-
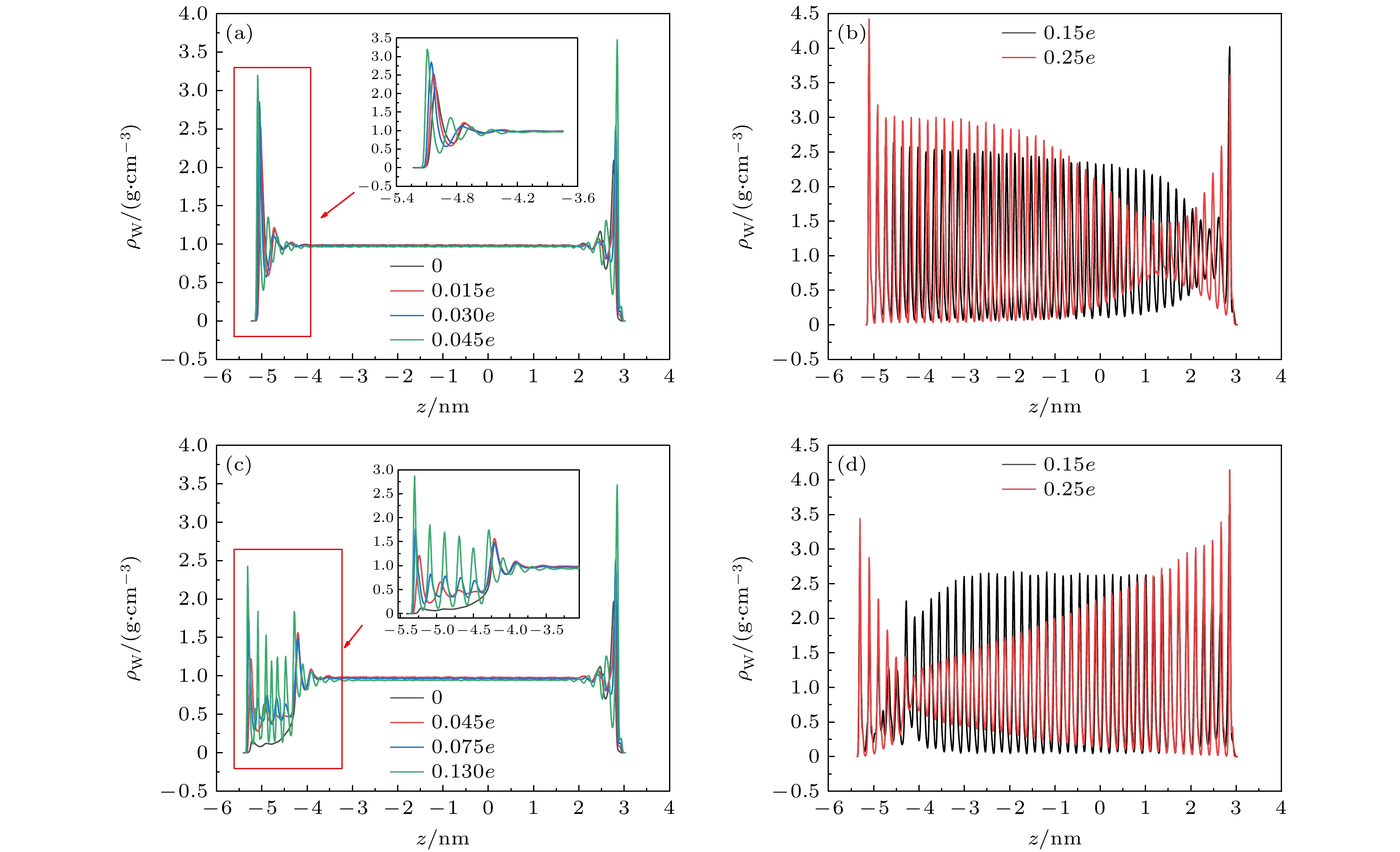
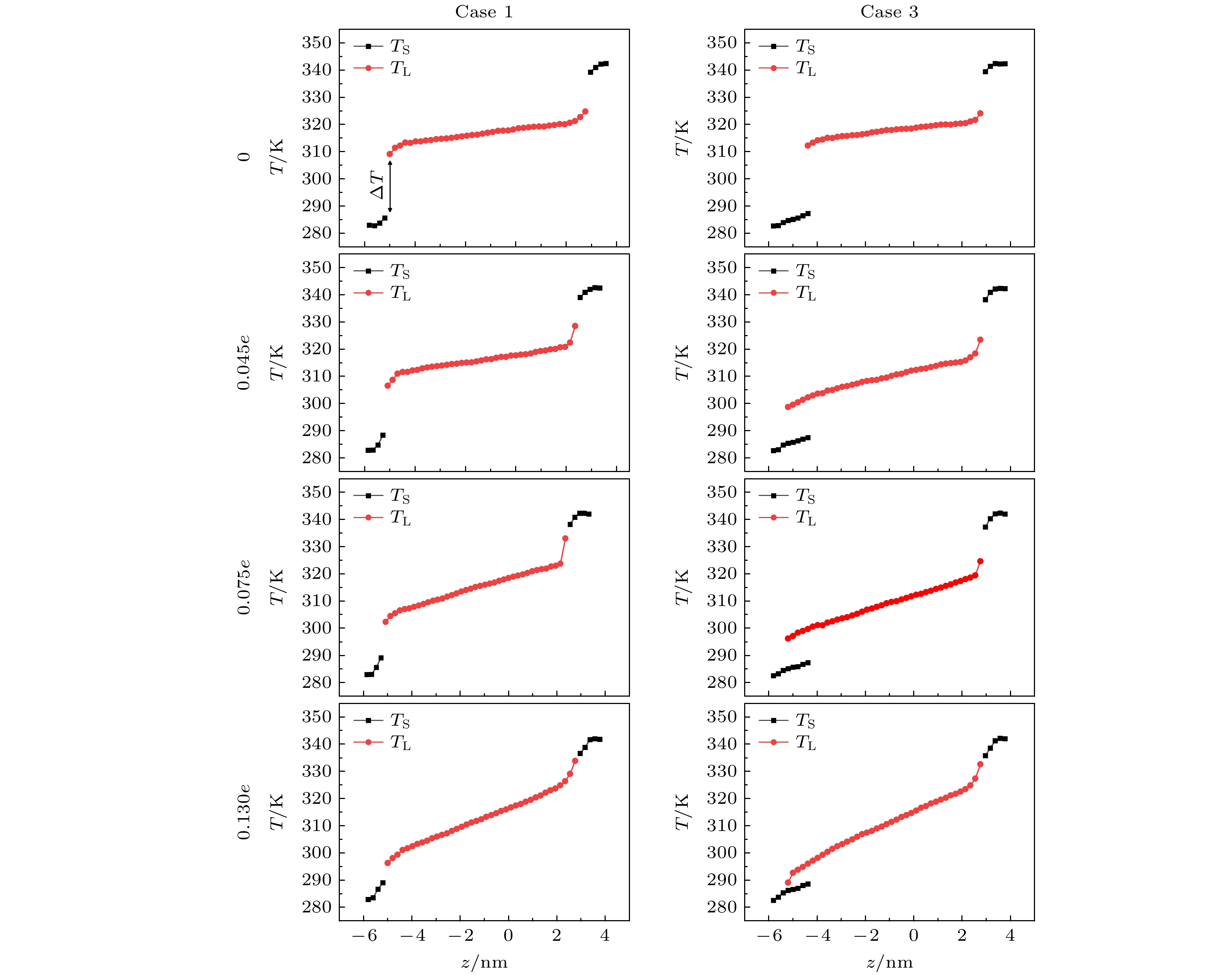
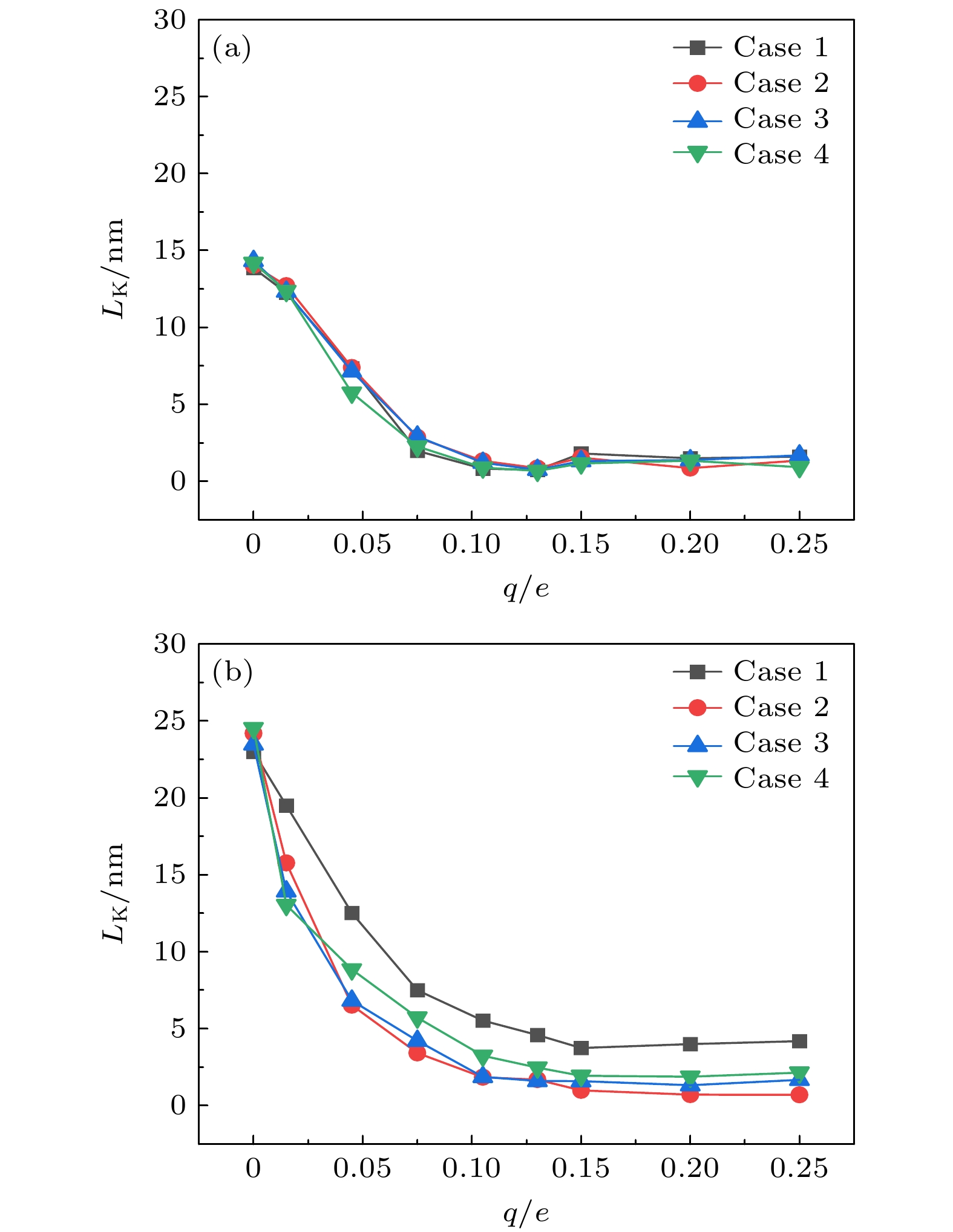
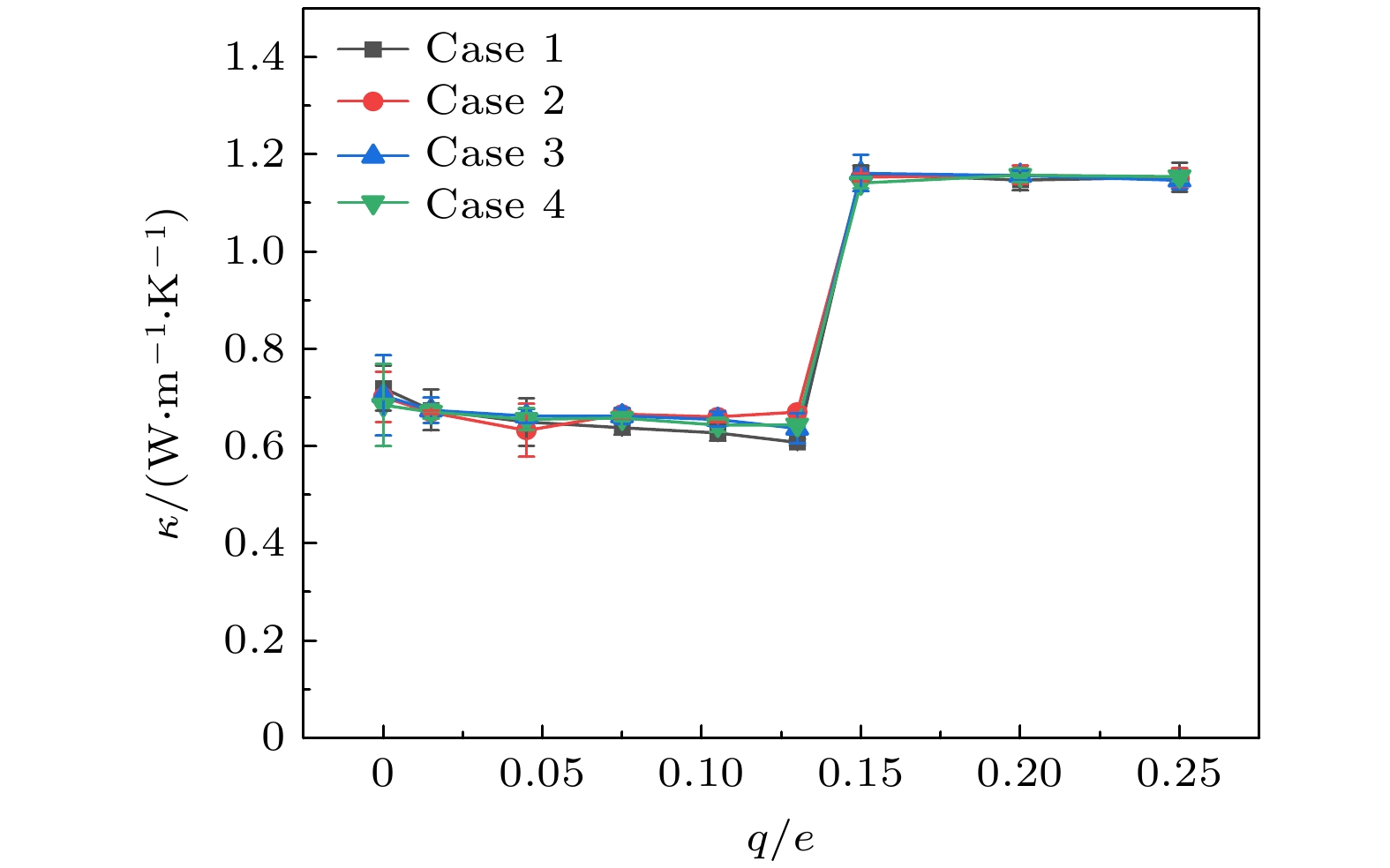
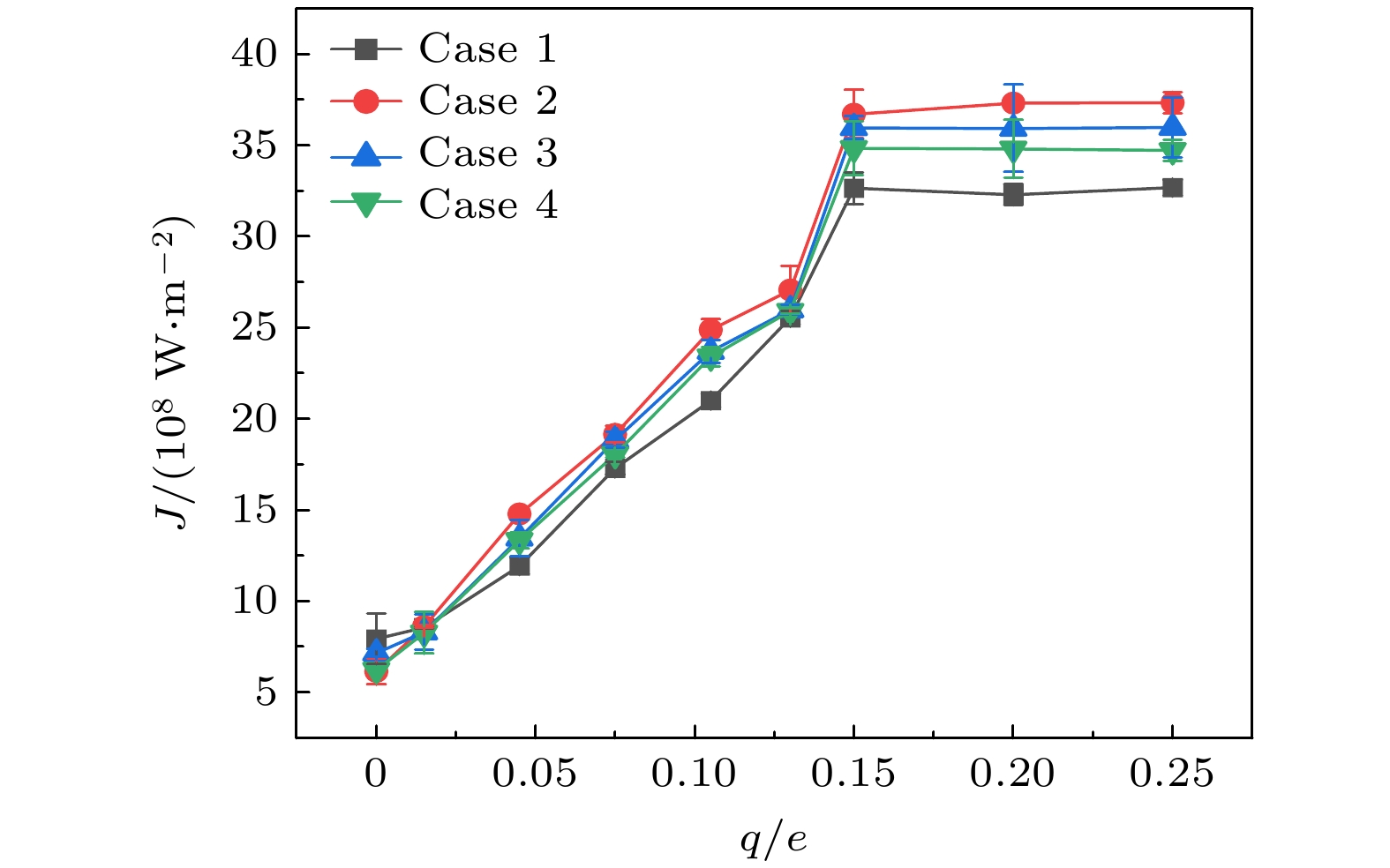
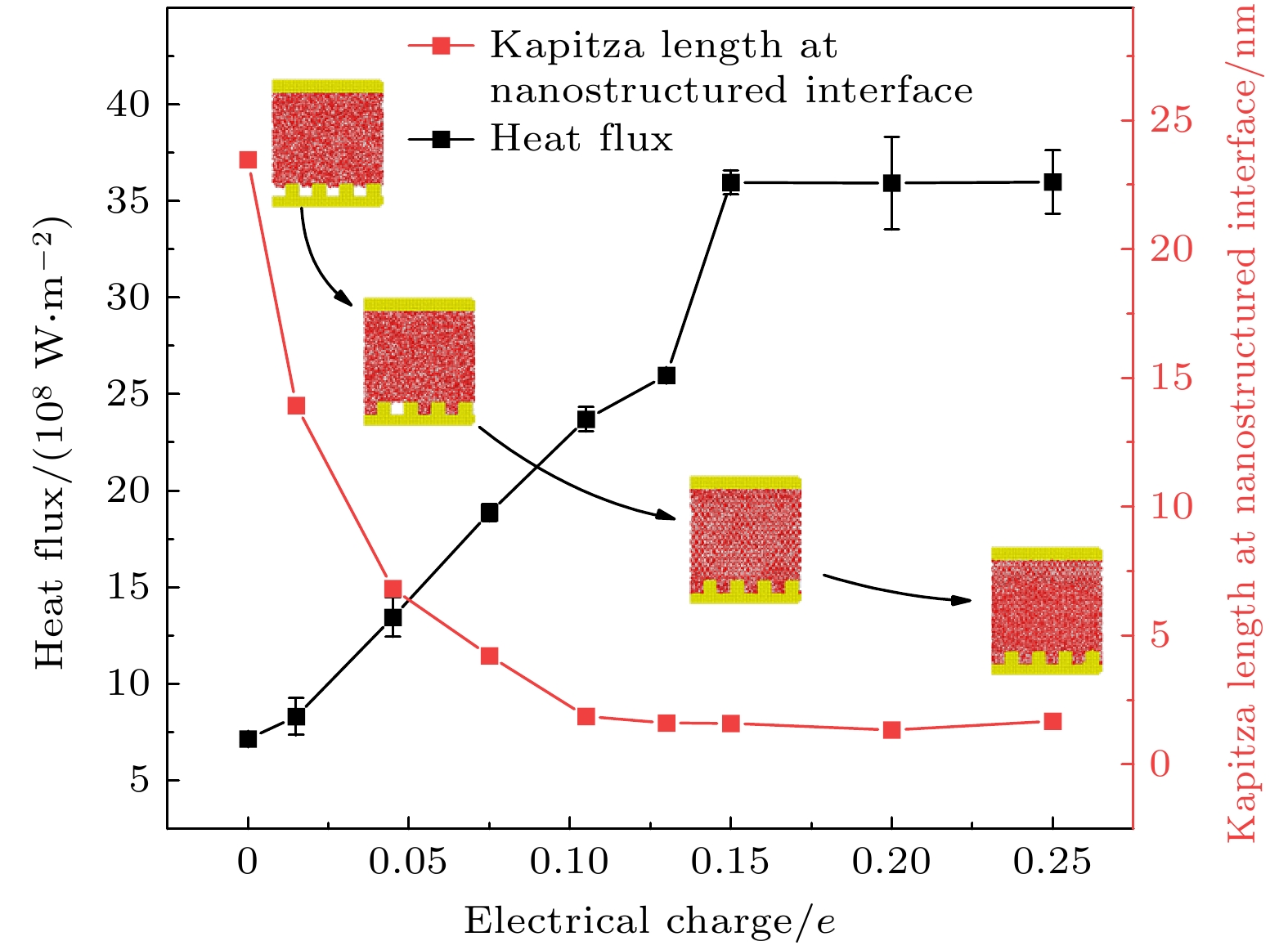
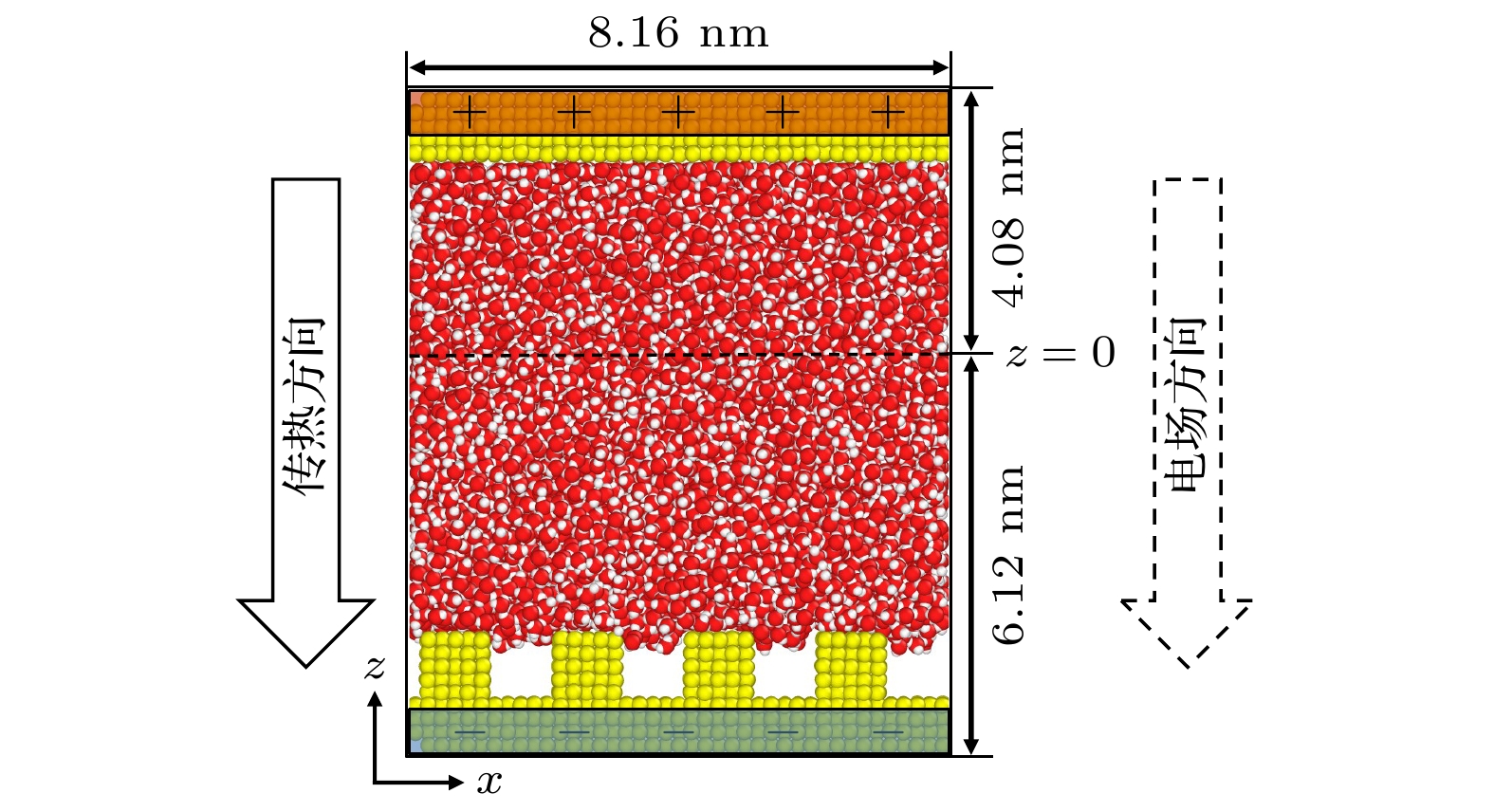
在固体表面布置纳米结构是一种强化固-液界面传热的简单有效的方法. 但是, 当固-液界面相互作用较弱时, 由于纳米结构并不能被液体浸润, 纳米结构的存在反而会弱化固-液界面之间的传热, 而外电场的施加则可以解决这一问题. 本文基于分子动力学模拟的方法, 研究了纳米结构固-液界面在外电场作用下的传热特性. 通过在2块平行金属板布置数量相同的正负电荷, 产生垂直于板面的均匀电场, 并在下层金属板上布置了不同尺寸的纳米结构. 结果表明: 在外电场作用下, 纳米结构处会产生电润湿现象, 固-液界面的润湿状态能够从Cassie态变为Wenzel态, 界面处的Kapitza热阻长度明显减小, 因而热流密度显著增大; 当电荷量增至发生电冻结的临界值, 液态水会产生电冻结现象, 其热导率骤增至1.2 W/(m·K), 热流密度也随之发生骤增; 继续增加电场强度, 由于电冻结现象的发生, 固-液界面热阻则基本保持不变.With the size of high-performance electronic device decreasing (down to nanoscale), and the accompanying heat dissipation becomes a big problem due to its extremely high heat generation density. To tackle the ever-demanding heat dissipation requirement, intensive work has been done to develop techniques for chip-level cooling. Among the techniques reported in the literature, liquid cooling appears to be a good candidate for cooling high-performance electronic devices. However, when the device size is reduced to the sub-micro or nanometer level, the thermal resistance on the solid-liquid interface cannot be ignored in the heat transfer process. Usually, the interfacial thermal transport can be enhanced by using nanostructures on the solid surface because of the confinement effect of the fluid molecules filling up the nano-grooves and the increase of the solid-liquid interfacial contact area. However, in the case of weak interfacial couplings, the fluid molecules cannot enter into the nano-grooves and the interfacial thermal transport is suppressed. In the present work, the heat transfer system between two parallel metal plates filled with deionized water is investigated by molecular dynamics simulation. Electronic charges are applied to the upper plate and lower plate to create a uniform electric field that is perpendicular to the surface, and three types of nanostructures with varying size are arranged on the lower plate. It is found that the wetting state at the solid-liquid interface can change from Cassie state into Wenzel state with strength of the electric field increasing. Owing to the transition from the dewetting state to wetting state (from Wenzel to Cassie wetting state), the Kapitza length can be degraded and the solid-liquid interfacial heat transfer can be enhanced. The mechanism of the enhancing hart transfer is discussed based on the calculation of the number density distribution of the water molecules between the two plates. When the charge is further increased, electrofreezing appears, and a solid hydrogen bonding network is formed in the system, resulting in the thermal conductivity increasing to 1.2 W/(m·K) while the thermal conductivity remains almost constant when the electric charge continues to increase.
-
Keywords:
- electrowetting /
- nanostructure /
- external electric field /
- solid-liquid interfacial thermal resistance /
- molecular dynamics
[1] Razeeb K M, Dalton E, Cross G L W, Robinson A J 2018 Int. Mater. 63 1
 Google Scholar
Google Scholar
[2] Pop E 2010 Nano Res. 3 147
 Google Scholar
Google Scholar
[3] Kapitza P L 1971 J. Phys. U.S.S.R. 4 181
 Google Scholar
Google Scholar
[4] Shenogina N, Godawat R, Keblinski P, Garde S 2009 Phys. Rev. Lett. 102 156101
 Google Scholar
Google Scholar
[5] Harikrishna H, Ducker W A, Huxtable S T 2013 Appl. Phys. Lett. 102 251606
 Google Scholar
Google Scholar
[6] Park S C, Cho H R, Kim D, Choi S H, Choi C, Yu D I 2024 Int. J. Heat Fluid Flow 107 109388
 Google Scholar
Google Scholar
[7] Song G, Min C 2013 Mol. Phys. 111 903
 Google Scholar
Google Scholar
[8] Rashidi M M, Ghahremanian S, Toghraie D, Roy P 2020 Int. Commun. Heat Mass 117 140741
 Google Scholar
Google Scholar
[9] 张程宾, 许兆林, 陈永平 2014 物理学报 63 214706
 Google Scholar
Google Scholar
Zhang C B, Xu Z L, Chen Y P 2014 Acta Phys. Sin. 63 214706
 Google Scholar
Google Scholar
[10] Chakraborty P, Ma T, Cao L, Wang Y 2019 Int. J. Heat Mass. Tran. 136 702
 Google Scholar
Google Scholar
[11] Yao S T, Wang J S, Jin S F, Tan F G, Chen S P 2024 Int. J. Therm. Sci. 203 109161
 Google Scholar
Google Scholar
[12] Qin S Y, Chen Z X, Wang Q, Li W G, Xing H W 2024 Int. Commun. Heat Mass 151 107257
 Google Scholar
Google Scholar
[13] Cassie A B D 1948 Disscussions of the Faraday Society 3 11
 Google Scholar
Google Scholar
[14] Wenzel R N 1936 Ind. Eng. Chem. 28 988
 Google Scholar
Google Scholar
[15] Bormashenko E 2015 Adv. Colloid Interface Sci. 222 92
 Google Scholar
Google Scholar
[16] Bormashenko E, Pogreb R, Stein T, Whyman G, Erlich M, Musin A, Machavariani V, Aurbach D 2008 Phys. Chem. Chem. Phys 10 4056
 Google Scholar
Google Scholar
[17] 李文, 马骁婧, 徐进良, 王艳, 雷骏鹏 2015 物理学报 70 126101
 Google Scholar
Google Scholar
Li W, Ma X J, Xu J L, Wang Y, Lei J P 2015 Acta Phys. Sin. 70 126101
 Google Scholar
Google Scholar
[18] Sur A, Lu Y, Pascente C, Ruchhoeft P, Liu D 2018 Int. J. Heat Mass Tran. 120 202
 Google Scholar
Google Scholar
[19] Lippmann G 1875 Ann. de Chim. et de Phys. 5 494
(in Chinese) [20] Orejon D, Sefiane K, Shanahan M E 2013 Appl. Phys. Lett. 102 201601
 Google Scholar
Google Scholar
[21] Daub C D, Bratko D, Leung K, Luzar A 2007 J. Phys. Chem. C 111 505
 Google Scholar
Google Scholar
[22] Song F H, Li B Q, Liu C 2013 Langmuir 29 4266
 Google Scholar
Google Scholar
[23] Lee M W, Latthe S S, Yarin A L, Yoon S S 2013 Langmuir 29 7758
 Google Scholar
Google Scholar
[24] Zhang B X, Wang S L, He X, Yang Y R, Wang X D, Lee D J 2021 J. Mol. Liq. 342 117468
 Google Scholar
Google Scholar
[25] Luedtke W D, Gao J P, Landman U 2011 J. Phys. Chem. C 115 20343
 Google Scholar
Google Scholar
[26] Zhu X Y, Yuan Q Z, Zhao Y P 2014 Nanoscale 6 5432
 Google Scholar
Google Scholar
[27] Sun W, Xu X B, Zhang H, Xu C X 2008 Cryobiology 56 93
 Google Scholar
Google Scholar
[28] Zangi R, Mark A E 2004 J. Chem. Phys. 120 7123
 Google Scholar
Google Scholar
[29] Jinesh K B, Frenken J W M 2008 Phys. Rev. Lett. 101 036101
 Google Scholar
Google Scholar
[30] Ahmad I, Ranjan A, Pathak M, Khan M K 2023 Int. J. Therm. Sci. 192 108440
 Google Scholar
Google Scholar
[31] Lu Y, Liu D 2023 Int J Heat Mass Tran. 208 124055
 Google Scholar
Google Scholar
[32] 胡剑, 张森, 娄钦 2023 物理学报 72 176401
 Google Scholar
Google Scholar
Hu J, Zhang S, Lou Q 2023 Acta Phys. Sin. 72 176401
 Google Scholar
Google Scholar
[33] Mugele F, Baret J C 2005 J. Phys.: Condens. Matter 17 R705
 Google Scholar
Google Scholar
[34] Plimpton S 1995 J. Comput. Phys. 117 1
 Google Scholar
Google Scholar
[35] Yenigun O, Barisik M 2019 Nanoscale Microscale Thermophys. Eng 4 304
 Google Scholar
Google Scholar
-
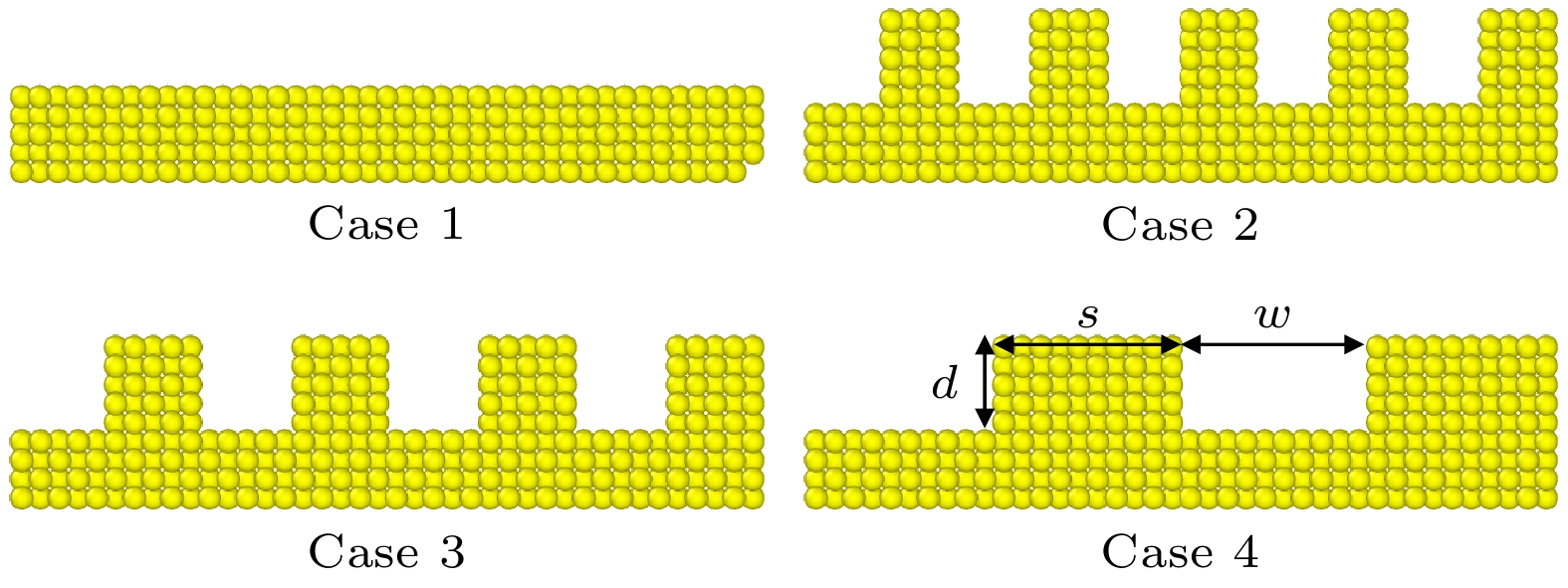
表 1 4种纳米结构的参数
Table 1. Parameters of four nanogroove configurations.
d/nm w/nm s/nm Case 1 — — — Case 2 1.02 0.816 0.816 Case 3 1.02 1.02 1.02 Case 4 1.02 2.04 2.04 表 2 L-J势函数的具体参数
Table 2. Parameters of L-J potential.
原子类型 σ/nm ε/eV q O-O 0.3166 0.0068 –0.8476e H-H 0 0 +0.4238e Au-O 0.2867 0.0114 — Au-H 0 0 — -
[1] Razeeb K M, Dalton E, Cross G L W, Robinson A J 2018 Int. Mater. 63 1
 Google Scholar
Google Scholar
[2] Pop E 2010 Nano Res. 3 147
 Google Scholar
Google Scholar
[3] Kapitza P L 1971 J. Phys. U.S.S.R. 4 181
 Google Scholar
Google Scholar
[4] Shenogina N, Godawat R, Keblinski P, Garde S 2009 Phys. Rev. Lett. 102 156101
 Google Scholar
Google Scholar
[5] Harikrishna H, Ducker W A, Huxtable S T 2013 Appl. Phys. Lett. 102 251606
 Google Scholar
Google Scholar
[6] Park S C, Cho H R, Kim D, Choi S H, Choi C, Yu D I 2024 Int. J. Heat Fluid Flow 107 109388
 Google Scholar
Google Scholar
[7] Song G, Min C 2013 Mol. Phys. 111 903
 Google Scholar
Google Scholar
[8] Rashidi M M, Ghahremanian S, Toghraie D, Roy P 2020 Int. Commun. Heat Mass 117 140741
 Google Scholar
Google Scholar
[9] 张程宾, 许兆林, 陈永平 2014 物理学报 63 214706
 Google Scholar
Google Scholar
Zhang C B, Xu Z L, Chen Y P 2014 Acta Phys. Sin. 63 214706
 Google Scholar
Google Scholar
[10] Chakraborty P, Ma T, Cao L, Wang Y 2019 Int. J. Heat Mass. Tran. 136 702
 Google Scholar
Google Scholar
[11] Yao S T, Wang J S, Jin S F, Tan F G, Chen S P 2024 Int. J. Therm. Sci. 203 109161
 Google Scholar
Google Scholar
[12] Qin S Y, Chen Z X, Wang Q, Li W G, Xing H W 2024 Int. Commun. Heat Mass 151 107257
 Google Scholar
Google Scholar
[13] Cassie A B D 1948 Disscussions of the Faraday Society 3 11
 Google Scholar
Google Scholar
[14] Wenzel R N 1936 Ind. Eng. Chem. 28 988
 Google Scholar
Google Scholar
[15] Bormashenko E 2015 Adv. Colloid Interface Sci. 222 92
 Google Scholar
Google Scholar
[16] Bormashenko E, Pogreb R, Stein T, Whyman G, Erlich M, Musin A, Machavariani V, Aurbach D 2008 Phys. Chem. Chem. Phys 10 4056
 Google Scholar
Google Scholar
[17] 李文, 马骁婧, 徐进良, 王艳, 雷骏鹏 2015 物理学报 70 126101
 Google Scholar
Google Scholar
Li W, Ma X J, Xu J L, Wang Y, Lei J P 2015 Acta Phys. Sin. 70 126101
 Google Scholar
Google Scholar
[18] Sur A, Lu Y, Pascente C, Ruchhoeft P, Liu D 2018 Int. J. Heat Mass Tran. 120 202
 Google Scholar
Google Scholar
[19] Lippmann G 1875 Ann. de Chim. et de Phys. 5 494
(in Chinese) [20] Orejon D, Sefiane K, Shanahan M E 2013 Appl. Phys. Lett. 102 201601
 Google Scholar
Google Scholar
[21] Daub C D, Bratko D, Leung K, Luzar A 2007 J. Phys. Chem. C 111 505
 Google Scholar
Google Scholar
[22] Song F H, Li B Q, Liu C 2013 Langmuir 29 4266
 Google Scholar
Google Scholar
[23] Lee M W, Latthe S S, Yarin A L, Yoon S S 2013 Langmuir 29 7758
 Google Scholar
Google Scholar
[24] Zhang B X, Wang S L, He X, Yang Y R, Wang X D, Lee D J 2021 J. Mol. Liq. 342 117468
 Google Scholar
Google Scholar
[25] Luedtke W D, Gao J P, Landman U 2011 J. Phys. Chem. C 115 20343
 Google Scholar
Google Scholar
[26] Zhu X Y, Yuan Q Z, Zhao Y P 2014 Nanoscale 6 5432
 Google Scholar
Google Scholar
[27] Sun W, Xu X B, Zhang H, Xu C X 2008 Cryobiology 56 93
 Google Scholar
Google Scholar
[28] Zangi R, Mark A E 2004 J. Chem. Phys. 120 7123
 Google Scholar
Google Scholar
[29] Jinesh K B, Frenken J W M 2008 Phys. Rev. Lett. 101 036101
 Google Scholar
Google Scholar
[30] Ahmad I, Ranjan A, Pathak M, Khan M K 2023 Int. J. Therm. Sci. 192 108440
 Google Scholar
Google Scholar
[31] Lu Y, Liu D 2023 Int J Heat Mass Tran. 208 124055
 Google Scholar
Google Scholar
[32] 胡剑, 张森, 娄钦 2023 物理学报 72 176401
 Google Scholar
Google Scholar
Hu J, Zhang S, Lou Q 2023 Acta Phys. Sin. 72 176401
 Google Scholar
Google Scholar
[33] Mugele F, Baret J C 2005 J. Phys.: Condens. Matter 17 R705
 Google Scholar
Google Scholar
[34] Plimpton S 1995 J. Comput. Phys. 117 1
 Google Scholar
Google Scholar
[35] Yenigun O, Barisik M 2019 Nanoscale Microscale Thermophys. Eng 4 304
 Google Scholar
Google Scholar
计量
- 文章访问数: 4512
- PDF下载量: 136
- 被引次数: 0













 下载:
下载: