-
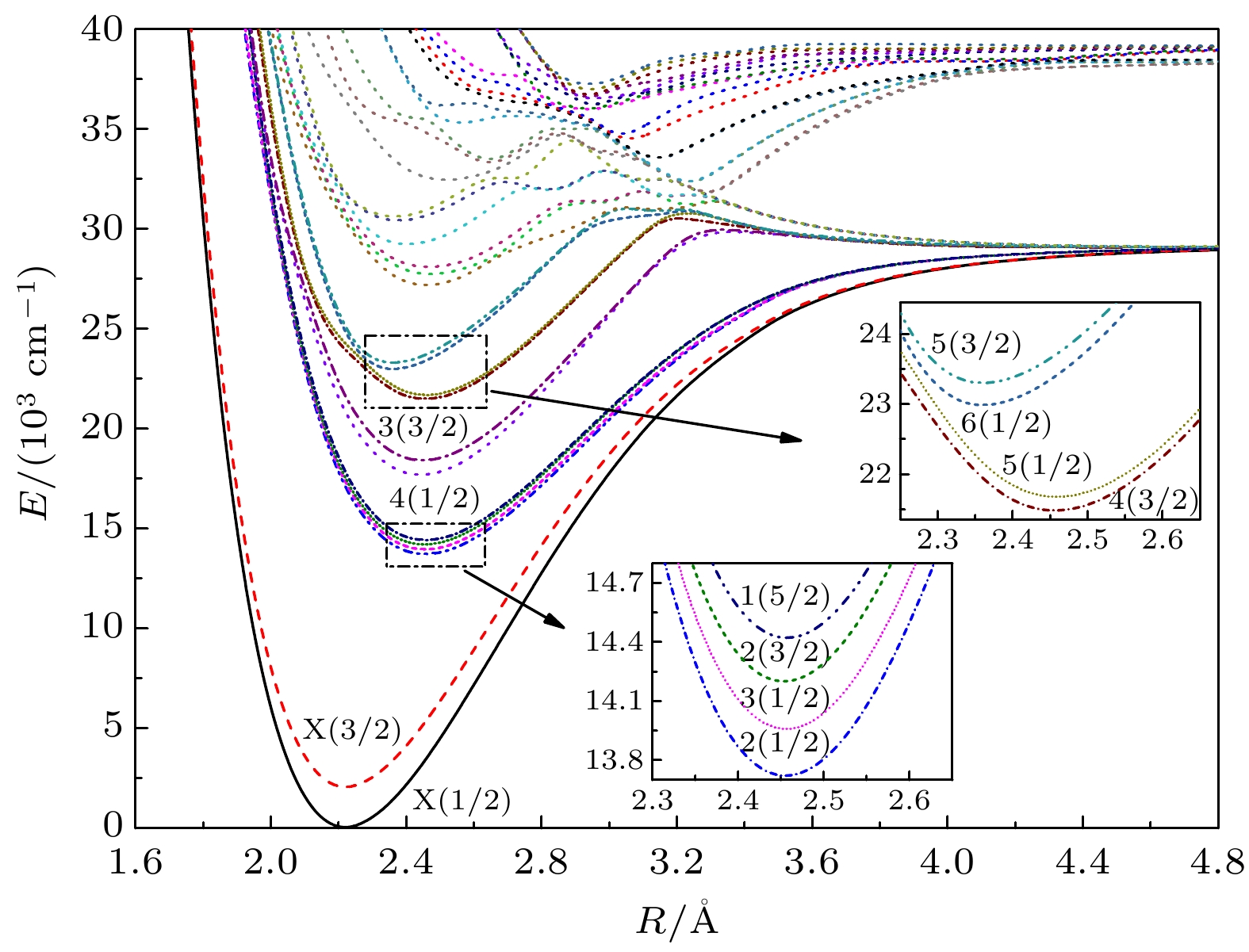
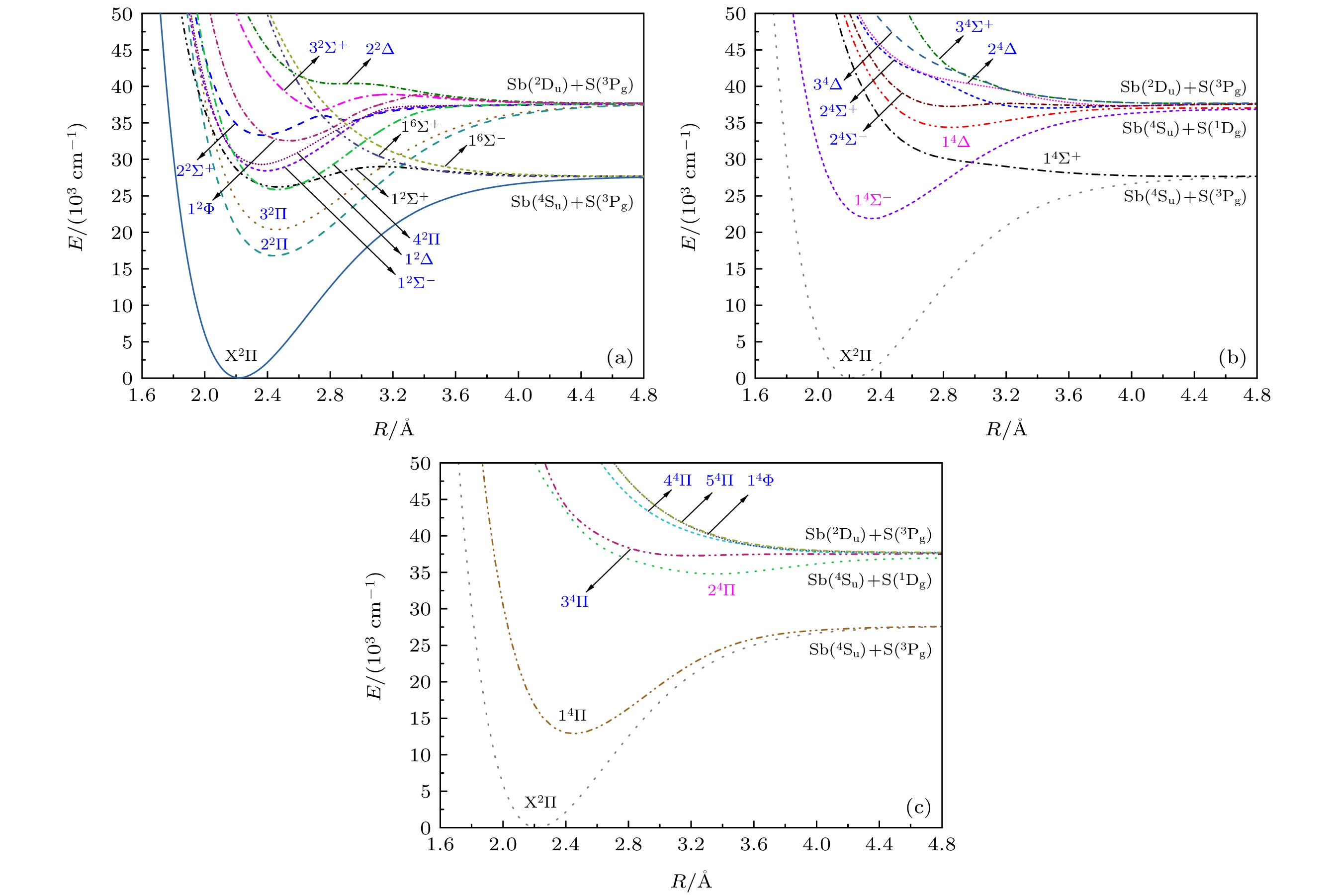
运用多参考组态相互作用(MRCI+Q)方法, 对硫化锑(SbS)能量最低的3个Ʌ-S离解极限的所有电子态以及考虑自旋-轨道耦合效应后分裂所得的Ω态进行了计算. 得到27个Ʌ-S电子态及能量最低的12个Ω态的电子结构、光谱常数和振动能级等信息. Sb原子和S原子能级的计算值与实验值相符很好. 分析表明自旋-轨道耦合效应对光谱常数与振动能级的影响总体上并不显著. 对X(3/2)→X(1/2), 2(1/2)→X(1/2), 4(1/2)→X(1/2), 5(1/2)→X(1/2)及6(1/2)→X(1/2)跃迁的振动光谱进行了模拟与分析, 其中X(3/2)→X(1/2)谱带位于中红外波段, 其他谱带均位于可见光波段. 此外, 对氮族元素硫化物的电子态进行了验证计算, 计算结果与已有实验结果吻合较好, 体现了同族元素代换后相关物性的渐变规律性.In this paper, highly correlated ab initio calculations are performed for accurately determining the electronic structures and spectroscopic features of the Λ-S and Ω low-lying electronic states of SbS . The potential energy curves for 27 Λ-S states of the first three dissociation asymptotes are constructed. Spectroscopic constants and vibrational states for all bound states are well determined. The calculated atomic states for both atoms are consistent with experimental data quite well. Several low-lying Ω electronic states are also investigated, and their respective spectroscopic constants and vibrational states are obtained and compared with those of corresponding Λ-S states, which indicates that the spin-orbit coupling effect gives rise to a minor influence on the electronic states of SbS. To verify our computational accuracy, the additional calculations for the low-lying electronic states of PS, AsS and BiS are also carried out. Our derived results are in reasonable agreement with available experimental data. In addition, vibrational spectra from the excited Ω states to the ground state of SbS are simulated, including bands of X(3/2)→X(1/2), 2(1/2)→X(1/2), 4(1/2)→X(1/2), 5(1/2)→X(1/2) and 6(1/2)→X(1/2). The X(3/2)→X(1/2) band is found in the mid-infrared region, while the others are located in the visible region. The predictive results provided in this paper are expected to serve as a guideline for further researches, such as assisting laboratorial detections and analyzing observed spectrum of SbS.
-
Keywords:
- SbS /
- electronic structure /
- spin-orbit coupling effect /
- spectroscopic constants
[1] Lian W T, Jiang C H, Yin Y W, Tang R F, Li G, Zhang L J, Che B, Chen T 2021 Nat. Commun. 12 3260
 Google Scholar
Google Scholar
[2] Zhao R M, Yang X L, Shi H L, Du M H 2021 Phys. Rev. Mater. 5 054605
 Google Scholar
Google Scholar
[3] Yang Y, Shi C W, Lv K, Wang Q, Sun X, Chen W C 2021 New J. Chem. 45 10357
 Google Scholar
Google Scholar
[4] Y Grad L, von Rohr F O, Hengsberger M, Osterwalder J 2021 Phys. Rev. Mater. 5 075401
 Google Scholar
Google Scholar
[5] Hu X K, Ma Y X, Pang Z X, Li P 2019 Chem. Phys. 523 110
 Google Scholar
Google Scholar
[6] Zhang J W, Lian W T, Yin Y W, et al. 2020 Solar RRL 4 2000048
 Google Scholar
Google Scholar
[7] Shimauchi M, Nishiyama Y 1968 Sci. Light 17 76
[8] Fowler A, Bakker C J 1932 Proc. Roy. Soc. (London) A 136 28
[9] Zeeman P B 1951 Can. J Phys. 29 174
 Google Scholar
Google Scholar
[10] Barrow R F, Drummond G, Zeeman P B 1954 Proc. Roy. Soc. (London) A 67 365
[11] Amano T, Saito S, Hirota E, Morino Y 1969 J. Mol. Spectrosc. 32 97
 Google Scholar
Google Scholar
[12] Jenouvrier A, Pascat B 1973 Can. J. Phys. 51 2143
 Google Scholar
Google Scholar
[13] Jenouvrier A, Pascat B 1980 Can. J. Phys. 58 1275
 Google Scholar
Google Scholar
[14] Wang T T, Li C Y, Zheng X F, Chen Y 2007 Chin. Sci. Bull. 52 596
 Google Scholar
Google Scholar
[15] Dressler K, Miescher E 1955 Proc. Roy. Soc. (London) A 68 542
 Google Scholar
Google Scholar
[16] Dressler K 1955 Ph. D. Dissertation (Basel: Universität Basel)
[17] Narasimham N A, Subramanian T K B 1969 J. Mol. Spectrosc. 29 294
 Google Scholar
Google Scholar
[18] Narasimham N A, Subramanian T K B 1971 J. Mol. Spectrosc. 37 371
 Google Scholar
Google Scholar
[19] Jenouvrier A, Pascat B 1978 Can. J. Phys. 56 1088
 Google Scholar
Google Scholar
[20] Balasubramanian T K, Dixit M N, Narasimham N A 1979 Pramana 12 707
 Google Scholar
Google Scholar
[21] Kawaguchi K, Hirota E, Ohishi M, Suzuki H, Takano S, Yamamoto S, Saito S 1988 J. Mol. Spectrosc. 130 81
 Google Scholar
Google Scholar
[22] Ohishi M, Yamamoto S, Saito S, et al. 1988 Astrophys. J 329 511
 Google Scholar
Google Scholar
[23] Klein H, Klisch E, Winnewisser G 1999 Z. Naturforschung A 54 137
 Google Scholar
Google Scholar
[24] Shimauchi M, 1969 Sci. Light 18 90
[25] Shimauchi M, 1971 Can. J. Phys. 49 1249
 Google Scholar
Google Scholar
[26] Shimauchi M, Sakaba Y, Kikuchi S 1972 Sci. Light 21 1
[27] Shimauchi M, Iwata H, Matsuno T, Sakaba Y, Lee S K, Karasawa S 1972 Sci. Light 21 145
[28] Shimauchi M, Karasawa S 1973 Sci. Light 22 127
[29] Barrow R F, Stobart O V, Vaughan H 1967 Proc. Phys. Soc. Lond. 90 555
 Google Scholar
Google Scholar
[30] Patiño P, Eland J H D, Barrow R F 1984 J. Phys. B:At. Mol. Phys. 17 1009
 Google Scholar
Google Scholar
[31] Izumi K, Cohen E A, Setzer K D, Fink E H, Kawaguchi K 2008 J. Mol. Spectrosc. 252 198
 Google Scholar
Google Scholar
[32] Setzer K D, Meinecke F, Fink E H 2009 J. Mol. Spectrosc. 258 56
 Google Scholar
Google Scholar
[33] O’Hare P A G 1970 J. Chem. Phys. 52 2992
 Google Scholar
Google Scholar
[34] Bialski M, Grein F 1976 J. Mol. Spectrosc. 61 321
 Google Scholar
Google Scholar
[35] Karna S P, Grein F 1986 J. Mol. Spectrosc. 120 284
 Google Scholar
Google Scholar
[36] Karna S P, Bruna P J, Grein F 1988 J. Phys. B:At. Mol. Opt. Phys. 21 1303
 Google Scholar
Google Scholar
[37] Karna S P, Grein F 1992 Mol. Phys. 77 135
 Google Scholar
Google Scholar
[38] Chong D P 1994 Chem. Phys. Lett. 220 102
 Google Scholar
Google Scholar
[39] Moussaoui Y, Ouamerali O, De Maré G R 1998 J Mol. Struct. Theochem. 425 237
 Google Scholar
Google Scholar
[40] Kalcher J 2002 Phys. Chem. Chem. Phys. 4 3311
 Google Scholar
Google Scholar
[41] Peebles L R, Marshall P 2002 Chem. Phys. Lett. 366 520
 Google Scholar
Google Scholar
[42] Czernek J, Živný O 2004 Chem. Phys. 303 137
 Google Scholar
Google Scholar
[43] Yaghlane S B, Francisco J S, Hochlaf M 2012 J. Chem. Phys. 136 244309
 Google Scholar
Google Scholar
[44] Yang, J, Kang Y, Wang X, Bai X 2013 J. Mol. Model 19 5199
 Google Scholar
Google Scholar
[45] Lingott R M, Liebermann H P, Alekseyev A B, Buenker R J 1999 J. Chem. Phys. 110 11294
 Google Scholar
Google Scholar
[46] Shi D H, Xing W, Sun J F, Zhu Z L 2012 Eur. Phys. J. D 66 173
 Google Scholar
Google Scholar
[47] Gao Y F, Gao T, Gong M 2013 J Quant. Spectrosc. Radiat. Transf. 129 193
 Google Scholar
Google Scholar
[48] 刘慧, 邢伟, 施德恒, 孙金锋, 朱遵略 2013 物理学报 62 203104
 Google Scholar
Google Scholar
Liu H, Xing W, Shi D H, Sun J F, Zhu Z L 2013 Acta Phys. Sin. 62 203104
 Google Scholar
Google Scholar
[49] Shi D H, Song Z Y, Niu X H, Sun J F, Zhu Z L 2016 Spectrochim. Acta A Mol. Biomol. Spectrosc. 153 30
 Google Scholar
Google Scholar
[50] Prajapat L, Jagoda P, Lodi L, Gorman M N, Yurchenko S N, Tennyson J 2017 MNRAS 472 3648
 Google Scholar
Google Scholar
[51] Zhou D, Shi D H, Sun J F 2019 J. Quant. Spectrosc. Radiat. Transf. 230 120
 Google Scholar
Google Scholar
[52] de Almeida A A, Andreazza C M, Borin A C 2020 Theor. Chem. Acc. 139 33
 Google Scholar
Google Scholar
[53] Reddy R R, Reddy A S R, Rao T V R 1985 Pramana 25 187
 Google Scholar
Google Scholar
[54] Knowles P J, Werner H J 1985 J. Chem. Phys. 82 5053
 Google Scholar
Google Scholar
[55] Werner H J, Knowles P J 1988 J. Chem. Phys. 89 5803
 Google Scholar
Google Scholar
[56] Peterson K A, Dunning Jr. T H 2002 J. Chem. Phys. 117 10548
 Google Scholar
Google Scholar
[57] Peterson K A, Yousaf K E 2010 J. Chem. Phys. 133 174116
 Google Scholar
Google Scholar
[58] Murrell J N, Sorbie K S 1974 J. Chem. Soc. Faraday Trans. 2 1552
[59] 李松, 韩立波, 陈善俊, 段传喜 2013 物理学报 62 113102
 Google Scholar
Google Scholar
Li S, Han L B, Chen S J, Duan C X 2013 Acta Phys. Sin. 62 113102
 Google Scholar
Google Scholar
[60] Li S, Chen S J, Zhu D S, Fan Q C 2013 Comput. Theor. Chem. 1017 136
 Google Scholar
Google Scholar
[61] Lu N, Wu W Q, Zhang C Z, Wan M J, Jin Y Y, Zhang W B, Chen S J, Li S 2020 Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 237 118301
 Google Scholar
Google Scholar
[62] Li S, Chen S J, Chen Y, Chen P 2016 Chin. Phys. B. 25 033101
 Google Scholar
Google Scholar
[63] Chen P, Wang N, Li S, Chen S J 2017 J. Quant. Spectrosc. Radiat. Transf. 201 104
 Google Scholar
Google Scholar
[64] 万明杰, 李松, 金成国, 罗华锋 2019 物理学报 68 063103
 Google Scholar
Google Scholar
Wan M J, Li S, Jin C G, Luo H F 2019 Acta Phys. Sin. 68 063103
 Google Scholar
Google Scholar
[65] Berning A, Schweizer M, Werner H J, Knowles P J, Palmieri P 2000 Mol. Phys. 98 1823
 Google Scholar
Google Scholar
[66] Werner H J, Knowles P J, Knizia G, et al. MOLPRO, version 2015.1, A Package of ab initio Programs, 2015
[67] Le Roy R J 2017 J. Quant. Spectrosc. Radiat. Transf. 186 167
 Google Scholar
Google Scholar
[68] Sansonetti J E, Martin W C 2005 J. Phys. Chem. Ref. Data 34 1559
 Google Scholar
Google Scholar
-
表 1 SbS的Λ-S态离解极限
Table 1. Dissociation relationships of the Λ-S states of SbS.
原子态 Λ-S态 ΔE/cm–1 实验值[68] 计算值 $ {\text{Sb}}({}^4{{\text{S}}_{\text{u}}}) + {\text{S}}{(^3}{{\text{P}}_{\text{g}}}) $ ${{\text{1}}^2}{\Sigma ^ + }$, ${{\rm X}^2}\Pi $ , ${{\text{1}}^4}{\Sigma ^ + }$, ${{\text{1}}^4}\prod $, ${{\text{1}}^6}{\Sigma ^ + }$, $ {1^6}\Pi $ 0 0 $ {\text{Sb}}({}^4{{\text{S}}_{\text{u}}}) + {\text{S}}{(^1}{{\text{D}}_{\text{g}}}) $ ${1^4}{\Sigma ^ - }$, ${1^4}\Delta $, $2{}^4\Pi $ 9238.609 9346 $ {\text{Sb}}({}^2{{\text{D}}_{\text{u}}}) + {\text{S}}{(^3}{{\text{P}}_{\text{g}}}) $ ${2^2}{\Sigma ^ + }$, ${3^2}{\Sigma ^ + }$, ${1^2}{\Sigma ^ - }$, ${1^2}\Delta $, ${2^2}\Delta $, $2{}^2\Pi $, $3{}^2\Pi $, $4{}^2\Pi $, ${1^2}\Phi $,
${2^4}{\Sigma ^ + }$, ${3^4}{\Sigma ^ + }$, ${2^4}{\Sigma ^ - }$, ${2^4}\Delta $, ${3^4}\Delta $, $3{}^4\Pi $, $4{}^4\Pi $, $5{}^4\Pi $, ${1^4}\Phi $9854.018 10022 表 2 SbS的Λ-S态光谱常数
Table 2. Spectroscopic constants of the Λ-S states of SbS.
Λ-S态 Re/Å De/eV Be/cm–1 ωe/cm–1 ωeχe /cm–1 Te/cm–1 RMS/cm–1 电子组态(组态系数) ${{\text{X}}^{\text{2}}}\Pi $ 2.2199 3.44 0.1348 479.8 1.51 0 0.57 15σαβ7πxαβyαβ8πxα (72.77) ${1^4}\Pi $ 2.4481 1.84 0.1108 343.6 1.17 12884 0.65 15σαβ7πxαβyα8πxαyα (84.83) ${{\text{2}}^{\text{2}}}\Pi $ 2.4388 2.60 0.1117 356.3 0.89 16721 0.64 15σαβ7πxαβyβ8πxαyα (52.26) ${{\text{3}}^{\text{2}}}\Pi $ 2.4513 2.16 0.1106 341.8 0.97 20306 0.88 15σαβ7πxαβyα8πxαyβ (31.90) ${1^4}{\Sigma ^ - }$ 2.3354 1.88 0.1218 361.1 2.54 21870 1.53 15σα7πxαβyαβ8πxαyα (83.08) ${4^{\text{2}}}\Pi $ 2.4554 1.49 0.1109 341.1 1.97 25796 2.85 15σαβ7πxαyαβ8πyαβ (59.44) ${{\text{1}}^{\text{2}}}{\Sigma ^ + }$第一势阱 2.4699 0.45 0.1089 254.4 0.88 26185 3.64 15σαβ16σα7πxαβyαβ (52.15) ${1^{\text{2}}}{\Sigma ^ - }$ 2.3735 1.17 0.1179 340.1 6.30 28350 7.03 15σα7πxαβyαβ8πxβyα (61.23) ${1^{\text{2}}}\Delta $ 2.3545 1.04 0.1198 343.5 2.51 29271 1.80 15σα7πxαβyαβ8πxαyβ (60.01) ${1^2}\Phi $ 2.5497 1.45 0.1022 265.1 1.96 32508 6.63 15σαβ7πxαyα8πxαβyβ(50.92) ${2^{\text{2}}}{\Sigma ^ + }$ 2.3678 0.63 0.1185 337.0 1.73 33250 2.19 15σα7πxαβyαβ8πyαβ (38.05) 15σα7πxαβyαβ8πxαβ (38.05) ${1^4}\Delta $ 2.8462 0.34 0.0820 188.3 2.40 34389 0.84 15σαβ16σα7πxαβyα8πyα (35.39) 15σαβ16σα7πxαyαβ8πxα (35.39) ${2^4}\Pi $ 3.3555 0.28 0.0590 134.1 1.52 34791 0.38 15σα16σα7πxαβyβ8πxαyα (27.31) ${3^{\text{2}}}{\Sigma ^ + }$ 3.0478 0.32 0.0715 189.7 4.99 36554 3.79 15σα7πxαβyαβ8πyαβ (16.85) 15σα7πxαβyαβ8πxαβ (16.85) ${2^4}{\Sigma ^ + }$ 3.4574 0.09 0.0556 87.1 1.80 37047 0.84 15σα7πxαyαβ8πxαyαβ (14.08) 15σα7πxαβyα8πxαβyα (14.08) ${2^4}{\Sigma ^ - }$第一势阱 2.8356 0.06 0.0826 184.4 6.87 37260 1.01 15σαβ16σα7πxαyαβ8πyα (35.84) 15σαβ16σα7πxαβyα 8πxα (35.84) ${2^4}\Delta $ 3.9570 0.05 0.0424 95.3 7.03 37339 0.96 15σα16σαβ7πxαyαβ8πxα (17.70) 15σα16σαβ7πxαβyα8πyα (17.70) 表 3 XS (X = N, P, As, Sb, Bi)自由基电子基态
${{\text{X}}^{\text{2}}}\Pi $ 的光谱常数Table 3. Spectroscopic constants of the ground state
${{\text{X}}^{\text{2}}}\Pi $ of XS (X = N, P, As, Sb, Bi) radicals.Re/Å De/eV ωe/cm–1 Be/cm–1 理论值[35] a 1.515 — 1220.5 0.7542 理论值[40] b 1.5058 — 1202.4 0.742 NS 理论值[46] c 1.4962 4.8504 1216.17 0.77323 理论值[47] d 1.498 — 1220.9 0.7715 实验值[9] 1.495(7) — — 0.7736(4) 实验值[11] 1.4938(2) — — — 理论值[36] e 1.944 — 735.6 0.2836 理论值[40] b 1.9148 — 728.0 0.292 理论值[43] f 1.879 — 732.0 0.2936 PS 理论值[48] g 1.8972 4.5272 741.0 0.2979 理论值[52] h 1.918 — 708 — 实验值[15] 1.92 — 739.5 0.29 实验值[18] 1.900(7) — — — 实验值[21] 1.8977405(45) — 739.13
(42)0.2975216
(14)本文工作 1.9014 4.41 739.5 0.2960 理论值[53] i — 4.15(13) — — 理论值[40] b 2.0395 — 559.2 0.181 AsS 理论值[44] j 2.045 3.94 — — 理论值[49] k 2.0180 4.0554 565.19 0.18472 实验值[28] 2.0174 — 567.94 0.18476 本文工作 2.0208 3.83 564.4 0.1839 SbS 本文工作 2.2199 3.44 479.8 0.1348 BiS 本文工作 2.3118 3.12 424.9 0.1135 注: a MRSDCI/modified basis sets; b CAS-ACPF/cc-pVQZ; c MRCI+Q/AV5Z+CV+DK; d MRCI+Q/aug-cc-pV5Z; e MRSDCI/modified basis sets; f MRCI/aug-cc-pV5Z; g MRCI+Q/56+CV+DK; h MRCI/modified basis sets; i Obtained from the RKR method; j MP2(full)/6-31G(d); k MRCI+Q/Q5+CV+DK. 表 4 SbS自由基Ω态的离解极限
Table 4. Dissociation relationships of the Ω states of SbS.
原子态 Ω态 ΔE/cm–1 实验值[68] 本文计算值 $\rm {{Sb} }({}^4{ {{S} }_{ {3/2} } }) + {{S} }{(^3}{ {{P} }_2})$ 7/2, 5/2(2),
3/2(3), 1/2(4)0 0 $\rm{{Sb} }({}^4{ {{S} }_{ {3/ } 2} }) + {{S} }{(^3}{ {{P} }_1})$ 5/2, 3/2(2),
1/2(3)396.055 410.46 $\rm {{Sb} }({}^4{ {{S} }_{ {3/2} } }) + {{S} }{(^3}{ {{P} }_0})$ 3/2, 1/2 573.640 605.81 表 5 SbS自由基的Ω态光谱常数
Table 5. Spectroscopic constants of the Ω states of SbS.
Ω态 Re/Å De/eV Be/cm–1 ωe/cm–1 ωeχe/cm–1 Te/cm–1 RMS/cm–1 X(1/2) 2.2195 3.62 0.1348 476.3 1.36 0 2.36 实验值[7] — — — 480 1.2 — X(3/2) 2.2201 3.36 0.1348 477.3 1.97 2025 2.25 实验值[7] — — — 470 1.6 — 2(1/2) 2.4527 1.93 0.1104 341.4 0.43 13646 2.70 3(1/2) 2.4538 1.90 0.1103 342.2 0.62 13888 2.28 2(3/2) 2.4503 1.86 0.1106 346.5 1.17 14123 0.30 1(5/2) 2.4537 1.83 0.1103 344.6 1.16 14346 0.38 3(3/2) 2.4428 2.56 0.1113 364.7 1.46 17632 2.94 4(1/2) 2.4467 2.48 0.1110 367.3 2.46 18340 9.74 4(3/2) 2.4524 2.18 0.1105 339.7 0.41 21413 3.17 5(1/2) 2.4560 2.13 0.1101 342.8 1.03 21610 2.17 6(1/2) 2.3462 1.95 0.1207 352.3 1.41 22947 6.01 5(3/2) 2.3476 1.91 0.1205 356.6 2.74 23268 8.04 表 6 XS (X = N, P, As, Sb, Bi)自由基Ω基态的光谱常数
Table 6. Spectroscopic constants of the ground Ω state of XS (X = N, P, As, Sb, Bi) radicals.
Ω态 Re/Å De/eV ωe/cm–1 Be/cm–1 Te/cm–1 NS X(1/2) 理论值[46] a 1.4962 4.8562 1216.43 0.77320 0 理论值[51] b 1.4976 4.7586 1213.30 — 0 实验值[12] 1.4955 — 1219.14 0.7730 0 实验值[13] 1.4955 — 1218.97 0.7730 0 实验值[14] 1.4931 — 1218.1 0.7758(11) 0 X(3/2) 理论值[46] a 1.4962 4.8446 1215.93 0.77326 223.64 理论值[51] b 1.4975 4.7412 1213.02 — 221.67 实验值[12] 1.4901 — 1218.90 0.7777 223.15 实验值[13] 1.4901 — 1218.90 0.7777 222.98 实验值[14] 1.4884 — 1218.0 0.7807(2) 220.4 PS X(1/2) 实验值[19] 1.899 — 739.54(2) 0.29724(5) 0 本文工作 1.9015 4.40 738.8 0.2960 0 X(3/2) 实验值[19] 1.897 4.566 739.45(2) 0.29765(5) 321.93 本文工作 1.9014 4.37 738.6 0.2960 324.8 AsS X(1/2) 实验值[24] — — 567.9(4) 0.18476 0 本文工作 2.0206 3.89 565.6 0.1839 0 X(3/2) 实验值[24] 2.0174 — 566.1(3) 0.18492 — 实验值[25] 2.0216(3) — 562.40(16) 0.18408(4) — 本文工作 2.0210 3.78 563.3 0.1838 893.3 SbS X(1/2) 本文工作 2.2195 3.62 476.3 0.1348 0 X(3/2) 本文工作 2.2201 3.36 477.3 0.1348 2025.0 BiS X(1/2) 理论值[45] c 2.365 — 407 — 0 实验值[29] 2.3194 — 408.71 0.11301 0 实验值[30] 2.3122(10) — 404.68(8) 0.11371(10) 0 实验值[31] 2.3188(1) — 408.67(7) 0.113063(10) 0 实验值[32] — — 408.66(3) — 0 本文工作 2.3131 3.58 429.5 0.1134 0 X(3/2) 理论值[45] c 2.361 — 404 — 7076 实验值[31] 2.31525(13) — 403.95(21) 0.113411(13) 6905.02(18) 实验值[32] 2.31489(11) — 404.501(94) — — 本文工作 2.3191 2.87 413.8 0.1128 5781 注: a MRCI+Q/AV5Z+CV+DK+SO; b MRCI+Q/56+CV+DK+SO; c MRDCI+Q/modified basis sets. 表 7 SbS的Λ-S及其对应Ω态的振动能级、转动常数和离心畸变常数(单位: cm–1)
Table 7. Vibrational energy levels, rotational constants and centrifugal distortion constants for the Ω and its respective Λ-S states of SbS (in cm–1).
v Gv Bv 108Dv Gv Bv 108Dv Gv Bv 108Dv X(1/2) X(3/2) ${\text{X}}{}^2\Pi $ 0 205.3 0.1350 4.30 200.7 0.1349 4.37 212.4 0.1351 4.18 1 682.7 0.1345 4.32 673.6 0.1344 4.39 696.4 0.1344 4.38 2 1156.9 0.1340 4.32 1143.1 0.1338 4.40 1171.8 0.1339 4.40 3 1628.2 0.1334 4.33 1609.4 0.1332 4.41 1642.2 0.1334 4.32 4 2096.6 0.1329 4.35 2072.6 0.1327 4.44 2110.0 0.1328 4.28 5 2561.9 0.1323 4.39 2532.6 0.1321 4.48 2575.7 0.1323 4.31 3(3/2) 4(1/2) ${2^2}\Pi $ 0 160.5 0.1118 4.32 165.5 0.1113 4.44 153.7 0.1119 4.42 1 519.5 0.1114 4.31 517.2 0.1109 4.44 508.8 0.1114 4.38 2 876.8 0.1109 4.29 866.9 0.1104 4.42 862.3 0.1108 4.37 3 1232.5 0.1105 4.29 1214.7 0.1100 4.43 1214.1 0.1104 4.33 4 1586.4 0.1101 4.32 1560.6 0.1095 4.46 1564.3 0.1099 4.34 5 1938.3 0.1097 4.34 1904.3 0.1090 4.49 1912.9 0.1095 4.35 4(3/2) 5(1/2) ${3^2}\Pi $ 0 169.7 0.1106 4.58 170.6 0.1103 4.59 163.4 0.1108 4.73 1 512.3 0.1101 4.59 511.8 0.1098 4.60 501.1 0.1102 4.54 2 852.6 0.1096 4.59 850.7 0.1093 4.60 839.6 0.1096 4.57 3 1190.7 0.1091 4.60 1187.4 0.1089 4.61 1176.7 0.1091 4.58 4 1526.5 0.1086 4.62 1521.9 0.1084 4.63 1512.0 0.1086 4.61 5 1860.0 0.1081 4.62 1854.0 0.1079 4.64 1845.0 0.1081 4.64 6(1/2) 5(3/2) ${1^4}{\Sigma ^ - }$ 0 131.6 0.1204 5.73 128.3 0.1203 5.80 151.1 0.1215 5.33 1 479.4 0.1197 5.60 473.6 0.1196 5.72 517.9 0.1214 5.42 2 826.2 0.1190 5.66 817.0 0.1189 5.75 881.0 0.1204 6.22 3 1170.2 0.1183 5.85 1157.7 0.1182 5.86 1230.6 0.1194 6.24 4 1510.2 0.1177 5.91 1494.9 0.1175 5.91 1571.7 0.1186 5.89 5 1846.6 0.1171 5.66 1828.6 0.1169 5.82 1909.2 0.1179 5.87 2(1/2) 3(1/2) $1{}^4\Pi $ 0 170.1 0.1106 4.55 170.3 0.1106 4.55 160.2 0.1111 4.73 1 514.3 0.1102 4.55 514.0 0.1101 4.56 499.2 0.1104 4.56 2 856.3 0.1097 4.56 855.5 0.1096 4.57 838.7 0.1099 4.63 3 1196.0 0.1092 4.57 1194.7 0.1091 4.58 1176.2 0.1094 4.65 4 1533.5 0.1087 4.58 1531.6 0.1086 4.59 1511.4 0.1089 4.67 5 1868.7 0.1082 4.58 1866.2 0.1081 4.60 1844.2 0.1084 4.69 2(3/2) 1(5/2) 0 168.8 0.1108 4.56 170.4 0.1105 4.57 1 513.4 0.1103 4.57 513.2 0.1100 4.58 2 855.6 0.1098 4.58 853.6 0.1095 4.58 3 1195.5 0.1093 4.59 1191.7 0.1090 4.60 4 1533.0 0.1088 4.60 1527.5 0.1085 4.61 5 1868.2 0.1083 4.61 1860.8 0.1080 4.62 表 8 XS (X = P, As, Bi)自由基Ω基态的振动能级、转动常数和离心畸变常数(单位: cm–1)
Table 8. Vibrational energy levels, rotational constants and centrifugal distortion constants for the ground Ω state of XS (X = P, As, Bi) radicals (in cm–1).
v Gv Bv 107Dv Gv Bv 107Dv v Gv Bv 108Dv PS X(1/2) PS X(3/2) AsS X(3/2) 0 368.4 0.29550 1.91 368.2 0.29551 1.91 3 1952.3 0.18108 7.90 — 0.29649 a 1.85 a — 0.29695 a 1.9 a — 0.18116(8) b 8.6(5) b 1 1101.3 0.29394 1.92 1100.8 0.29394 1.92 4 2501.2 0.18024 7.93 — 0.29469 a 1.7 a — 0.29543 a 1.8 a — 0.18033(4) b 8.7(8) b 2 1828.2 0.29237 1.92 1827.2 0.29237 1.92 5 3046.0 0.17939 7.93 — 0.29333 a 1.9 a — 0.29385 a 2.0 a — 0.17950(4) b 8.8(7) b 3 2549.0 0.29078 1.92 2547.6 0.29078 1.92 6 3587.0 0.17853 7.89 — 0.29161 a 1.85 a — 0.29223 a 1.95 a — 0.17865(5) b 9.1(9) b 4 3264.0 0.28918 1.92 3262.2 0.28917 1.93 7 4124.1 0.17765 7.85 — 0.29015 a 1.9 a — 0.29065 a 1.8 a — 0.17782(4) b 9.7(8) b 5 3973.2 0.28756 1.93 3971.0 0.28755 1.93 — 0.28855 a 2.0 a — 0.28933 a 1.8 a BiS X(1/2) 6 4676.7 0.28595 1.94 4674.0 0.28594 1.94 0 213.9 0.11344 3.19 — 0.28710 a 1.9 a — 0.28740 a 2.0 a — 0.112764(5) c 3.34(4) c 7 5374.2 0.28434 1.95 5371.0 0.28432 1.95 1 641.0 0.11302 3.19 — — — — 0.28653 a 1.7 a 2 1065.6 0.11260 3.19 8 6065.7 0.28273 1.96 6061.9 0.28271 1.96 3 1488.0 0.11218 3.20 — — — — 0.28416 a 2.0 a 注: a为文献[19]实验值; b为文献[25]实验值; c为文献[29]实验值. -
[1] Lian W T, Jiang C H, Yin Y W, Tang R F, Li G, Zhang L J, Che B, Chen T 2021 Nat. Commun. 12 3260
 Google Scholar
Google Scholar
[2] Zhao R M, Yang X L, Shi H L, Du M H 2021 Phys. Rev. Mater. 5 054605
 Google Scholar
Google Scholar
[3] Yang Y, Shi C W, Lv K, Wang Q, Sun X, Chen W C 2021 New J. Chem. 45 10357
 Google Scholar
Google Scholar
[4] Y Grad L, von Rohr F O, Hengsberger M, Osterwalder J 2021 Phys. Rev. Mater. 5 075401
 Google Scholar
Google Scholar
[5] Hu X K, Ma Y X, Pang Z X, Li P 2019 Chem. Phys. 523 110
 Google Scholar
Google Scholar
[6] Zhang J W, Lian W T, Yin Y W, et al. 2020 Solar RRL 4 2000048
 Google Scholar
Google Scholar
[7] Shimauchi M, Nishiyama Y 1968 Sci. Light 17 76
[8] Fowler A, Bakker C J 1932 Proc. Roy. Soc. (London) A 136 28
[9] Zeeman P B 1951 Can. J Phys. 29 174
 Google Scholar
Google Scholar
[10] Barrow R F, Drummond G, Zeeman P B 1954 Proc. Roy. Soc. (London) A 67 365
[11] Amano T, Saito S, Hirota E, Morino Y 1969 J. Mol. Spectrosc. 32 97
 Google Scholar
Google Scholar
[12] Jenouvrier A, Pascat B 1973 Can. J. Phys. 51 2143
 Google Scholar
Google Scholar
[13] Jenouvrier A, Pascat B 1980 Can. J. Phys. 58 1275
 Google Scholar
Google Scholar
[14] Wang T T, Li C Y, Zheng X F, Chen Y 2007 Chin. Sci. Bull. 52 596
 Google Scholar
Google Scholar
[15] Dressler K, Miescher E 1955 Proc. Roy. Soc. (London) A 68 542
 Google Scholar
Google Scholar
[16] Dressler K 1955 Ph. D. Dissertation (Basel: Universität Basel)
[17] Narasimham N A, Subramanian T K B 1969 J. Mol. Spectrosc. 29 294
 Google Scholar
Google Scholar
[18] Narasimham N A, Subramanian T K B 1971 J. Mol. Spectrosc. 37 371
 Google Scholar
Google Scholar
[19] Jenouvrier A, Pascat B 1978 Can. J. Phys. 56 1088
 Google Scholar
Google Scholar
[20] Balasubramanian T K, Dixit M N, Narasimham N A 1979 Pramana 12 707
 Google Scholar
Google Scholar
[21] Kawaguchi K, Hirota E, Ohishi M, Suzuki H, Takano S, Yamamoto S, Saito S 1988 J. Mol. Spectrosc. 130 81
 Google Scholar
Google Scholar
[22] Ohishi M, Yamamoto S, Saito S, et al. 1988 Astrophys. J 329 511
 Google Scholar
Google Scholar
[23] Klein H, Klisch E, Winnewisser G 1999 Z. Naturforschung A 54 137
 Google Scholar
Google Scholar
[24] Shimauchi M, 1969 Sci. Light 18 90
[25] Shimauchi M, 1971 Can. J. Phys. 49 1249
 Google Scholar
Google Scholar
[26] Shimauchi M, Sakaba Y, Kikuchi S 1972 Sci. Light 21 1
[27] Shimauchi M, Iwata H, Matsuno T, Sakaba Y, Lee S K, Karasawa S 1972 Sci. Light 21 145
[28] Shimauchi M, Karasawa S 1973 Sci. Light 22 127
[29] Barrow R F, Stobart O V, Vaughan H 1967 Proc. Phys. Soc. Lond. 90 555
 Google Scholar
Google Scholar
[30] Patiño P, Eland J H D, Barrow R F 1984 J. Phys. B:At. Mol. Phys. 17 1009
 Google Scholar
Google Scholar
[31] Izumi K, Cohen E A, Setzer K D, Fink E H, Kawaguchi K 2008 J. Mol. Spectrosc. 252 198
 Google Scholar
Google Scholar
[32] Setzer K D, Meinecke F, Fink E H 2009 J. Mol. Spectrosc. 258 56
 Google Scholar
Google Scholar
[33] O’Hare P A G 1970 J. Chem. Phys. 52 2992
 Google Scholar
Google Scholar
[34] Bialski M, Grein F 1976 J. Mol. Spectrosc. 61 321
 Google Scholar
Google Scholar
[35] Karna S P, Grein F 1986 J. Mol. Spectrosc. 120 284
 Google Scholar
Google Scholar
[36] Karna S P, Bruna P J, Grein F 1988 J. Phys. B:At. Mol. Opt. Phys. 21 1303
 Google Scholar
Google Scholar
[37] Karna S P, Grein F 1992 Mol. Phys. 77 135
 Google Scholar
Google Scholar
[38] Chong D P 1994 Chem. Phys. Lett. 220 102
 Google Scholar
Google Scholar
[39] Moussaoui Y, Ouamerali O, De Maré G R 1998 J Mol. Struct. Theochem. 425 237
 Google Scholar
Google Scholar
[40] Kalcher J 2002 Phys. Chem. Chem. Phys. 4 3311
 Google Scholar
Google Scholar
[41] Peebles L R, Marshall P 2002 Chem. Phys. Lett. 366 520
 Google Scholar
Google Scholar
[42] Czernek J, Živný O 2004 Chem. Phys. 303 137
 Google Scholar
Google Scholar
[43] Yaghlane S B, Francisco J S, Hochlaf M 2012 J. Chem. Phys. 136 244309
 Google Scholar
Google Scholar
[44] Yang, J, Kang Y, Wang X, Bai X 2013 J. Mol. Model 19 5199
 Google Scholar
Google Scholar
[45] Lingott R M, Liebermann H P, Alekseyev A B, Buenker R J 1999 J. Chem. Phys. 110 11294
 Google Scholar
Google Scholar
[46] Shi D H, Xing W, Sun J F, Zhu Z L 2012 Eur. Phys. J. D 66 173
 Google Scholar
Google Scholar
[47] Gao Y F, Gao T, Gong M 2013 J Quant. Spectrosc. Radiat. Transf. 129 193
 Google Scholar
Google Scholar
[48] 刘慧, 邢伟, 施德恒, 孙金锋, 朱遵略 2013 物理学报 62 203104
 Google Scholar
Google Scholar
Liu H, Xing W, Shi D H, Sun J F, Zhu Z L 2013 Acta Phys. Sin. 62 203104
 Google Scholar
Google Scholar
[49] Shi D H, Song Z Y, Niu X H, Sun J F, Zhu Z L 2016 Spectrochim. Acta A Mol. Biomol. Spectrosc. 153 30
 Google Scholar
Google Scholar
[50] Prajapat L, Jagoda P, Lodi L, Gorman M N, Yurchenko S N, Tennyson J 2017 MNRAS 472 3648
 Google Scholar
Google Scholar
[51] Zhou D, Shi D H, Sun J F 2019 J. Quant. Spectrosc. Radiat. Transf. 230 120
 Google Scholar
Google Scholar
[52] de Almeida A A, Andreazza C M, Borin A C 2020 Theor. Chem. Acc. 139 33
 Google Scholar
Google Scholar
[53] Reddy R R, Reddy A S R, Rao T V R 1985 Pramana 25 187
 Google Scholar
Google Scholar
[54] Knowles P J, Werner H J 1985 J. Chem. Phys. 82 5053
 Google Scholar
Google Scholar
[55] Werner H J, Knowles P J 1988 J. Chem. Phys. 89 5803
 Google Scholar
Google Scholar
[56] Peterson K A, Dunning Jr. T H 2002 J. Chem. Phys. 117 10548
 Google Scholar
Google Scholar
[57] Peterson K A, Yousaf K E 2010 J. Chem. Phys. 133 174116
 Google Scholar
Google Scholar
[58] Murrell J N, Sorbie K S 1974 J. Chem. Soc. Faraday Trans. 2 1552
[59] 李松, 韩立波, 陈善俊, 段传喜 2013 物理学报 62 113102
 Google Scholar
Google Scholar
Li S, Han L B, Chen S J, Duan C X 2013 Acta Phys. Sin. 62 113102
 Google Scholar
Google Scholar
[60] Li S, Chen S J, Zhu D S, Fan Q C 2013 Comput. Theor. Chem. 1017 136
 Google Scholar
Google Scholar
[61] Lu N, Wu W Q, Zhang C Z, Wan M J, Jin Y Y, Zhang W B, Chen S J, Li S 2020 Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 237 118301
 Google Scholar
Google Scholar
[62] Li S, Chen S J, Chen Y, Chen P 2016 Chin. Phys. B. 25 033101
 Google Scholar
Google Scholar
[63] Chen P, Wang N, Li S, Chen S J 2017 J. Quant. Spectrosc. Radiat. Transf. 201 104
 Google Scholar
Google Scholar
[64] 万明杰, 李松, 金成国, 罗华锋 2019 物理学报 68 063103
 Google Scholar
Google Scholar
Wan M J, Li S, Jin C G, Luo H F 2019 Acta Phys. Sin. 68 063103
 Google Scholar
Google Scholar
[65] Berning A, Schweizer M, Werner H J, Knowles P J, Palmieri P 2000 Mol. Phys. 98 1823
 Google Scholar
Google Scholar
[66] Werner H J, Knowles P J, Knizia G, et al. MOLPRO, version 2015.1, A Package of ab initio Programs, 2015
[67] Le Roy R J 2017 J. Quant. Spectrosc. Radiat. Transf. 186 167
 Google Scholar
Google Scholar
[68] Sansonetti J E, Martin W C 2005 J. Phys. Chem. Ref. Data 34 1559
 Google Scholar
Google Scholar
计量
- 文章访问数: 10277
- PDF下载量: 96
- 被引次数: 0













 下载:
下载: