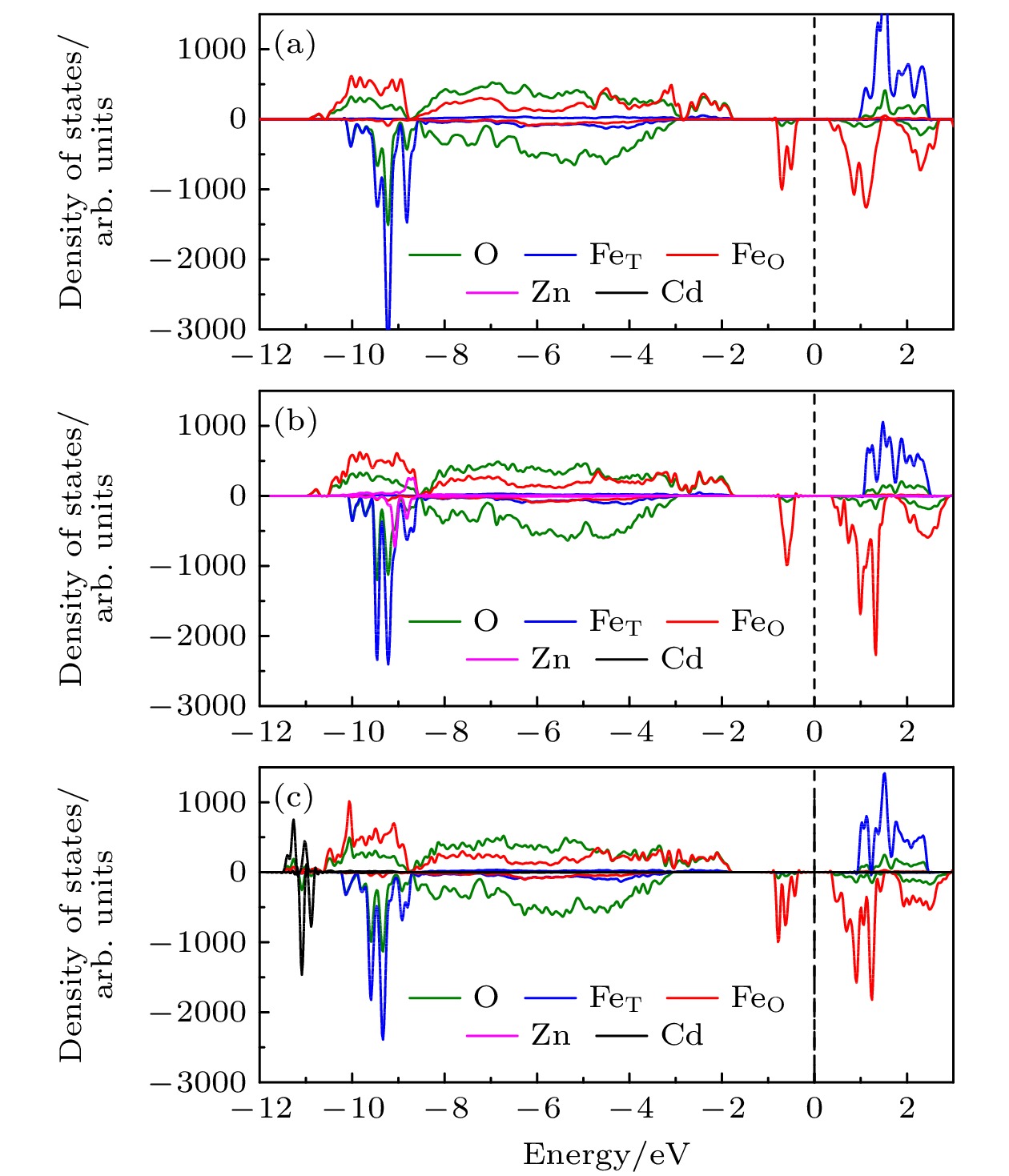
图 4 (a) 未掺杂的完美 Fe3O4, (b) Zn0.125Fe2.875O4, (c) Cd0.125Fe2.875O4 的投影态密度, 费米能级归零, 如黑色虚线所示(数据来源于CRYSTAL17的杂化泛函计算)
Fig. 4. Projected density of states of (a) perfect Fe3O4 without doping, (b) Zn0.125Fe2.875O4, (c) Cd0.125Fe2.875O4. The legend of colors is on the top, the Fermi level is scaled to zero as indicated by the dashed black lines (data is from HSE calculation with CRYSTAL17).
表 1 过渡金属掺杂Fe3O4的能量差ΔE = ET – EO, 其中ET和EO分别表示掺杂剂取代四面体 Fe 和八面体 Fe 的掺杂 Fe3O4 的总能量(数据来源于CRYSTAL17的杂化泛函计算)
Table 1. Energy difference ΔE = ET – EO for transition metal doped Fe3O4, where ET and EO represent the total energy of doped Fe3O4 with the dopant replacing tetrahedral Fe and octahedral Fe, respectively (data is from HSE calculation with CRYSTAL17).
杂质 Sc Ti V Cr Mn Co Ni Cu Zn ΔE /eV 0.70 0.10 0.37 1.48 0.10 0.13 0.95 0.19 –0.14 杂质 Y Zr Nb Mo Tc Ru Rh Pd Ag Cd ΔE /eV 4.23 0.13 0.01 0.72 2.14 2.27 1.31 0.96 –0.29 –0.32 表 2 过渡金属掺杂 Fe3O4 的形成能 Ef (数据来源于VASP的PBE + U计算)
Table 2. Formation energy Ef of transition metal doped Fe3O4 (data is from PBE + U calculation with VASP).
杂质 Sc Ti V Cr Mn Co Ni Cu Zn Ef /eV –6.45 –5.75 –4.94 –4.97 –3.72 –2.09 –1.69 0.21 –1.93 杂质 Y Zr Nb Mo Tc Ru Rh Pd Ag Cd Ef /eV –5.42 –6.09 –5.23 –2.60 –2.18 –1.00 –0.62 0.54 1.12 –0.61 表 3 Fe3O4掺杂时3d和4d过渡金属原子的原子磁矩(m)(数据来源于CRYSTAL17的杂化泛函计算)
Table 3. Atomic magnetic moment (m) of 3d and 4d transition metal atoms when doped in Fe3O4 (data is from HSE calculation with CRYSTAL17).
杂质 Sc Ti V Cr Mn Co Ni Cu Zn m/μB –0.06 –0.12 –2.02 –3.05 –4.76 –2.76 –1.78 0.01 0.07 杂质 Y Zr Nb Mo Tc Ru Rh Pd Ag Cd m/μB –0.09 –0.22 –0.52 –2.66 –1.91 0.11 –0.09 1.30 0.12 0.07 -
[1] Perez J M, Josephson L, O'Loughlin T, Högemann D, Weissleder R 2002 Nat. Biotechnol. 20 816
 Google Scholar
Google Scholar
[2] Liu J, Sun Z, Deng Y, Zou Y, Li C, Guo X, Xiong L, Gao Y, Li F, Zhao D 2009 Angew. Chem. Int. Ed. 48 5875
 Google Scholar
Google Scholar
[3] Wu W, Wu Z, Yu T, Jiang C, Kim W S 2015 Sci. Technol. Adv. Mater. 16 023501
 Google Scholar
Google Scholar
[4] Martinkova P, Brtnicky M, Kynicky J, Pohanka M 2018 Adv. Healthc. Mater. 7 1700932
 Google Scholar
Google Scholar
[5] Pankhurst Q A, Thanh N T K, Jones S K, Dobson J 2009 J. Phys. D: Appl. Phys. 42 224001
 Google Scholar
Google Scholar
[6] Gupta A K, Gupta M 2005 Biomaterials 26 3995
 Google Scholar
Google Scholar
[7] Sun C, Lee J S H, Zhang M 2008 Adv. Drug. Deliv. Rev. 60 1252
 Google Scholar
Google Scholar
[8] Pankhurst Q A, Connolly J, Jones S K, Dobson J 2003 J. Phys. D: Appl. Phys. 36 R167
 Google Scholar
Google Scholar
[9] Laurent S, Forge D, Port M, Roch A, Robic C, Vander Elst L, Muller R N 2008 Chem. Rev. 108 2064
 Google Scholar
Google Scholar
[10] Colombo M, Carregal-Romero S, Casula M F, Gutiérrez L, Morales M P, Böhm I B, Heverhagen J T, Prosperi D, Parak W J 2012 Chem. Soc. Rev. 41 4306
 Google Scholar
Google Scholar
[11] Mitchell M J, Billingsley M M, Haley R M, Wechsler M E, Peppas N A, Langer R 2021 Nat. Rev. Drug Discov. 20 101
 Google Scholar
Google Scholar
[12] Dilnawaz F, Singh A, Mohanty C, Sahoo S K 2010 Biomaterials 31 3694
 Google Scholar
Google Scholar
[13] Wang Y, Zhao R B, Wang S B, Liu Z M, Tang R K 2016 Biomaterials 75 71
 Google Scholar
Google Scholar
[14] Liao S H, Liu C H, Bastakoti B P, Suzuki N, Chang Y, Yamauchi Y, Lin F H, Wu K C 2015 Int. J. Nanomed. 10 3315
[15] Rajan A, Sharma M, Sahu N K 2020 Sci. Rep. 10 15045
 Google Scholar
Google Scholar
[16] Sun S, Zeng H 2002 J. Am. Chem. Soc. 124 8204
 Google Scholar
Google Scholar
[17] Hou Y, Yu J, Gao S 2003 J. Mater. Chem. 13 1983
 Google Scholar
Google Scholar
[18] Sun S, Zeng H, Robinson D B, Raoux S, Rice P M, Wang S X, Li G 2004 J. Am. Chem. Soc. 126 273
 Google Scholar
Google Scholar
[19] Park J, An K, Hwang Y, Park J G, Noh H J, Kim J Y, Park J H, Hwang N M, Hyeon T 2004 Nat. Mater. 3 891
 Google Scholar
Google Scholar
[20] Tian Y, Yu B B, Li X, Li K 2011 J. Mater. Chem. 21 2476
 Google Scholar
Google Scholar
[21] Kovalenko M V, Bodnarchuk M I, Lechner R T, Hesser G, Schäffler F, Heiss W 2007 J. Am. Chem. Soc. 129 6352
 Google Scholar
Google Scholar
[22] Yang H, Ogawa T, Hasegawa D, Takahashi M 2008 J. Appl. Phys. 103 07D526
 Google Scholar
Google Scholar
[23] Kim D, Lee N, Park M, Kim B H, An K, Hyeon T 2009 J. Am. Chem. Soc. 131 454
 Google Scholar
Google Scholar
[24] Zhao L, Duan L 2010 Eur. J. Inorg. Chem. 2010 5635
 Google Scholar
Google Scholar
[25] Zhang L H, Wu J J, Liao H B, Hou Y L, Gao S 2009 Chem. Commun. 29 4378
[26] Li X, Liu D, Song S, Wang X, Ge X, Zhang H 2011 CrystEngComm 13 6017
[27] Cheng X L, Jiang J S, Jiang D M, Zhao Z J 2014 J. Phys. Chem. C 118 12588
 Google Scholar
Google Scholar
[28] Zheng R K, Gu H, Xu B, Fung K K, Zhang X X, Ringer S P 2006 Adv. Mater. 18 2418
 Google Scholar
Google Scholar
[29] Zhao L J, Zhang H J, Xing Y, Song S Y, Yu S Y, Shi W D, Guo X M, Yang J H, Lei Y Q, Cao F 2008 Chem. Mat. 20 198
 Google Scholar
Google Scholar
[30] Woo K, Hong J, Choi S, Lee H W, Ahn J P, Kim C S, Lee S W 2004 Chem. Mat. 16 2814
 Google Scholar
Google Scholar
[31] Li Q, Kartikowati C W, Horie S, Ogi T, Iwaki T, Okuyama K 2017 Sci. Rep. 7 9894
 Google Scholar
Google Scholar
[32] Liu J, Bin Y, Matsuo M 2012 J. Phys. Chem. C 116 134
 Google Scholar
Google Scholar
[33] Ahghari M R, Amiri-khamakani Z, Maleki A 2023 Sci. Rep. 13 1007
 Google Scholar
Google Scholar
[34] Qi Z L, Joshi T P, Liu R P, Liu H J, Qu J H 2017 J. Hazard. Mater. 329 193
 Google Scholar
Google Scholar
[35] Kresse G, Furthmüller J 1996 Phys. Rev. B 54 11169
 Google Scholar
Google Scholar
[36] Kresse G, Joubert D 1999 Phys. Rev. B 59 1758
[37] Perdew J P, Burke K, Ernzerhof M 1996 Phys. Rev. Lett. 77 3865
 Google Scholar
Google Scholar
[38] Liu H, Di Valentin C 2017 J. Phys. Chem. C 121 25736
 Google Scholar
Google Scholar
[39] Dovesi R, Orlando R, Erba A, Zicovich-Wilson C M, Civalleri B, Casassa S, Maschio L, Ferrabone M, De La Pierre M, D'Arco P, Noël Y, Causà M, Rérat M, Kirtman B 2014 Int. J. Quantum Chem. 114 1287
 Google Scholar
Google Scholar
[40] Krukau A V, Vydrov O A, Izmaylov A F, Scuseria G E 2006 J. Chem. Phys. 125 224106
 Google Scholar
Google Scholar
[41] Liu H, Di Valentin C 2018 Nanoscale 10 11021
[42] Liu H, Di Valentin C 2019 Phys. Rev. Lett. 123 186101
 Google Scholar
Google Scholar
[43] Hay P J, Wadt W R 1985 J. Chem. Phys. 82 299
 Google Scholar
Google Scholar
[44] Durand P, Barthelat J C 1975 Theor. Chim. Acta 38 283
 Google Scholar
Google Scholar
计量
- 文章访问数: 5028
- PDF下载量: 169
- 被引次数: 0













 下载:
下载: