-
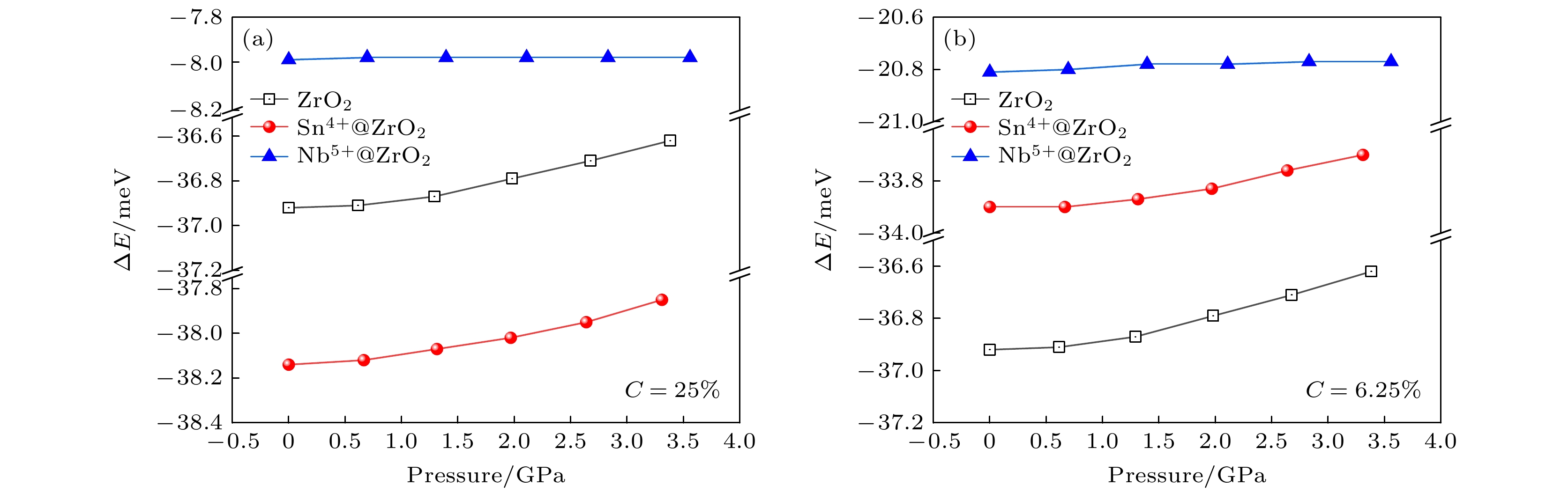
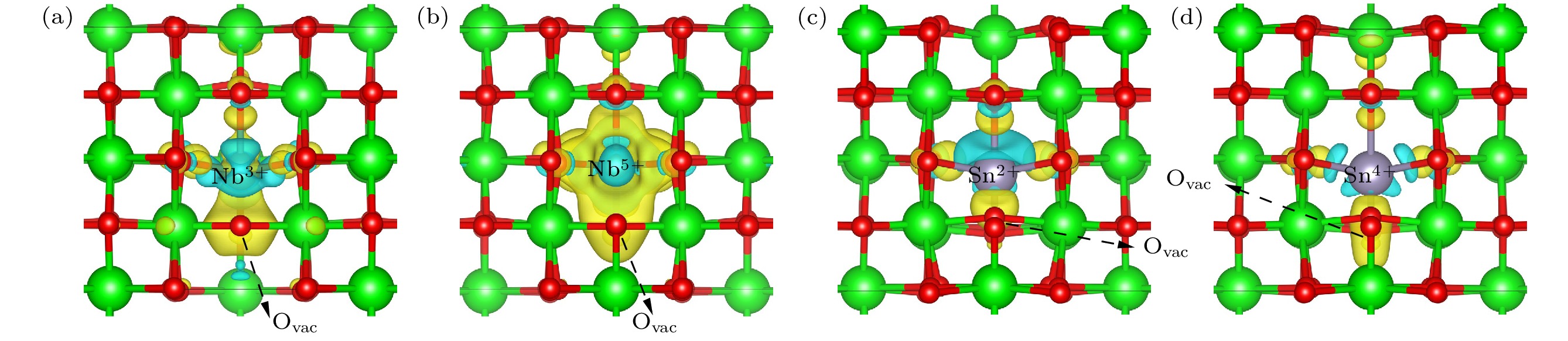
锆合金的水侧腐蚀是核燃料棒包壳材料设计的关键问题之一. 包壳材料的耐腐蚀性能与锆合金氧化膜中t-ZrO2含量和t-m相变密切相关. 目前, Zr-Sn-Nb系合金是新型锆合金发展的主流方向. 合金元素Sn, Nb在氧化膜中可呈现多种价态, 显著影响ZrO2稳定性, 然而Sn, Nb对t-ZrO2含量和t-m相变的影响机制尚不明晰. 本文基于第一性原理计算了不同价态Sn, Nb掺杂ZrO2的晶体结构性质、形成焓和氧空位形成能, 从原子尺度揭示了Sn, Nb对ZrO2稳定性的影响机理. 研究表明Sn2+, Nb3+引起显著晶格膨胀; Sn4+则造成轻微晶格膨胀, 而Nb5+引起晶格收缩, 可见高氧化态下Nb比Sn更利于减小氧化膜的内应力. 低价合金元素降低ZrO2稳定性, 且会增大t, m相形成能差距; 高价的Nb5+, Sn4+均可提高t-ZrO2相对稳定性从而抑制t-m相变, 其中Nb5+效果显著, Sn4+则作用微弱. 0—3.5 GPa范围内, t-ZrO2相对稳定性随压力增大而增强. 合金元素的低价态比高价态更利于在t-ZrO2中形成氧空位, 因而在氧化膜/金属界面附近低氧化态区域, 低价元素和压应力是稳定t-ZrO2的主要因素. 通过电子结构分析, 发现氧空位形成能与合金元素离子和氧空位间的电荷转移幅度(或电子局域化程度)呈正相关. 这些结果有助于针对锆合金耐腐蚀性的成分优化和结构设计 .
-
关键词:
- 第一性原理 /
- Zr-Sn-Nb合金 /
- 氧化膜 /
- 相稳定性
Water-side oxidative corrosion of zirconium alloy is a key problem in the design of nuclear fuel rods cladding materials in pressurised water reactors (PWRs), and its corrosion resistance is one of the main factors limiting service life. At present, Zr-Sn-Nb system alloys are still the main development direction of advanced zirconium alloys. Sn and Nb can exhibit a variety of valence states in the oxide film of the cladding and significantly affect the stability of ZrO2. However, the influence mechanism of Sn and Nb on the fraction of t-ZrO2 and the t-m phase transition is unclear. In this work, the lattice properties, formation enthalpies, and oxygen vacancy formation energy of ZrO2 under the doping conditions of Sn and Nb with different valence states are calculated based on the first-principles, and the influence mechanism of Sn and Nb on the stability of ZrO2 is revealed at an atomic scale. The results show that there is a significant difference between the effects of Sn and Nb, as well as between low-valent and high-valent elements. Sn2+ and Nb3+ cause lattice swelling to be significantly distorted , Nb5+ causes lattice to shrink, which contributes to reducing the stresses within the film, and Sn4+ leads the lattice to slightly swell. The low-valent elements all make ZrO2 less stable and are unfavourable for the stability of t-ZrO2 relative to m-ZrO2. The high-valent Nb5+and Sn4+ promote the relative stability of t-ZrO2, thus inhibiting the t-m phase transition, with Nb5+ having a significant effect and Sn4+ having a weak effect. The relative stability of t-ZrO2 increases with pressure rising in a range of 0–3.5 GPa. Compared with high-valent elements, the low-valent elements are favourable for introduing oxygen vacancies into t-ZrO2, thus stabilising the interfacial t-ZrO2 and enhancing the corrosion resistance of the cladding. By investigating the electronic structure, it is found that the oxygen vacancy formation energy is positively correlated with the magnitude of charge transfer (or degree of electron localisation) between the alloying element ion and the oxygen vacancy. These results contribute to optimizing the composition and designing the structure for corrosion resistance of zirconium alloys.-
Keywords:
- first-principles /
- Zr-Sn-Nb alloys /
- oxide films /
- phase stability
[1] Yuan R, Xie Y P, Li T, Xu C H, Yao M Y, Xu J X, Guo H B, Zhou B X 2021 Acta Mater. 209 116804
 Google Scholar
Google Scholar
[2] Kim T, Couet A, Kim S, Lee Y, Yoo S C, Kim J H 2020 Corros. Sci. 173 108745
 Google Scholar
Google Scholar
[3] Liu S M, Beyerlein I J, Han W Z 2020 Nat. Commun. 11 5766
 Google Scholar
Google Scholar
[4] Wei K J, Chen L, Qu Y, Zhang Y F, Jin X Y, Xue W B, Zhang J L 2018 Corros. Sci. 143 129
 Google Scholar
Google Scholar
[5] Zhou B F, Feng K Q 2018 RSC Adv. 8 26251
 Google Scholar
Google Scholar
[6] Bell B D C, Murphy S T, Burr P A, Comstock R J, Partezana J M, Grimes R W, Wenman M R 2016 Corros. Sci. 105 36
 Google Scholar
Google Scholar
[7] Nikulina A V 2004 Met. Sci. Heat Treat. 46 458
 Google Scholar
Google Scholar
[8] Ruiz-Hervias J, Simbruner K, Cristobal-Beneyto M, Perez-Gallego D, Zencker U 2021 J. Nucl. Mater. 544 152668
 Google Scholar
Google Scholar
[9] Zhang F L, Chai L J, Qi L, Wang Y Y, Wu L, Pan H C, Teng C Q, Murty K L 2023 J. Nucl. Mater. 577 154284
 Google Scholar
Google Scholar
[10] Qin W, Nam C, Li H L, Szpunar J A 2007 Acta Mater. 55 1695
 Google Scholar
Google Scholar
[11] Liao J J, Yang Z B, Qiu S Y, Peng Q, Li Z C, Zhang J S 2019 J. Nucl. Mater. 524 101
 Google Scholar
Google Scholar
[12] Motta A T, Couet A, Comstock R J 2015 Annu. Rev. Mater. Res. 45 311
 Google Scholar
Google Scholar
[13] Hillner E, Franklin D G, Smee J D 2000 J. Nucl. Mater. 278 334
 Google Scholar
Google Scholar
[14] Couet A, Motta A T, Ambard A 2015 Corros. Sci. 100 73
 Google Scholar
Google Scholar
[15] Yilmazbayhan A, Motta A T, Comstock R J, Sabol G P, Lai B, Cai Z 2004 J. Nucl. Mater. 324 6
 Google Scholar
Google Scholar
[16] Massih A R, Vesterlund G 1992 Nucl. Eng. Des. 137 57
 Google Scholar
Google Scholar
[17] Motta A T 2011 JOM 63 59
 Google Scholar
Google Scholar
[18] Bouineau V, Bénier G, Pêcheur D, Thomazet J, Ambard A, Blat M 2010 Nucl. Technol. 170 444
 Google Scholar
Google Scholar
[19] Takagi I, Une K, Miyamura S, Kobayashi T 2011 J. Nucl. Mater. 419 339
 Google Scholar
Google Scholar
[20] Couet A, Borrel L, Liu J L, Hu J, Grovenor C 2019 Corros. Sci. 159 108134
 Google Scholar
Google Scholar
[21] Hu J, Garner A, Frankel P, Li M, Kirk M A, Lozano-Perez S, Preuss M, Grovenor C 2019 Acta Mater. 173 313
 Google Scholar
Google Scholar
[22] Liu J L, Yu H B, Karamched P, Hu J, He G Z, Goran D, Hughes G M, Wilkinson A J, Lozano-Perez S, Grovenor C R M 2019 Acta Mater. 179 328
 Google Scholar
Google Scholar
[23] Kurpaska L, Favergeon J, Lahoche L, Moulin G, Marssi M El, Roelandt J M 2013 Oxid. Met. 79 261
 Google Scholar
Google Scholar
[24] Xun G, Qingdong L, Wenqing L, Meiyi Y, Bangxin Z 2008 Rare Met. Mater. Eng. 37 1415
 Google Scholar
Google Scholar
[25] Beie H, Mitwalsky A, Garzarolli F, Ruhmann H, Sell H 1994 Zirconium in the Nuclear Industry: Tenth International Symposium (West Conshohocken, PA: ASTM International) pp615–643
[26] Barberis P 1995 J. Nucl. Mater. 226 34
 Google Scholar
Google Scholar
[27] Polatidis E, Frankel P, Wei J, Klaus M, Comstock R J, Ambard A, Lyon S, Cottis R A, Preuss M 2013 J. Nucl. Mater. 432 102
 Google Scholar
Google Scholar
[28] Bouvier P, Godlewski J, Lucazeau G 2002 J. Nucl. Mater. 300 118
 Google Scholar
Google Scholar
[29] Preuss M, Frankel P, Polatidis E, Wei J, Smith J W, Wang C, Cottis R A, Lyon S B, Lozano-Perez S, Hudson D, Ni N, Grovenor C R M, Smith G D W, Sykes J, Cerezo A, Storer S, Fitzpatrick M E 2011 16th International Symposium on Zirconium in the Nuclear Industry Chengdu, China, May 9–13, 2010
[30] Wei J, Frankel P, Polatidis E, Blat M, Ambard A, Comstock R J, Hallstadius L, Hudson D, Smith G D W, Grovenor C R M, Klaus M, Cottis R A, Lyon S, Preuss M 2013 Acta Mater. 61 4200
 Google Scholar
Google Scholar
[31] Garner A, Hu J, Harte A, Frankel P, Grovenor C, Lozano-Perez S, Preuss M 2015 Acta Mater. 99 259
 Google Scholar
Google Scholar
[32] Jeong Y H, Lee K O, Kim H G 2002 J. Nucl. Mater. 302 9
 Google Scholar
Google Scholar
[33] Jeong Y H, Kim H G, Kim T H 2003 J. Nucl. Mater. 317 1
 Google Scholar
Google Scholar
[34] Sakamoto K, Une K, Aomi M, Otsuka T, Hashizume K 2015 J. Nucl. Sci. Technol. 52 1259
 Google Scholar
Google Scholar
[35] Liao J J, Xu F, Peng Q, Yang Z B, Li Z C, Qiu S Y 2020 J. Nucl. Mater. 528 151846
 Google Scholar
Google Scholar
[36] Froideval A, Degueldre C, Segre C U, Pouchon M A, Grolimund D 2008 Corros. Sci. 50 1313
 Google Scholar
Google Scholar
[37] Hulme H, Baxter F, Babu R P, Denecke M A, Gass M, Steuwer A, Norén K, Carlson S, Preuss M 2016 Corros. Sci. 105 202
 Google Scholar
Google Scholar
[38] Wu J W, Xie Y P, Yao M Y, Guan S H, Zhao Y, Pan R J, Wu L, Liu Z P 2023 Phys. Chem. Chem. Phys. 25 8934
 Google Scholar
Google Scholar
[39] Zhao X S, Shang S L, Liu Z K, Shen J Y 2011 J. Nucl. Mater. 415 13
 Google Scholar
Google Scholar
[40] 赵毅, 王栋, 徐晨皓, 吴江桅, 王洋, 谢耀平 2021 上海金属 43 112
 Google Scholar
Google Scholar
Zhao Y, Wang D, Xu C H, Wu J W, Wang Y, Xie Y P 2021 Shanghai Met. 43 112
 Google Scholar
Google Scholar
[41] Kresse G, Hafner J 1993 Phys. Rev. B 47 558
 Google Scholar
Google Scholar
[42] Kresse G, Furthmüller J 1996 Comput. Mater. Sci. 6 15
 Google Scholar
Google Scholar
[43] Kresse G, Furthmüller J 1996 Phys. Rev. B 54 11169
 Google Scholar
Google Scholar
[44] Blöchl P E 1994 Phys. Rev. B 50 17953
 Google Scholar
Google Scholar
[45] Perdew J P, Burke K, Ernzerhof M 1997 Phys. Rev. Lett. 78 1396
 Google Scholar
Google Scholar
[46] Monkhorst H J, Pack J D 1976 Phys. Rev. B 13 5188
 Google Scholar
Google Scholar
[47] Methfessel M, Paxton A T 1989 Phys. Rev. B 40 3616
 Google Scholar
Google Scholar
[48] Barbour O, Crocombette J P, Schuler T, Tupin M 2020 J. Nucl. Mater. 539 152333
 Google Scholar
Google Scholar
[49] Zhang P, Lu Y, He C H, Zhang P 2011 J. Nucl. Mater. 418 143
 Google Scholar
Google Scholar
[50] Fukui H, Fujimoto M, Akahama Y, Sano-Furukawa A, Hattori T 2019 Acta Crystallogr. Sect. B Struct. Sci. Cryst. Eng. Mater. 75 742
 Google Scholar
Google Scholar
[51] Delaforce P M, Yeomans J A, Filkin N C, Wright G J, Thomson R C 2007 J. Am. Ceram. Soc. 90 918
 Google Scholar
Google Scholar
[52] Torres F J, Amigó J M, Alarcón J 2003 J. Solid State Chem. 173 40
 Google Scholar
Google Scholar
[53] Lutterotti L, Scardi P 1990 J. Appl. Crystallogr. 23 246
 Google Scholar
Google Scholar
[54] Bouvier P, Djurado E, Lucazeau G, Le Bihan T 2000 Phys. Rev. B 62 8731
 Google Scholar
Google Scholar
[55] Bondars B, Heidemane G, Grabis J, Laschke K, Boysen H, Schneider J, Frey F 1995 J. Mater. Sci. 30 1621
 Google Scholar
Google Scholar
[56] Puchala B, der Ven A 2013 Phys. Rev. B 88 94108
 Google Scholar
Google Scholar
[57] Fisher E S, Renken C J 1964 Phys. Rev. 135 A482
 Google Scholar
Google Scholar
[58] Anada H, Takeda K 1996 Zirconium in the Nuclear Industry: Eleventh International Symposium (West Conshohocken, PA: ASTM International) pp35–54
[59] Wang C, Zinkevich M, Aldinger F 2004 Calphad 28 281
 Google Scholar
Google Scholar
[60] Chevalier P Y, Fischer E, Cheynet B 2004 Calphad 28 15
 Google Scholar
Google Scholar
[61] Schubert H, Frey F 2005 J. Eur. Ceram. Soc. 25 1597
 Google Scholar
Google Scholar
[62] Becke A D, Edgecombe K E 1990 J. Chem. Phys. 92 5397
 Google Scholar
Google Scholar
-
图 3 三维差分电荷密度(e/Bohr3)([001]方向观察得到, 蓝色等值面值为–0.008 e/Bohr3, 黄色等值面值为0.008 e/Bohr3) (a) t-ZrO2中Nb3+与1NN氧空位; (b) t-ZrO2中Nb5+与1NN氧空位; (c) t-ZrO2中Sn2+与1NN氧空位; (d) t-ZrO2中Sn4+与1NN氧空位
Fig. 3. Three-dimensional differential charge density (e/Bohr3), observed in the [001] direction: (a) Nb3+ with 1NN-Ovac in t-ZrO2; (b) Nb5+ with 1NN-Ovac in t-ZrO2; (c) Sn2+ with 1NN-Ovac in t-ZrO2; (d) Sn4+ with 1NN-Ovac in t-ZrO2. The value of the blue isosurface is –0.008 e/Bohr3, yellow isosurface is 0.008 e/Bohr3.
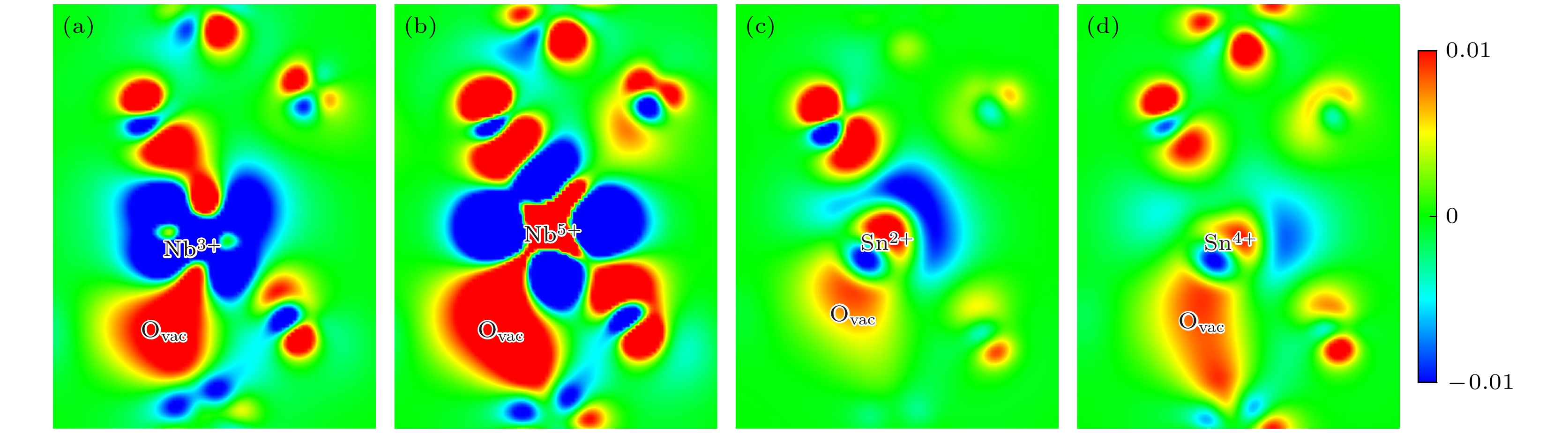
图 4 二维差分电荷密度(e/Bohr3), 取自$ {M}^{x+} $-Ovac对所在的(100)面 (a) Nb3+-Ovac; (b) Nb5+-Ovac; (c) Sn2+-Ovac; (d) Sn4+-Ovac
Fig. 4. Two-dimensional differential charge density (e/Bohr3), taken from the (100) plane in which $ {M}^{x+} $-Ovac is located: (a) Nb3+-Ovac; (b) Nb5+-Ovac; (c) Sn2+-Ovac; (d) Sn4+-Ovac.
表 1 锆(亚)氧化物的晶格常数(a, b, c)、晶胞矢量夹角(α, β, γ)及形成焓(Hf)
Table 1. Lattice constants (a, b, c), unit cell vector angles (α, β, γ ) and formation enthalpy (Hf) of Zr (sub)oxides.
Phase a/Å b/Å c/Å α/(°) β/(°) γ/(°) $ {H}_{{\mathrm{f}}} $/eV Ref. m-ZrO2 5.1901 5.2441 5.3760 90.00 99.63 90.00 –3.860 This work 5.1850 5.2780 5.2860 90.00 99.65 90.00 –3.778 DFT-GGA[40] 5.0791 5.1785 5.2340 90.00 99.49 90.00 — DFT-LDA[50] 5.0930 5.1760 5.2440 90.00 99.10 90.00 — DFT-LDA[51] 5.0790 5.2080 5.3110 90.00 99.23 90.00 — Exp.[52] 5.1473 5.2088 5.3166 90.00 99.21 90.00 — Exp.[50] t-ZrO2 3.6220 3.6220 5.2733 90.00 90.00 90.00 –3.823 This work 3.6370 3.6370 5.2820 90.00 90.00 90.00 –3.743 DFT-GGA[40] 3.5780 3.5780 5.1630 90.00 90.00 90.00 — DFT-LDA[39] 3.5948 3.5948 5.1824 90.00 90.00 90.00 — Exp.[53] 3.5916 3.5916 5.1790 90.00 90.00 90.00 — Exp.[54] 3.5961 3.5961 5.1770 90.00 90.00 90.00 — Exp.[55] h-ZrO 5.3110 5.3110 3.2005 90.00 90.00 120.00 –2.975 This work 5.2850 5.2850 3.1790 90.00 90.00 120.00 — DFT-GGA[56] 5.3100 5.3100 3.2000 90.00 90.00 120.00 — Exp.[22] 表 2 12原子超胞的晶格常数(a, b, c)、基矢夹角(α, β, γ), ZrO2单元平均体积(V ), $ {M}^{x+} $掺杂超胞体积的变化($\Delta V $)
Table 2. Lattice constants (a, b, c), basis vector angles (α, β, γ), average volume of ZrO2 unit (V ) of the 12-atom supercell, and the volume change ($ \Delta V$) of supercell with $ {M}^{x+} $ doping.
Phase a/Å b/Å c/Å α/(°) β/(°) γ/(°) V/Å3 ΔV/Å3 ΔV/V0/% pure phases t 3.62 3.62 5.28 90.00 90.00 90.00 34.62 — — m 5.19 5.24 5.38 90.00 99.63 90.00 36.07 — — Nb5+ doping t 3.52 3.51 5.07 90.00 90.00 90.00 31.27 –3.34 –10.69 m 4.94 5.18 5.00 90.10 96.75 89.69 31.76 –4.31 –13.56 Nb4+ doping t 3.61 3.61 5.24 90.00 90.00 90.00 34.07 –0.55 –1.61 m 5.14 5.20 5.36 89.90 100.48 89.23 35.28 –0.79 –2.23 Nb3+ doping t 3.33 4.23 7.02 90.00 90.00 90.00 49.49 14.88 30.06 m 5.32 5.30 5.52 90.14 102.46 89.71 38.02 1.95 5.14 Sn4+ doping t 3.65 3.59 5.35 90.00 90.00 90.00 35.08 0.46 1.31 m 5.20 5.16 5.47 89.62 97.14 88.22 36.41 0.34 0.93 Sn2+ doping t 4.14 3.41 7.00 90.00 90.00 90.00 49.34 14.73 29.84 m 5.80 5.90 6.18 90.26 115.55 89.93 47.77 11.70 24.48 表 3 $ {E}_{{\mathrm{f}}}({\mathrm{O}}x, P) $, $ {{\Delta }}E({\mathrm{O}}x, P) $的计算值, 其中浓度$ C= $ $ {n}_{M}/({n}_{{\mathrm{Z}}{\mathrm{r}}}+{n}_{M}) $
Table 3. Values of $ {E}_{{\mathrm{f}}}({\mathrm{O}}x, P) $ and $ {{\Delta }}E({\mathrm{O}}x, P) $. $ C= $$ {n}_{M}/({n}_{{\mathrm{Z}}{\mathrm{r}}}+{n}_{M}) $.
M x+ Phases C/% 25 6.25 3.125 E(Ox, 0)
/(eV·atom–1)Pure t –3.82 m –3.86 Sn4+ t –3.47 –3.74 –3.78 m –3.51 –3.77 –3.82 Sn2+ t –2.72 –3.54 –3.68 m –2.83 –3.59 –3.72 Nb5+ t –4.38 –3.96 –3.89 m –4.39 –3.98 –3.92 Nb4+ t –3.44 –3.75 –3.79 m –3.49 –3.79 –3.82 Nb3+ t –2.72 –3.54 –3.68 m –2.68 –3.60 –3.73 $ \Delta $E($ {\mathrm{O}}x $, 0)
/(meV·atom–1)Pure –36.92 Sn4+ –38.14 –33.90 –35.47 Sn2+ –115.96 –49.31 –42.26 Nb5+ –7.99 –20.81 –29.65 Nb4+ –51.89 –38.89 –38.11 Nb3+ 38.71 –60.04 –48.30 表 4 t-ZrO2中氧空位形成能. $ {\mathrm{O}}x $表示纯ZrO2或3.125%浓度$ {M}^{x+} $掺杂的ZrO2; $ {M}^{x+} $包括Nb3+, Nb4+, Nb5+, Sn2+, Sn4+; $ {E}_{{\mathrm{f}}}^{{1{\mathrm{N}}{\mathrm{N}}{\text{-}}{\mathrm{O}}}_{{\mathrm{v}}{\mathrm{a}}{\mathrm{c}}}} $, $ {E}_{{\mathrm{f}}}^{{2{\mathrm{N}}{\mathrm{N}}{\text{-}}{\mathrm{O}}}_{{\mathrm{v}}{\mathrm{a}}{\mathrm{c}}}} $分别表示$ {M}^{x+} $的1NN, 2NN氧空位形成能
Table 4. Oxygen vacancy formation energy in t-ZrO2. $ {\mathrm{O}}x $ represent pure ZrO2 or ZrO2 with 3.125% $ {M}^{x+} $ doping, and $ {M}^{x+}= $ Nb3+, Nb4+, Nb5+, Sn2+, Sn4+. $ {E}_{{\mathrm{f}}}^{{1{\mathrm{N}}{\mathrm{N}}{\text{-}}{\mathrm{O}}}_{{\mathrm{v}}{\mathrm{a}}{\mathrm{c}}}} $ and $ {E}_{{\mathrm{f}}}^{{2{\mathrm{N}}{\mathrm{N}}{\text{-}}{\mathrm{O}}}_{{\mathrm{v}}{\mathrm{a}}{\mathrm{c}}}} $ represent the 1NN and 2NN oxygen vacancy formation energies of $ {M}^{x+} $, respectively.
$ {\mathrm{O}}x $ Pure Nb5+ Nb4+ Nb3+ Sn4+ Sn2+ $ {E}_{{\mathrm{f}}}^{{1{\mathrm{N}}{\mathrm{N}}{\text{-}}{\mathrm{O}}}_{{\mathrm{v}}{\mathrm{a}}{\mathrm{c}}}} $/eV 6.353 5.771 5.691 5.252 4.474 3.905 $ {E}_{{\mathrm{f}}}^{{2{\mathrm{N}}{\mathrm{N}}{\text{-}}{\mathrm{O}}}_{{\mathrm{v}}{\mathrm{a}}{\mathrm{c}}}} $/eV 5.938 5.919 5.362 5.257 5.029 -
[1] Yuan R, Xie Y P, Li T, Xu C H, Yao M Y, Xu J X, Guo H B, Zhou B X 2021 Acta Mater. 209 116804
 Google Scholar
Google Scholar
[2] Kim T, Couet A, Kim S, Lee Y, Yoo S C, Kim J H 2020 Corros. Sci. 173 108745
 Google Scholar
Google Scholar
[3] Liu S M, Beyerlein I J, Han W Z 2020 Nat. Commun. 11 5766
 Google Scholar
Google Scholar
[4] Wei K J, Chen L, Qu Y, Zhang Y F, Jin X Y, Xue W B, Zhang J L 2018 Corros. Sci. 143 129
 Google Scholar
Google Scholar
[5] Zhou B F, Feng K Q 2018 RSC Adv. 8 26251
 Google Scholar
Google Scholar
[6] Bell B D C, Murphy S T, Burr P A, Comstock R J, Partezana J M, Grimes R W, Wenman M R 2016 Corros. Sci. 105 36
 Google Scholar
Google Scholar
[7] Nikulina A V 2004 Met. Sci. Heat Treat. 46 458
 Google Scholar
Google Scholar
[8] Ruiz-Hervias J, Simbruner K, Cristobal-Beneyto M, Perez-Gallego D, Zencker U 2021 J. Nucl. Mater. 544 152668
 Google Scholar
Google Scholar
[9] Zhang F L, Chai L J, Qi L, Wang Y Y, Wu L, Pan H C, Teng C Q, Murty K L 2023 J. Nucl. Mater. 577 154284
 Google Scholar
Google Scholar
[10] Qin W, Nam C, Li H L, Szpunar J A 2007 Acta Mater. 55 1695
 Google Scholar
Google Scholar
[11] Liao J J, Yang Z B, Qiu S Y, Peng Q, Li Z C, Zhang J S 2019 J. Nucl. Mater. 524 101
 Google Scholar
Google Scholar
[12] Motta A T, Couet A, Comstock R J 2015 Annu. Rev. Mater. Res. 45 311
 Google Scholar
Google Scholar
[13] Hillner E, Franklin D G, Smee J D 2000 J. Nucl. Mater. 278 334
 Google Scholar
Google Scholar
[14] Couet A, Motta A T, Ambard A 2015 Corros. Sci. 100 73
 Google Scholar
Google Scholar
[15] Yilmazbayhan A, Motta A T, Comstock R J, Sabol G P, Lai B, Cai Z 2004 J. Nucl. Mater. 324 6
 Google Scholar
Google Scholar
[16] Massih A R, Vesterlund G 1992 Nucl. Eng. Des. 137 57
 Google Scholar
Google Scholar
[17] Motta A T 2011 JOM 63 59
 Google Scholar
Google Scholar
[18] Bouineau V, Bénier G, Pêcheur D, Thomazet J, Ambard A, Blat M 2010 Nucl. Technol. 170 444
 Google Scholar
Google Scholar
[19] Takagi I, Une K, Miyamura S, Kobayashi T 2011 J. Nucl. Mater. 419 339
 Google Scholar
Google Scholar
[20] Couet A, Borrel L, Liu J L, Hu J, Grovenor C 2019 Corros. Sci. 159 108134
 Google Scholar
Google Scholar
[21] Hu J, Garner A, Frankel P, Li M, Kirk M A, Lozano-Perez S, Preuss M, Grovenor C 2019 Acta Mater. 173 313
 Google Scholar
Google Scholar
[22] Liu J L, Yu H B, Karamched P, Hu J, He G Z, Goran D, Hughes G M, Wilkinson A J, Lozano-Perez S, Grovenor C R M 2019 Acta Mater. 179 328
 Google Scholar
Google Scholar
[23] Kurpaska L, Favergeon J, Lahoche L, Moulin G, Marssi M El, Roelandt J M 2013 Oxid. Met. 79 261
 Google Scholar
Google Scholar
[24] Xun G, Qingdong L, Wenqing L, Meiyi Y, Bangxin Z 2008 Rare Met. Mater. Eng. 37 1415
 Google Scholar
Google Scholar
[25] Beie H, Mitwalsky A, Garzarolli F, Ruhmann H, Sell H 1994 Zirconium in the Nuclear Industry: Tenth International Symposium (West Conshohocken, PA: ASTM International) pp615–643
[26] Barberis P 1995 J. Nucl. Mater. 226 34
 Google Scholar
Google Scholar
[27] Polatidis E, Frankel P, Wei J, Klaus M, Comstock R J, Ambard A, Lyon S, Cottis R A, Preuss M 2013 J. Nucl. Mater. 432 102
 Google Scholar
Google Scholar
[28] Bouvier P, Godlewski J, Lucazeau G 2002 J. Nucl. Mater. 300 118
 Google Scholar
Google Scholar
[29] Preuss M, Frankel P, Polatidis E, Wei J, Smith J W, Wang C, Cottis R A, Lyon S B, Lozano-Perez S, Hudson D, Ni N, Grovenor C R M, Smith G D W, Sykes J, Cerezo A, Storer S, Fitzpatrick M E 2011 16th International Symposium on Zirconium in the Nuclear Industry Chengdu, China, May 9–13, 2010
[30] Wei J, Frankel P, Polatidis E, Blat M, Ambard A, Comstock R J, Hallstadius L, Hudson D, Smith G D W, Grovenor C R M, Klaus M, Cottis R A, Lyon S, Preuss M 2013 Acta Mater. 61 4200
 Google Scholar
Google Scholar
[31] Garner A, Hu J, Harte A, Frankel P, Grovenor C, Lozano-Perez S, Preuss M 2015 Acta Mater. 99 259
 Google Scholar
Google Scholar
[32] Jeong Y H, Lee K O, Kim H G 2002 J. Nucl. Mater. 302 9
 Google Scholar
Google Scholar
[33] Jeong Y H, Kim H G, Kim T H 2003 J. Nucl. Mater. 317 1
 Google Scholar
Google Scholar
[34] Sakamoto K, Une K, Aomi M, Otsuka T, Hashizume K 2015 J. Nucl. Sci. Technol. 52 1259
 Google Scholar
Google Scholar
[35] Liao J J, Xu F, Peng Q, Yang Z B, Li Z C, Qiu S Y 2020 J. Nucl. Mater. 528 151846
 Google Scholar
Google Scholar
[36] Froideval A, Degueldre C, Segre C U, Pouchon M A, Grolimund D 2008 Corros. Sci. 50 1313
 Google Scholar
Google Scholar
[37] Hulme H, Baxter F, Babu R P, Denecke M A, Gass M, Steuwer A, Norén K, Carlson S, Preuss M 2016 Corros. Sci. 105 202
 Google Scholar
Google Scholar
[38] Wu J W, Xie Y P, Yao M Y, Guan S H, Zhao Y, Pan R J, Wu L, Liu Z P 2023 Phys. Chem. Chem. Phys. 25 8934
 Google Scholar
Google Scholar
[39] Zhao X S, Shang S L, Liu Z K, Shen J Y 2011 J. Nucl. Mater. 415 13
 Google Scholar
Google Scholar
[40] 赵毅, 王栋, 徐晨皓, 吴江桅, 王洋, 谢耀平 2021 上海金属 43 112
 Google Scholar
Google Scholar
Zhao Y, Wang D, Xu C H, Wu J W, Wang Y, Xie Y P 2021 Shanghai Met. 43 112
 Google Scholar
Google Scholar
[41] Kresse G, Hafner J 1993 Phys. Rev. B 47 558
 Google Scholar
Google Scholar
[42] Kresse G, Furthmüller J 1996 Comput. Mater. Sci. 6 15
 Google Scholar
Google Scholar
[43] Kresse G, Furthmüller J 1996 Phys. Rev. B 54 11169
 Google Scholar
Google Scholar
[44] Blöchl P E 1994 Phys. Rev. B 50 17953
 Google Scholar
Google Scholar
[45] Perdew J P, Burke K, Ernzerhof M 1997 Phys. Rev. Lett. 78 1396
 Google Scholar
Google Scholar
[46] Monkhorst H J, Pack J D 1976 Phys. Rev. B 13 5188
 Google Scholar
Google Scholar
[47] Methfessel M, Paxton A T 1989 Phys. Rev. B 40 3616
 Google Scholar
Google Scholar
[48] Barbour O, Crocombette J P, Schuler T, Tupin M 2020 J. Nucl. Mater. 539 152333
 Google Scholar
Google Scholar
[49] Zhang P, Lu Y, He C H, Zhang P 2011 J. Nucl. Mater. 418 143
 Google Scholar
Google Scholar
[50] Fukui H, Fujimoto M, Akahama Y, Sano-Furukawa A, Hattori T 2019 Acta Crystallogr. Sect. B Struct. Sci. Cryst. Eng. Mater. 75 742
 Google Scholar
Google Scholar
[51] Delaforce P M, Yeomans J A, Filkin N C, Wright G J, Thomson R C 2007 J. Am. Ceram. Soc. 90 918
 Google Scholar
Google Scholar
[52] Torres F J, Amigó J M, Alarcón J 2003 J. Solid State Chem. 173 40
 Google Scholar
Google Scholar
[53] Lutterotti L, Scardi P 1990 J. Appl. Crystallogr. 23 246
 Google Scholar
Google Scholar
[54] Bouvier P, Djurado E, Lucazeau G, Le Bihan T 2000 Phys. Rev. B 62 8731
 Google Scholar
Google Scholar
[55] Bondars B, Heidemane G, Grabis J, Laschke K, Boysen H, Schneider J, Frey F 1995 J. Mater. Sci. 30 1621
 Google Scholar
Google Scholar
[56] Puchala B, der Ven A 2013 Phys. Rev. B 88 94108
 Google Scholar
Google Scholar
[57] Fisher E S, Renken C J 1964 Phys. Rev. 135 A482
 Google Scholar
Google Scholar
[58] Anada H, Takeda K 1996 Zirconium in the Nuclear Industry: Eleventh International Symposium (West Conshohocken, PA: ASTM International) pp35–54
[59] Wang C, Zinkevich M, Aldinger F 2004 Calphad 28 281
 Google Scholar
Google Scholar
[60] Chevalier P Y, Fischer E, Cheynet B 2004 Calphad 28 15
 Google Scholar
Google Scholar
[61] Schubert H, Frey F 2005 J. Eur. Ceram. Soc. 25 1597
 Google Scholar
Google Scholar
[62] Becke A D, Edgecombe K E 1990 J. Chem. Phys. 92 5397
 Google Scholar
Google Scholar
计量
- 文章访问数: 5502
- PDF下载量: 119
- 被引次数: 0













 下载:
下载: