-
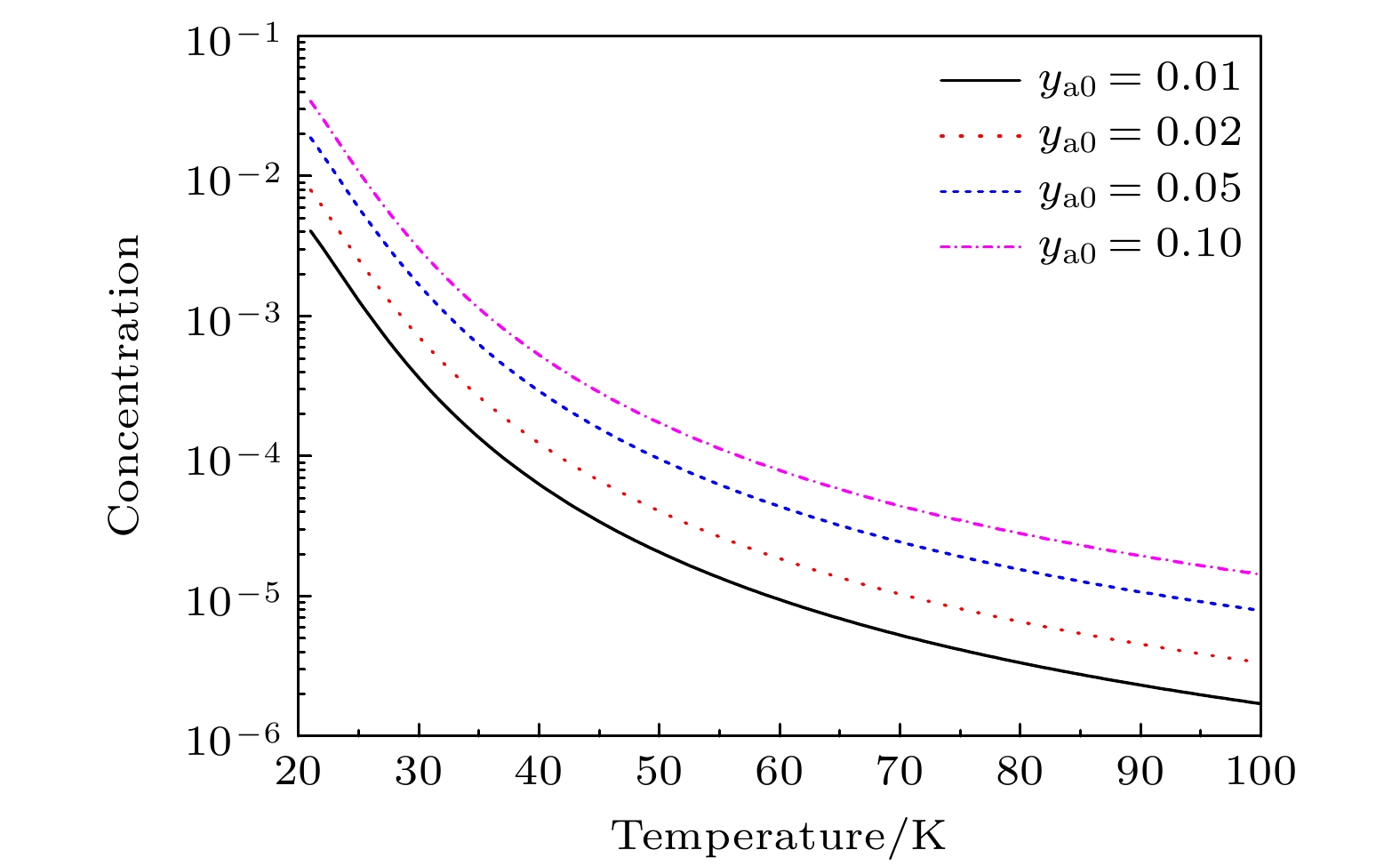
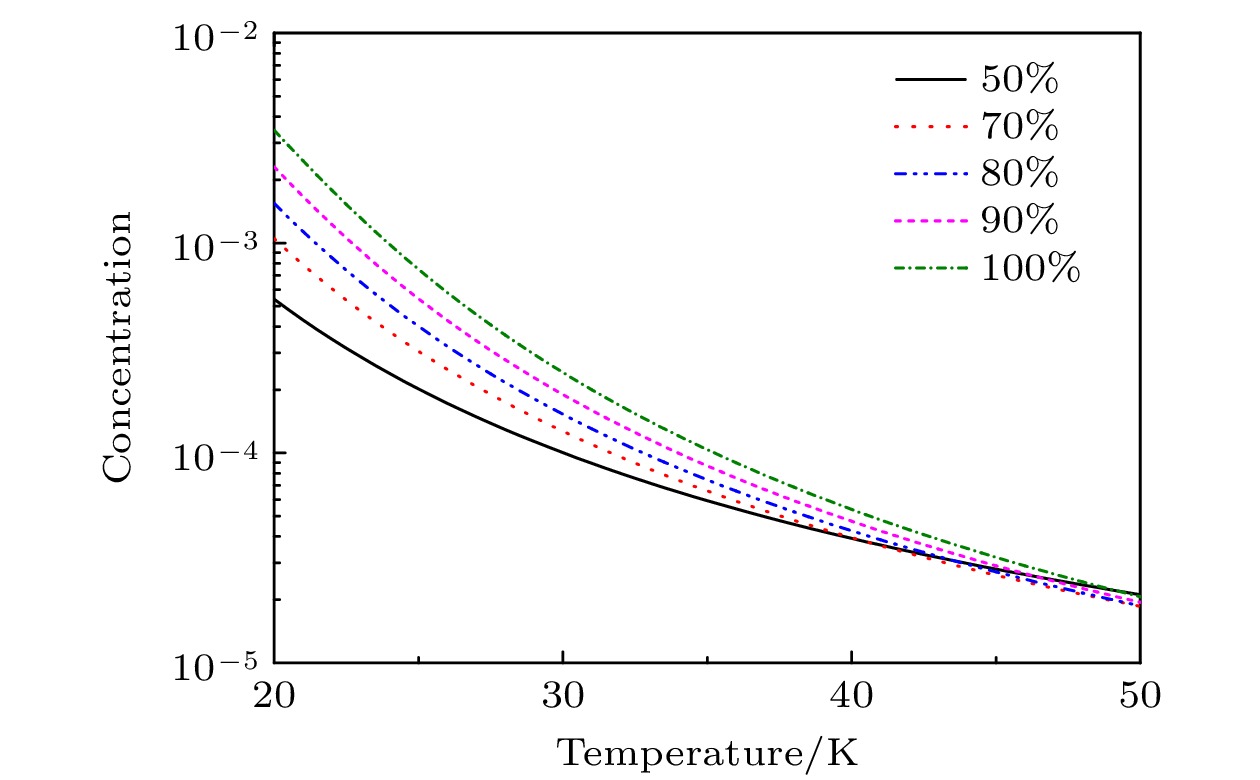
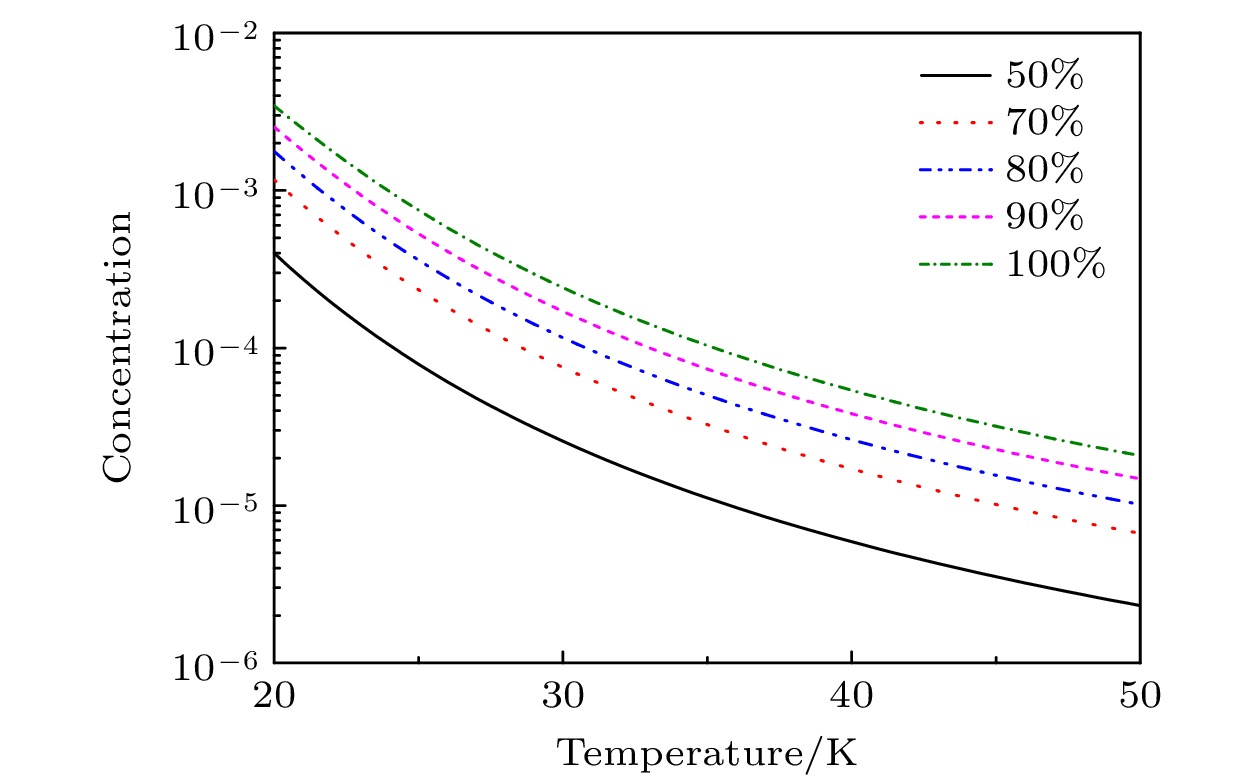
In the low temperature environment generated by supersonic flow in the process of laser assisted retardation of condensation of isotope separation for BCl3, the molecular isotopes BCl3 and carrier gas (rare gas (RG): He, Ne, Ar, Kr, Xe) can form BCl3:RG dimer via contact collision process. The mechanism and relationship between dimer concentration and absolute temperature of dimer involving BCl3 molecules are of great significance for regulating and selecting the isotope separation parameters. In this work, based on the analytic description of the anharmonic interaction potential function of BCl3:RG, and considering the two-body and three-body collision induced association and dissociation of dimers, the concentration of BCl3:RG dimers is obtained at the absolute temperature in a range of 20–40 K. The obtained results are as follows. The two-body collision is dominant in the formation of dimer in the low temperature range. When the initial molar fraction of BCl3 is in a range of 0.01–0.10, the BCl3:RG dimer concentration changes approximately linearly with the initial molar fraction of BCl3, indicating that the initial molar fraction not only determines the theoretical upper limit of the dimer concentration, but also dominates the dimer concentration in a low temperature range. When the temperature of the supersonic flow chamber is about 20 K, the concentration of BCl3:Kr dimers is largest, and the concentrations of other dimers are also presented. Furthermore, we explain the mechanism of laser assisted retardation of condensation in separation of isotopes by using a simple model on a molecular scale by adjusting the parameters of dissociation energy and stretching vibration frequency of the dimer.
[1] Apatin V M, Lokhman V N, Makarov G N, Ogurok N D D, Ryabov E A 2018 Quantum Electron. 48 157
 Google Scholar
Google Scholar
[2] Makarov G N, Ogurok N D D, Petin A N 2018 Quantum Electron. 48 667
 Google Scholar
Google Scholar
[3] Lokhman V N, Makarov G N, Malinovskii A L, Petin A N, Poydashev D G, Ryabov E A 2018 Laser Phys. 28 105703
 Google Scholar
Google Scholar
[4] Lyakhov K A, Lee H J, Pechen A N 2017 Sep. Purif. Technol. 176 402
 Google Scholar
Google Scholar
[5] Silex Systems-SILEX Laser Enrichment Technology https://wp.silex.com.au/[2022-07-25]
[6] Snyder R 2016 Sci. Glob. Secur. 24 68
[7] Baldwin A 2016 M. S. Thesis (Cambridge: Massachusetts Institute of Technology)
[8] Eerkens J W 2005 Laser Part. Beams 23 225
 Google Scholar
Google Scholar
[9] Lyakhov K A 2022 J. Phys. Conf. Ser. 2147 012009
 Google Scholar
Google Scholar
[10] Lyakhov K A, Lee H J 2013 Ann. Nucl. Energy 54 274
 Google Scholar
Google Scholar
[11] Lowry J T, Snider R F 1974 J. Chem. Phys. 61 2320
 Google Scholar
Google Scholar
[12] Eerkens J W 2001 Chem. Phys. 269 189
 Google Scholar
Google Scholar
[13] Eerkens J W 2006 Chem. Phys. Lett. 430 271
 Google Scholar
Google Scholar
[14] Eerkens J W 2005 Nucl. Sci. Eng. 150 1
 Google Scholar
Google Scholar
[15] Lyakhov K A, Pechen A N 2021 Proc. Steklov Inst. Math. 313 131
 Google Scholar
Google Scholar
[16] Borner A, Li Z, Levin D A 2013 J. Chem. Phys. 138 064302
 Google Scholar
Google Scholar
[17] Lyakhov K A, Pechen A N 2020 Lobachevskii J. Math. 41 2345
 Google Scholar
Google Scholar
[18] Lyakhov K, Lee H J 2015 J. Nanosci. Nanotechnol. 15 8502
 Google Scholar
Google Scholar
[19] Lyakhov K, Lee H J, Pechen A 2016 IEEE J. Quantum Electron. 52 1400208
 Google Scholar
Google Scholar
[20] Guo J, Li Y J, Ma J P, Tang X, Liu X S 2021 Chem. Phys. Lett. 773 138572
 Google Scholar
Google Scholar
[21] Li Y J, Guo J, Ma J P, Tang X, Liu X S 2021 Chem. Phys. Lett. 781 138948
 Google Scholar
Google Scholar
-
表 1 不同温度(20—40 K)下二聚体浓度(初始摩尔分数比0.02∶0.98)
Table 1. Concentration of BCl3:RG at different absolute temperature (20—40 K). Initial mole fractions ration is 0.02∶0.98.
温度T/K BCl3:He/10–4 BCl3:Ne/10–4 BCl3:Ar/10–4 BCl3:Kr/10–4 BCl3:Xe/10–4 20.0 21.7 20.1 34.5 n/a 66.2 20.5 20.0 18.0 29.1 91.0 55.3 21.0 18.5 16.1 24.6 79.9 46.1 21.5 17.2 14.5 20.9 69.7 38.3 22.0 16.0 13.1 17.8 60.5 31.8 22.5 14.9 11.9 15.3 52.4 26.4 23.0 13.9 10.8 13.1 45.3 22.0 23.5 13.0 9.82125 11.3 39.2 18.4 24.0 12.2 8.97569 9.83335 33.9 15.5 24.5 11.4 8.22618 8.56576 29.3 13.0 25.0 10.7 7.55977 7.49333 25.4 11.0 25.5 10.1 6.96548 6.58238 22.1 9.37370 26.0 9.54682 6.43398 5.80547 19.3 8.00714 26.5 9.02508 5.95728 5.14022 16.8 6.87095 27.0 8.54503 5.52857 4.56831 14.7 5.92244 27.5 8.10250 5.14199 4.07473 13.0 5.12731 28.0 7.69381 4.79251 3.64712 11.4 4.45793 28.5 7.31570 4.47579 3.27529 10.1 3.89202 29.0 6.96527 4.18808 2.95080 8.97688 3.41159 29.5 6.63995 3.92610 2.66662 7.99078 3.00205 30.0 6.33746 3.68704 2.41692 7.13453 2.65152 30.5 6.05576 3.46841 2.19678 6.38891 2.35034 31.0 5.79303 3.26806 2.00210 5.73774 2.09055 31.5 5.54762 3.08409 1.82941 5.16742 1.86565 32.0 5.31808 2.91484 1.67578 4.66648 1.67025 32.5 5.10308 2.75884 1.53871 4.22524 1.49989 33.0 4.90144 2.61480 1.41609 3.83551 1.35086 33.5 4.71209 2.48156 1.30610 3.49035 1.22007 34.0 4.53406 2.35812 1.20720 3.18384 1.10494 34.5 4.36648 2.24357 1.11805 2.91096 1.00327 35.0 4.20854 2.13710 1.03750 2.66739 0.913239 35.5 4.05952 2.03799 0.964548 2.44947 0.833287 36.0 3.91878 1.94560 0.898342 2.25403 0.762093 36.5 3.78571 1.85936 0.838129 2.07834 0.698532 37.0 3.65976 1.77874 0.783253 1.92005 0.641643 37.5 3.54044 1.70329 0.733144 1.77713 0.590602 38.0 3.42728 1.63257 0.687301 1.64781 0.544699 38.5 3.31988 1.56621 0.645283 1.53056 0.503325 39.0 3.21784 1.50388 0.606702 1.42405 0.465950 39.5 3.12081 1.44524 0.571217 1.32709 0.432116 40.0 3.02848 1.39004 0.538526 1.23868 0.401426 注: n/a = not available, 表示该点的数值计算未收敛. -
[1] Apatin V M, Lokhman V N, Makarov G N, Ogurok N D D, Ryabov E A 2018 Quantum Electron. 48 157
 Google Scholar
Google Scholar
[2] Makarov G N, Ogurok N D D, Petin A N 2018 Quantum Electron. 48 667
 Google Scholar
Google Scholar
[3] Lokhman V N, Makarov G N, Malinovskii A L, Petin A N, Poydashev D G, Ryabov E A 2018 Laser Phys. 28 105703
 Google Scholar
Google Scholar
[4] Lyakhov K A, Lee H J, Pechen A N 2017 Sep. Purif. Technol. 176 402
 Google Scholar
Google Scholar
[5] Silex Systems-SILEX Laser Enrichment Technology https://wp.silex.com.au/[2022-07-25]
[6] Snyder R 2016 Sci. Glob. Secur. 24 68
[7] Baldwin A 2016 M. S. Thesis (Cambridge: Massachusetts Institute of Technology)
[8] Eerkens J W 2005 Laser Part. Beams 23 225
 Google Scholar
Google Scholar
[9] Lyakhov K A 2022 J. Phys. Conf. Ser. 2147 012009
 Google Scholar
Google Scholar
[10] Lyakhov K A, Lee H J 2013 Ann. Nucl. Energy 54 274
 Google Scholar
Google Scholar
[11] Lowry J T, Snider R F 1974 J. Chem. Phys. 61 2320
 Google Scholar
Google Scholar
[12] Eerkens J W 2001 Chem. Phys. 269 189
 Google Scholar
Google Scholar
[13] Eerkens J W 2006 Chem. Phys. Lett. 430 271
 Google Scholar
Google Scholar
[14] Eerkens J W 2005 Nucl. Sci. Eng. 150 1
 Google Scholar
Google Scholar
[15] Lyakhov K A, Pechen A N 2021 Proc. Steklov Inst. Math. 313 131
 Google Scholar
Google Scholar
[16] Borner A, Li Z, Levin D A 2013 J. Chem. Phys. 138 064302
 Google Scholar
Google Scholar
[17] Lyakhov K A, Pechen A N 2020 Lobachevskii J. Math. 41 2345
 Google Scholar
Google Scholar
[18] Lyakhov K, Lee H J 2015 J. Nanosci. Nanotechnol. 15 8502
 Google Scholar
Google Scholar
[19] Lyakhov K, Lee H J, Pechen A 2016 IEEE J. Quantum Electron. 52 1400208
 Google Scholar
Google Scholar
[20] Guo J, Li Y J, Ma J P, Tang X, Liu X S 2021 Chem. Phys. Lett. 773 138572
 Google Scholar
Google Scholar
[21] Li Y J, Guo J, Ma J P, Tang X, Liu X S 2021 Chem. Phys. Lett. 781 138948
 Google Scholar
Google Scholar
Catalog
Metrics
- Abstract views: 6387
- PDF Downloads: 66
- Cited By: 0















 DownLoad:
DownLoad: