-
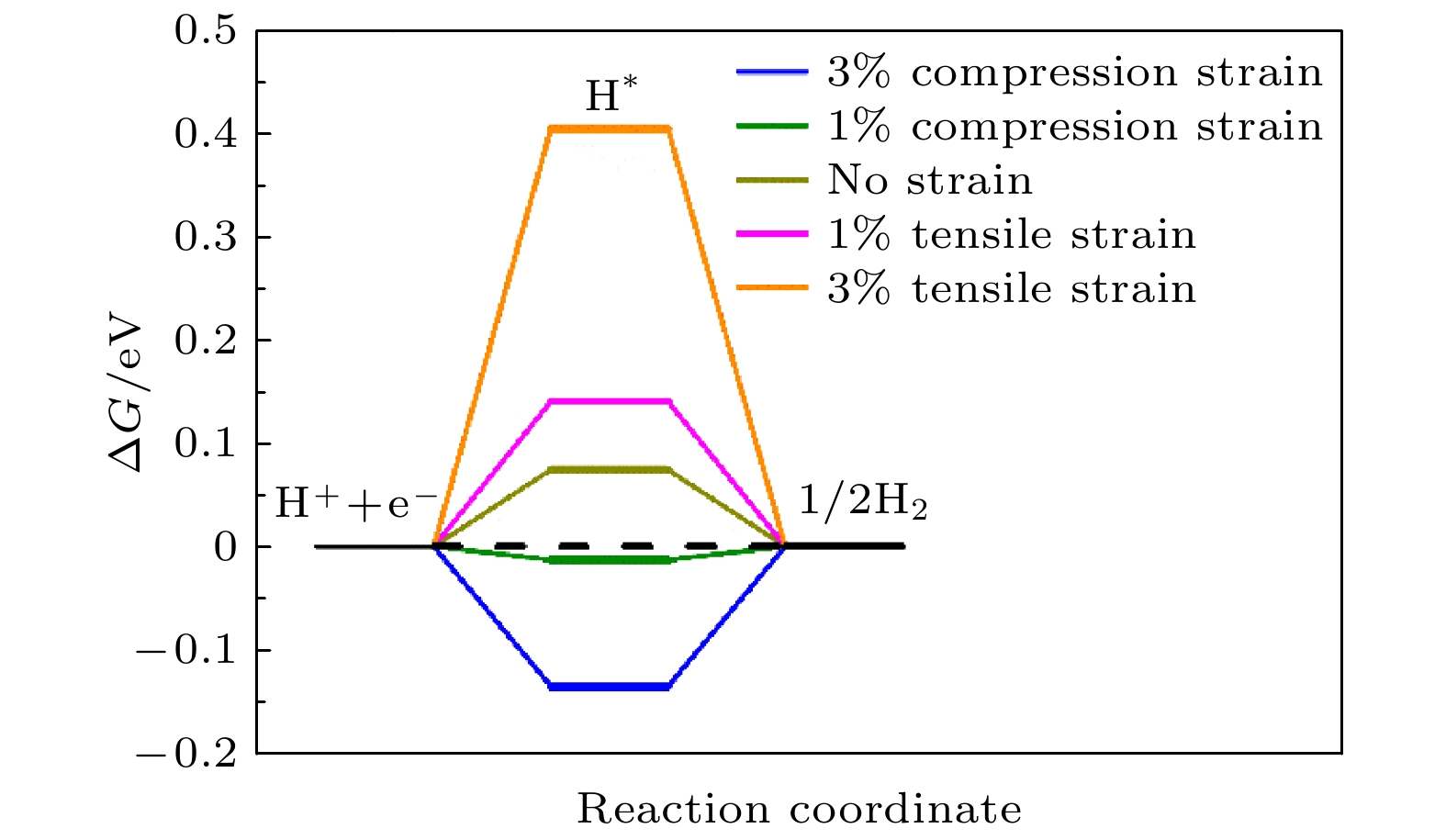
电解水制氢可以将太阳能、风能等可持续能源中的能量转移到氢气这种高能量密度的清洁能源载体中. NiP2作为一种具有较高析氢反应(HER)催化活性的廉价催化剂而备受关注. 本文计算了NiP2(100)表面上氢的吸附能、吉布斯自由能、交换电流密度等属性, 得出其表面上P的top位点是催化活性位点的结论. 并着重分析了掺杂和应变对NiP2催化活性的影响: 1)基于常见的实验手段能产生的应变范围, 计算了1%和3%的拉伸和压缩应变的影响, 发现在NiP2上施加1%的压缩应变可以提高其HER催化性能; 2)计算了过渡金属元素及非金属元素(N, C, S)掺杂对NiP2催化性能的影响, 发现掺杂S可以显著提高P的top位点的HER催化性能, 而过渡金属元素(催化活性: Mn > Mo > W > Co > Cr > Fe > Ni)中Mn的掺杂, 可以将NiP2非活性位点的催化性能提升到跟活性位点同一个数量级, 从而间接提升NiP2的催化性能. 本工作揭示了掺杂和应变对HER催化剂性能影响的微观机理, 为设计高性能HER催化剂提供了指导.Hydrogen production through electrolyzing water can transfer the energy from solar energy, wind energy and other sustainable energy to hydrogen, a clean energy carrier with high energy density. The NiP2 has attracted much attention as a cheap electrocatalyst with high catalytic performance for hydrogen evolution reaction (HER). In this paper, the adsorption energy, Gibbs free energy and exchange current densities at different sites on NiP2 (100) surface are calculated. On this basis, the effect of strain and doping on the HER catalytic performance of NiP2 are studied. By calculation, we find that when H is adsorbed on the top site of P atom on NiP2 (100) surface, the exchange current density is the closest to the top of volcanic curve, so the top site of P atom on NiP2 (100) surface is the catalytic active site. The effect of doping and strain on the catalytic performance of NiP2 are analyzed. 1) According to the range of strain produced by the common experimental technology, the effects of 1% and 3% tensile and compressive strain are calculated. It is found that 1% compressive strain can improve the catalytic performance of NiP2, while when 3% compressive strain or a 1% or 3% tensile strain is applied, the catalytic performance of NiP2 is not enhanced. 2) The effects of doping transition metal elements (Co, Fe, Mn, Mo, Cu, W, Cr) and non-metallic elements (N, C, S) on the catalytic performance of NiP2 are calculated. It is found that doping non-metallic element S can significantly improve the HER catalytic performance of the top site of P atom, while the doping of transition metal elements Mn, Mo, W, Co, Cr, Fe, Cu and non-metallic elements N, C have no effect on this site. The doping of transition metal element (catalytic activity: Mn > Mo > W > Co > Cr > Fe > Ni) Mn can make the catalytic performance of inactive site improved to that of the active site, thus indirectly improving the catalytic performance of NiP2. Our work reveals the micro mechanism of the effect of doping and strain on the performance of HER electrocatalyst, which provides a new perspective for designing the high performance HER electrocatalyst.
[1] Xu X, Pan Y, Zhong Y, Ran R, Shao Z 2020 Mater. Horiz. 7 2519
 Google Scholar
Google Scholar
[2] Chen M N, Xuan Y, Zhang F, He L, Wang X, Pan H Q, Ren J F, Lin Z J 2020 Int. J. Hydrogen Energy 45 14964
 Google Scholar
Google Scholar
[3] Geng C L, Yu X X, Wang P P, Cheng J G, Hong T 2020 J. Eur. Ceram. Soc. 40 3104
 Google Scholar
Google Scholar
[4] Zhu Z W, Zhu L, Li J R, Tang J F, Li G, Hsieh Y K, Wang T H, Wang C F 2016 J. Colloid. Interface Sci. 466 28
 Google Scholar
Google Scholar
[5] Lin X H, Gossenberger F, Groβ A 2016 Ind. Eng. Chem. Res. 55 11107
 Google Scholar
Google Scholar
[6] Lin J, Chen L, Liu T, Xia C R, Chen C S, Zhan Z L 2018 J. Power Sources 374 175
 Google Scholar
Google Scholar
[7] Yue J L, Zhou Y N, Shi S Q, Shadike Z, Huang X Q, Luo J, Yang Z Z, Li H, Gu L, Yang X Q, Fu Z W 2015 Reports 5 8810
 Google Scholar
Google Scholar
[8] Sun M, Xuan Y, Liu G Y, Liu Y L, Zhang F, Ren J F, Chen M N 2020 J. Magn. Magn. Mater. 504 166670
 Google Scholar
Google Scholar
[9] 陈美娜, 张蕾, 高慧颖, 宣言, 任俊峰, 林子敬 2018 物理学报 67 088202
 Google Scholar
Google Scholar
Chen M N, Zhang L, Gao H Y, Xuan Y, Ren J F, Lin Z J 2018 Acta Phys. Sin. 67 088202
 Google Scholar
Google Scholar
[10] Chen M N, Gao H Y, Zhang L, Xuan Y, Ren J F, Ni M, Lin Z J 2019 Ceram. Int. 45 3977
 Google Scholar
Google Scholar
[11] He L, Xuan Y, Zhang F, Wang X, Pan H Q, Ren J F, Chen M N 2020 Int. J. Hydrogen Energy 46 1096
 Google Scholar
Google Scholar
[12] Wei Z L, Hou J, Zhu Z W 2016 J. Alloys. Compd. 683 474
 Google Scholar
Google Scholar
[13] Yang W C, Bi Y J, Qin Y P, Liu Y, Zhang X H, Yang B C, Wu Q, Wang D Y, Shi S Q 2015 J. Power Sources 275 785
 Google Scholar
Google Scholar
[14] Shi S Q, Gao J, Liu Y, Zhao Y, Wu Q, Ju W W, Ouyang C Y, Xiao R J 2016 Chin. Phys. B 25 018212
 Google Scholar
Google Scholar
[15] He W J, Han L L, Hao Q Y, Zheng X R, Li Y, Zhang J, Liu C C, Liu H, Xin H L 2019 ACS Energy Lett. 4 2905
 Google Scholar
Google Scholar
[16] Conway B E, Tilak B V 2002 Electrochim. Acta 47 3571
 Google Scholar
Google Scholar
[17] Ouyang T, Chen A N, He Z Z, Liu Z Q, Tong Y X 2018 Chem. Commun. 54 9901
 Google Scholar
Google Scholar
[18] Gong S Q, Jiang Z J, Shi P H, Fan J C, Xu Q J, Min Y L 2018 Appl. Catal. B 238 318
 Google Scholar
Google Scholar
[19] Zhang Y Y, Lei H W, Duan D L, Villota E, Liu C, Ruan R 2018 ACS Appl. Mater. Interfaces 10 20429
 Google Scholar
Google Scholar
[20] Xiao P, Sk M A, Thia L, Ge X M, Lim R J, Wang J Y, Lim K H, Wang X 2014 Energy Environ. Sci. 7 2624
 Google Scholar
Google Scholar
[21] Aravind S S J, Ramanujachary K, Mugweru A, Vaden T D 2015 Appl. Catal. A Gen. 490 101
 Google Scholar
Google Scholar
[22] Wu Z X, Wang J, Xia K D, Lei W, Liu X, Wang D L 2018 J. Mater. Chem. A 6 616
 Google Scholar
Google Scholar
[23] Xu Y L, Yan M F, Liu Z, Wang J Y, Zhai Z Z, Ren B, Dong X X, Miao J F, Liu Z F 2020 Electrochim. Acta 363 137151
 Google Scholar
Google Scholar
[24] Wang J B, Chen W L, Wang T, Bate N, Wang C, Wang E 2018 Nano Res. 011 4535
 Google Scholar
Google Scholar
[25] Harnisch F, Sievers G, Schrder U 2009 Appl. Catal. B 89 455
 Google Scholar
Google Scholar
[26] Ojha K, Saha S, Kumar B, Hazra K S, Ganguli A K 2016 Chemcatchem 8 1218
 Google Scholar
Google Scholar
[27] Han L, Dong S J, Wang E 2016 Adv. Mater. 28 9266
 Google Scholar
Google Scholar
[28] Wang X D, Zhou H P, Zhang D K, Pi M Y, Feng J J, Chen S J 2018 J. Power Sources 387 1
 Google Scholar
Google Scholar
[29] Tian H, Wang X D, Li H Y, Pi M Y, Zhang D K, Chen S J 2020 Energy Technol. 8 1900936
 Google Scholar
Google Scholar
[30] Owens-Baird B, Sousa J P S, Ziouani Y, Petrovykh D Y, Zarkevich N A, Johnson D D, Kolen'Ko Y V, Kovnir K 2020 Chem. Sci. 11 5007
 Google Scholar
Google Scholar
[31] Wang T T, Guo X S, Zhang J Y, Xiao W, Xi P X, Peng S L, Gao D Q 2019 J. Mater. Chem. A 7 4971
 Google Scholar
Google Scholar
[32] Jain A, Sadan M B, Ramasubramaniam A 2020 J. Phys. Chem. C 124 12324
 Google Scholar
Google Scholar
[33] Li Z, Feng Y, Liang Y L, Cheng C Q, Dong C K, Liu H, Du X W 2020 Adv. Mater. 32 1908521
 Google Scholar
Google Scholar
[34] Ling T, Yan D Y, Wang H, Jiao Y, Hu Z P, Zheng Y, Zheng L R, Mao J, Liu H, Du X W, Jaroniec M, Qiao S Z 2017 Nat. Commun. 8 1509
 Google Scholar
Google Scholar
[35] Wu J B, Li P P, Pan Y T, Warren S, Yin X, Yang H 2012 Chem. Soc. Rev. 41 8066
 Google Scholar
Google Scholar
[36] Navickas E, Chen Y, Lu Q Y, Wallisch W, Huber T M, Bernardi J, Stöger-Pollach M, Friedbacher G, Hutter H, Yildiz B, Fleig J 2017 ACS Nano 11 11475
 Google Scholar
Google Scholar
[37] Gopal C B, García-Melchor M, Lee S C, Shi Y Z, Shavorskiy A, Monti M, Guan Z X, Sinclair R, Bluhm H, Vojvodic A, Chueh W C 2017 Nat. Commun. 8 15360
 Google Scholar
Google Scholar
[38] Kato H, Tottori Y, Sasaki K 2014 Exp. Mech. 54 489
 Google Scholar
Google Scholar
[39] Castellanos-Gomez A, Roldán R, Cappelluti E, Buscema M, Guinea F, Zant H S J, Steele G A 2013 Nano Lett. 13 5361
 Google Scholar
Google Scholar
[40] Du M S, Cui L S, Cao Y, Bard A J 2015 J. Am. Chem. Soc. 137 7397
 Google Scholar
Google Scholar
[41] Kresse G, Furthmüller J 1996 Phys. Rev. B Condens. Matter. 54 11169
 Google Scholar
Google Scholar
[42] Perdew J P, Burke K, Ernzerhof M 1998 Phys. Rev. Lett. 77 3865
 Google Scholar
Google Scholar
[43] He B, Chi S, Ye A, Mi P, Zhang L, Pu B, Zou Z, Ran Y, Zhao Q, Wang D 2020 Scientific Data 7 151
 Google Scholar
Google Scholar
[44] Hansen M H, Stern L A, Feng L G, Rossmeisl J, Hu X L 2015 Phys. Chem. Chem. Phys. 17 10823
 Google Scholar
Google Scholar
[45] Qu Y J, Pan H, Kwok C T, Wang Z S 2015 Phys. Chem. Chem. Phys. 17 24820
 Google Scholar
Google Scholar
[46] Medford A J, Vojvodic A, Hummelshøj J S, Voss J, Abild-Pedersen F, Studt F, Bligaard T, Nilsson A, Nørskov J K 2015 J. Catal. 328 36
 Google Scholar
Google Scholar
[47] Zhang Y, Jiao Y, Jaroniec M, Qiao S Z 2014 Angew. Chem. Int. Ed. Engl. 54 52
 Google Scholar
Google Scholar
[48] Tsai C, Chan K, Abild-Pedersen F, NøRskov J K 2014 Phys. Chem. Chem. Phys. 16 13156
 Google Scholar
Google Scholar
[49] Jiao Y, Zheng Y, Davey K, Qiao S Z 2016 Nat. Energy 1 16130
 Google Scholar
Google Scholar
[50] Noerskov J K, Bligaard T, Logadottir A, Kitchin J R, Chen J G, Pandelov S, Stimming U 2005 J. Cheminform 36 e12154
 Google Scholar
Google Scholar
[51] Liu X, Zhang L, Zheng Y, Guo Z, Zhu Y M, Chen H J, Li F, Liu P P, Yu B, Wang X W, Liu J, Chen Y 2019 Adv. Sci. 6 1801898
 Google Scholar
Google Scholar
[52] Wang L, Zeng Z H, Gao W P, Maxson T, Raciti D, Giroux M, Pan X Q, Wang C, Greeley J 2019 Science 363 870
 Google Scholar
Google Scholar
[53] Luo M C, Guo S J 2017 Nat. Rev. Mater. 2 17059
 Google Scholar
Google Scholar
[54] Zhu H, Gao G H, Du M L, Zhou J H, Wang K, Wu W B, Chen X, Li Y, Ma P M, Dong W F, Duan F, Chen M Q, Wu G M, Wu J D, Yang H T, Guo S J 2018 Adv. Mater. 30 1707301
 Google Scholar
Google Scholar
[55] Potapenko D V, Li Z S, Kysar J W, Osgood R M 2014 Nano Lett. 14 6185
 Google Scholar
Google Scholar
-
图 3 当电荷转移系数α = 0.45(黑色曲线)时, NiP2 (100)表面ΔG与
${j_0}$ 的火山型曲线, 空心符号表示通过吉布斯自由能获得的单个位点的理论交换电流密度Fig. 3. Volcano plot between
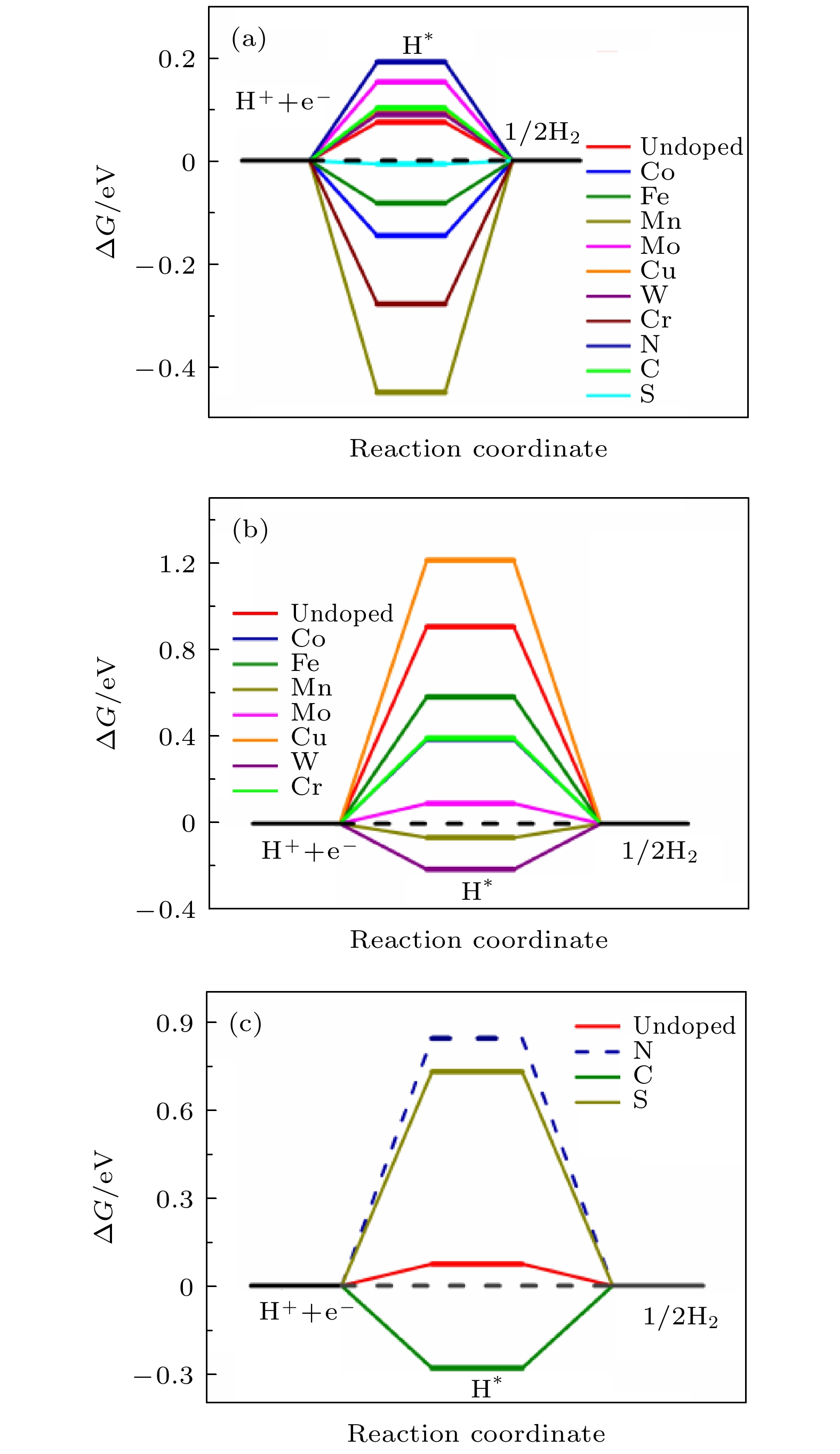
${j_0}$ and ΔG with charge-transfer coefficient α = 0.45 (black solid line) of NiP2 (100) surface. The hollow symbols represent the theoretical exchange current density of a single site obtained by the value of Gibbs free energy.图 4 (a) 过渡金属和非金属元素掺杂前后H吸附在NiP2(100)表面位点1(P的top位点)上和掺杂原子上的ΔG; (b) 过渡金属掺杂前后H吸附在NiP2(100)表面位点2(Ni的top位点)上和掺杂的过渡金属原子上的ΔG; (c)掺杂非金属原子前后H分别吸附在NiP2(100)表面位点1(P的top位点)和掺杂的非金属原子上的ΔG; 虚线表示H吸附位点有所偏移
Fig. 4. (a) ΔG of H adsorbed on site 1 (top site of P atom) of NiP2 (100) surface before and after doping, (b) ΔG of H adsorbed on site 2 (top site of Ni atom) and top site of doped transition metal atom before and after doping transition metal atom respectively, (c) ΔG of H adsorbed on site 1 (top site of P atom) and top site of doped non-metallic atom on NiP2 (100) surface before and after doping non-metallic atom respectively. The dashed line indicates the shift of H adsorption site.
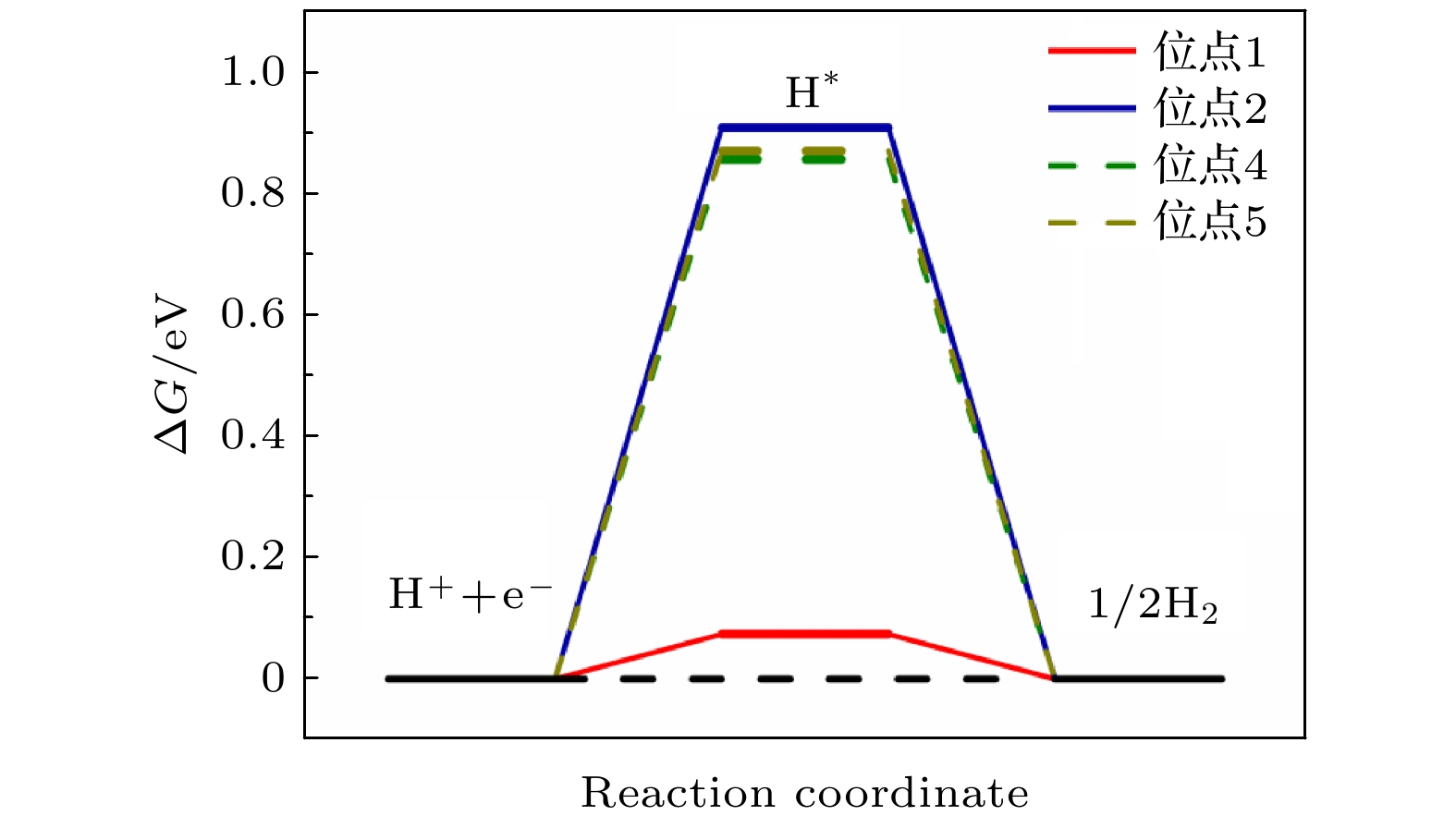
表 1 NiP2 (100)表面的不同位点的吸附能ΔE
Table 1. Adsorption energy (ΔE ) of different sites of NiP2 (100) surface.
位点 ΔE/eV 1 –0.209 2 0.674 3 移动到位点1 4 0.596 5 0.614 -
[1] Xu X, Pan Y, Zhong Y, Ran R, Shao Z 2020 Mater. Horiz. 7 2519
 Google Scholar
Google Scholar
[2] Chen M N, Xuan Y, Zhang F, He L, Wang X, Pan H Q, Ren J F, Lin Z J 2020 Int. J. Hydrogen Energy 45 14964
 Google Scholar
Google Scholar
[3] Geng C L, Yu X X, Wang P P, Cheng J G, Hong T 2020 J. Eur. Ceram. Soc. 40 3104
 Google Scholar
Google Scholar
[4] Zhu Z W, Zhu L, Li J R, Tang J F, Li G, Hsieh Y K, Wang T H, Wang C F 2016 J. Colloid. Interface Sci. 466 28
 Google Scholar
Google Scholar
[5] Lin X H, Gossenberger F, Groβ A 2016 Ind. Eng. Chem. Res. 55 11107
 Google Scholar
Google Scholar
[6] Lin J, Chen L, Liu T, Xia C R, Chen C S, Zhan Z L 2018 J. Power Sources 374 175
 Google Scholar
Google Scholar
[7] Yue J L, Zhou Y N, Shi S Q, Shadike Z, Huang X Q, Luo J, Yang Z Z, Li H, Gu L, Yang X Q, Fu Z W 2015 Reports 5 8810
 Google Scholar
Google Scholar
[8] Sun M, Xuan Y, Liu G Y, Liu Y L, Zhang F, Ren J F, Chen M N 2020 J. Magn. Magn. Mater. 504 166670
 Google Scholar
Google Scholar
[9] 陈美娜, 张蕾, 高慧颖, 宣言, 任俊峰, 林子敬 2018 物理学报 67 088202
 Google Scholar
Google Scholar
Chen M N, Zhang L, Gao H Y, Xuan Y, Ren J F, Lin Z J 2018 Acta Phys. Sin. 67 088202
 Google Scholar
Google Scholar
[10] Chen M N, Gao H Y, Zhang L, Xuan Y, Ren J F, Ni M, Lin Z J 2019 Ceram. Int. 45 3977
 Google Scholar
Google Scholar
[11] He L, Xuan Y, Zhang F, Wang X, Pan H Q, Ren J F, Chen M N 2020 Int. J. Hydrogen Energy 46 1096
 Google Scholar
Google Scholar
[12] Wei Z L, Hou J, Zhu Z W 2016 J. Alloys. Compd. 683 474
 Google Scholar
Google Scholar
[13] Yang W C, Bi Y J, Qin Y P, Liu Y, Zhang X H, Yang B C, Wu Q, Wang D Y, Shi S Q 2015 J. Power Sources 275 785
 Google Scholar
Google Scholar
[14] Shi S Q, Gao J, Liu Y, Zhao Y, Wu Q, Ju W W, Ouyang C Y, Xiao R J 2016 Chin. Phys. B 25 018212
 Google Scholar
Google Scholar
[15] He W J, Han L L, Hao Q Y, Zheng X R, Li Y, Zhang J, Liu C C, Liu H, Xin H L 2019 ACS Energy Lett. 4 2905
 Google Scholar
Google Scholar
[16] Conway B E, Tilak B V 2002 Electrochim. Acta 47 3571
 Google Scholar
Google Scholar
[17] Ouyang T, Chen A N, He Z Z, Liu Z Q, Tong Y X 2018 Chem. Commun. 54 9901
 Google Scholar
Google Scholar
[18] Gong S Q, Jiang Z J, Shi P H, Fan J C, Xu Q J, Min Y L 2018 Appl. Catal. B 238 318
 Google Scholar
Google Scholar
[19] Zhang Y Y, Lei H W, Duan D L, Villota E, Liu C, Ruan R 2018 ACS Appl. Mater. Interfaces 10 20429
 Google Scholar
Google Scholar
[20] Xiao P, Sk M A, Thia L, Ge X M, Lim R J, Wang J Y, Lim K H, Wang X 2014 Energy Environ. Sci. 7 2624
 Google Scholar
Google Scholar
[21] Aravind S S J, Ramanujachary K, Mugweru A, Vaden T D 2015 Appl. Catal. A Gen. 490 101
 Google Scholar
Google Scholar
[22] Wu Z X, Wang J, Xia K D, Lei W, Liu X, Wang D L 2018 J. Mater. Chem. A 6 616
 Google Scholar
Google Scholar
[23] Xu Y L, Yan M F, Liu Z, Wang J Y, Zhai Z Z, Ren B, Dong X X, Miao J F, Liu Z F 2020 Electrochim. Acta 363 137151
 Google Scholar
Google Scholar
[24] Wang J B, Chen W L, Wang T, Bate N, Wang C, Wang E 2018 Nano Res. 011 4535
 Google Scholar
Google Scholar
[25] Harnisch F, Sievers G, Schrder U 2009 Appl. Catal. B 89 455
 Google Scholar
Google Scholar
[26] Ojha K, Saha S, Kumar B, Hazra K S, Ganguli A K 2016 Chemcatchem 8 1218
 Google Scholar
Google Scholar
[27] Han L, Dong S J, Wang E 2016 Adv. Mater. 28 9266
 Google Scholar
Google Scholar
[28] Wang X D, Zhou H P, Zhang D K, Pi M Y, Feng J J, Chen S J 2018 J. Power Sources 387 1
 Google Scholar
Google Scholar
[29] Tian H, Wang X D, Li H Y, Pi M Y, Zhang D K, Chen S J 2020 Energy Technol. 8 1900936
 Google Scholar
Google Scholar
[30] Owens-Baird B, Sousa J P S, Ziouani Y, Petrovykh D Y, Zarkevich N A, Johnson D D, Kolen'Ko Y V, Kovnir K 2020 Chem. Sci. 11 5007
 Google Scholar
Google Scholar
[31] Wang T T, Guo X S, Zhang J Y, Xiao W, Xi P X, Peng S L, Gao D Q 2019 J. Mater. Chem. A 7 4971
 Google Scholar
Google Scholar
[32] Jain A, Sadan M B, Ramasubramaniam A 2020 J. Phys. Chem. C 124 12324
 Google Scholar
Google Scholar
[33] Li Z, Feng Y, Liang Y L, Cheng C Q, Dong C K, Liu H, Du X W 2020 Adv. Mater. 32 1908521
 Google Scholar
Google Scholar
[34] Ling T, Yan D Y, Wang H, Jiao Y, Hu Z P, Zheng Y, Zheng L R, Mao J, Liu H, Du X W, Jaroniec M, Qiao S Z 2017 Nat. Commun. 8 1509
 Google Scholar
Google Scholar
[35] Wu J B, Li P P, Pan Y T, Warren S, Yin X, Yang H 2012 Chem. Soc. Rev. 41 8066
 Google Scholar
Google Scholar
[36] Navickas E, Chen Y, Lu Q Y, Wallisch W, Huber T M, Bernardi J, Stöger-Pollach M, Friedbacher G, Hutter H, Yildiz B, Fleig J 2017 ACS Nano 11 11475
 Google Scholar
Google Scholar
[37] Gopal C B, García-Melchor M, Lee S C, Shi Y Z, Shavorskiy A, Monti M, Guan Z X, Sinclair R, Bluhm H, Vojvodic A, Chueh W C 2017 Nat. Commun. 8 15360
 Google Scholar
Google Scholar
[38] Kato H, Tottori Y, Sasaki K 2014 Exp. Mech. 54 489
 Google Scholar
Google Scholar
[39] Castellanos-Gomez A, Roldán R, Cappelluti E, Buscema M, Guinea F, Zant H S J, Steele G A 2013 Nano Lett. 13 5361
 Google Scholar
Google Scholar
[40] Du M S, Cui L S, Cao Y, Bard A J 2015 J. Am. Chem. Soc. 137 7397
 Google Scholar
Google Scholar
[41] Kresse G, Furthmüller J 1996 Phys. Rev. B Condens. Matter. 54 11169
 Google Scholar
Google Scholar
[42] Perdew J P, Burke K, Ernzerhof M 1998 Phys. Rev. Lett. 77 3865
 Google Scholar
Google Scholar
[43] He B, Chi S, Ye A, Mi P, Zhang L, Pu B, Zou Z, Ran Y, Zhao Q, Wang D 2020 Scientific Data 7 151
 Google Scholar
Google Scholar
[44] Hansen M H, Stern L A, Feng L G, Rossmeisl J, Hu X L 2015 Phys. Chem. Chem. Phys. 17 10823
 Google Scholar
Google Scholar
[45] Qu Y J, Pan H, Kwok C T, Wang Z S 2015 Phys. Chem. Chem. Phys. 17 24820
 Google Scholar
Google Scholar
[46] Medford A J, Vojvodic A, Hummelshøj J S, Voss J, Abild-Pedersen F, Studt F, Bligaard T, Nilsson A, Nørskov J K 2015 J. Catal. 328 36
 Google Scholar
Google Scholar
[47] Zhang Y, Jiao Y, Jaroniec M, Qiao S Z 2014 Angew. Chem. Int. Ed. Engl. 54 52
 Google Scholar
Google Scholar
[48] Tsai C, Chan K, Abild-Pedersen F, NøRskov J K 2014 Phys. Chem. Chem. Phys. 16 13156
 Google Scholar
Google Scholar
[49] Jiao Y, Zheng Y, Davey K, Qiao S Z 2016 Nat. Energy 1 16130
 Google Scholar
Google Scholar
[50] Noerskov J K, Bligaard T, Logadottir A, Kitchin J R, Chen J G, Pandelov S, Stimming U 2005 J. Cheminform 36 e12154
 Google Scholar
Google Scholar
[51] Liu X, Zhang L, Zheng Y, Guo Z, Zhu Y M, Chen H J, Li F, Liu P P, Yu B, Wang X W, Liu J, Chen Y 2019 Adv. Sci. 6 1801898
 Google Scholar
Google Scholar
[52] Wang L, Zeng Z H, Gao W P, Maxson T, Raciti D, Giroux M, Pan X Q, Wang C, Greeley J 2019 Science 363 870
 Google Scholar
Google Scholar
[53] Luo M C, Guo S J 2017 Nat. Rev. Mater. 2 17059
 Google Scholar
Google Scholar
[54] Zhu H, Gao G H, Du M L, Zhou J H, Wang K, Wu W B, Chen X, Li Y, Ma P M, Dong W F, Duan F, Chen M Q, Wu G M, Wu J D, Yang H T, Guo S J 2018 Adv. Mater. 30 1707301
 Google Scholar
Google Scholar
[55] Potapenko D V, Li Z S, Kysar J W, Osgood R M 2014 Nano Lett. 14 6185
 Google Scholar
Google Scholar
计量
- 文章访问数: 9381
- PDF下载量: 150
- 被引次数: 0














 下载:
下载: