-
自旋器件有望实现量子信息存储、传感和计算, 是下一代数据存储和通信的理想器件. 与无机自旋器件相比, 有机自旋器件不仅可以实现传统无机自旋器件的功能, 而且在同一有机自旋阀器件中会同时测到正负磁电阻信号, 这是因为有机分子与铁磁电极在界面会发生自旋杂化而产生独特的自旋界面. 通过控制自旋界面, 可以改变界面处分子能级展宽和偏移程度, 从而实现对磁电阻信号的可控调制. 有机自旋阀器件发展迅速, 但仍有一些问题亟待研究, 如对自旋界面进行识别和表征, 以及利用自旋界面对有机自旋阀信号进行操控等. 针对上述问题, 本文首先综述了有机自旋阀的基本原理, 通过对比无机有机材料能级结构的差异解释了有机自旋阀中自旋界面形成的原因, 对于有机自旋阀中磁电阻信号的增强和反转现象, 利用自旋界面模型中能级展宽和偏移进行了解释; 接着列举了自旋界面的实验识别案例, 如利用对表面敏感的表征技术对自旋界面进行识别以及设计新颖的器件结构验证自旋界面的存在等; 然后汇总了利用自旋界面调制自旋信号的相关工作, 自旋界面的调制可以通过电场调节铁电层的铁电极化、诱导铁磁电极相变、界面化学工程和磁交换相互作用等方式实现; 最后总结了有机自旋界面中仍需解决的问题, 并对有机自旋界面的识别和可控利用进行了展望.Spintronics are attractive to the utilization in next-generation quantum-computing and memory. Compared with inorganic spintronics, organic spintronics not only controls the spin degree-of-freedom but also possesses advantages such as chemical tailorability, flexibility, and low-cost fabrication process. Besides, the organic spin valve with a sandwich configuration that is composed of two ferromagnetic electrodes and an organic space layer is one of the classical devices in organic spintronics. Greatly enhanced or inversed magnetoresistance (MR) sign appearing in organic spin valve is induced by the unique interfacial effect an organic semiconductor/ferromagnetic interface. The significant enhancement or inversion of MR is later proved to be caused by the spin-dependent hybridization between molecular and ferromagnetic interface, i.e., the spinterface. The hybridization is ascribed to spin-dependent broadening and shifting of molecular orbitals. The spinterface takes place at one molecular layer when attaching to the surface of ferromagnetic metal. It indicates that the MR response can be modulated artificially in a specific device by converting the nature of spinterface. Despite lots of researches aiming at exploring the mechanism of spinterface, several questions need urgently to be resolved. For instance, the spin polarization, which is difficult to identify and observe with the surface sensitive technique and the inversion or enhancement of MR signal, which is also hard to explain accurately. The solid evidence of spinterface existing in real spintronic device also needs to be further testified. Besides, the precise manipulation of the MR sign by changing the nature of spinterface is quite difficult. According to the above background, this review summarizes the advance in spinterface and prospects future controllable utilization of spinterface. In Section 2, we introduce the basic principle of spintronic device and spinterface. The formation of unique spinterface in organic spin valve is clarified by using the difference in energy level alignment between inorganic and organic materials. Enhancement and inversion of MR sign are related to the broadening and shifting of the molecular level. In Section 3, several examples about identification of spinterface are listed, containing characterization by surface sensitive techniques and identification in real working devices. In Section 4 some methods about the manipulation of spinterface are exhibited, including modulation of ferroelectric organic barrier, interface engineering, regulation of electronic phase separation in ferromagnetic electrodes, etc. Finally, in this review some unresolved questions in spintronics are given, such as multi-functional and room-temperature organic spin valve and improvement of the spin injection efficiency. Spinterface is of great importance for both scientific research and future industrial interest in organic spintronics. The present study paves the way for the further development of novel excellent organic spin valves.
-
Keywords:
- organic spin valve /
- spinterface /
- organic spintronics /
- magnetoresistance
[1] Chiang C K, Fincher C R, Park Y W, Heeger A J, Shirakawa H, Louis E J, Gau S C, MacDiarmid A G 1977 Phys. Rev. Lett. 39 1098
 Google Scholar
Google Scholar
[2] Aviram A, Ratner M A 1974 Chem. Phys. Lett. 29 277
 Google Scholar
Google Scholar
[3] Aragonès A C, Aravena D, Cerdá J I, Acís-Castillo Z, Li H, Real J A, Sanz F, Hihath J, Ruiz E, Díez-Pérez I 2016 Nano Lett. 16 218
 Google Scholar
Google Scholar
[4] Guo L, Gu X, Zhu X, Sun X 2019 Adv. Mater. 31 1805355
 Google Scholar
Google Scholar
[5] Devkota J, Geng R G, Subedi R C, Nguyen T D 2016 Adv. Funct. Mater. 26 3881
 Google Scholar
Google Scholar
[6] Xin N, Guan J X, Zhou C G, Chen X J N, Gu C H, Li Y, Ratner M A, Nitzan A, Stoddart J F, Guo X F 2019 Nat. Rev. Phys. 1 211
 Google Scholar
Google Scholar
[7] Gehring P, Thijssen J M, van der Zant H S J 2019 Nat. Rev. Phys. 1 381
 Google Scholar
Google Scholar
[8] Ding S S, Tian Y, Li Y, Zhang H T, Zhou K, Liu J Y, Qin L, Zhang X X, Qiu X H, Dong H L, Zhu D B, Hu W P 2019 ACS Nano 13 9491
 Google Scholar
Google Scholar
[9] Sun D, Ehrenfreund E, Valy Vardeny Z 2014 Chem. Commun. 50 1781
 Google Scholar
Google Scholar
[10] Sun X, Gobbi M, Bedoya-Pinto A, Txoperena O, Golmar F, Llopis R, Chuvilin A, Casanova F, Hueso L E 2013 Nat. Commun. 4 2794
 Google Scholar
Google Scholar
[11] Gobbi M, Golmar F, Llopis R, Casanova F, Hueso L E 2011 Adv. Mater. 23 1609
 Google Scholar
Google Scholar
[12] Tran T L A, Le T Q, Sanderink J G M, van der Wiel W G, de Jong M P 2012 Adv. Funct. Mater. 22 1180
 Google Scholar
Google Scholar
[13] Dediu V A, Hueso L E, Bergenti I, Taliani C 2009 Nat. Mater. 8 707
 Google Scholar
Google Scholar
[14] Ding S, Tian Y, Hu W 2021 Nano Res. 14 3653
 Google Scholar
Google Scholar
[15] Xiong Z H, Wu D, Valy Vardeny Z, Shi J 2004 Nature 427 821
 Google Scholar
Google Scholar
[16] Nguyen T D, Ehrenfreund E, Vardeny Z V 2012 Science 337 204
 Google Scholar
Google Scholar
[17] Wang C, Fu B, Zhang X, Li R, Dong H, Hu W 2020 ACS. Central. Sci. 6 636
 Google Scholar
Google Scholar
[18] Zhou K, Dai K, Liu C, Shen C 2020 SmartMat 1 e1010
[19] Yao Y, Chen Y, Wang H, Samorì P 2020 SmartMat 1 e1009
[20] Huang Y, Gong X, Meng Y, Wang Z, Chen X, Li J, Ji D, Wei Z, Li L, Hu W 2021 Nat. Commun. 12 21
 Google Scholar
Google Scholar
[21] Chow P C Y, Someya T 2020 Adv. Mater. 32 1902045
 Google Scholar
Google Scholar
[22] Krinichnyi V I, Chemerisov S D, Lebedev Y S 1997 Phy. Rev. B 55 16233
 Google Scholar
Google Scholar
[23] Zhang X, Tong J, Ruan L, Yao X, Zhou L, Tian F, Qin G 2020 Phys. Chem. Chem. Phys. 22 11663
 Google Scholar
Google Scholar
[24] Boehme C, Lupton J M 2013 Nat. Nanotechnol. 8 612
 Google Scholar
Google Scholar
[25] Tsurumi J, Matsui H, Kubo T, Häusermann R, Mitsui C, Okamoto T, Watanabe S, Takeya J 2017 Nat. Phys. 13 994
 Google Scholar
Google Scholar
[26] Sanvito S 2011 Chem. Soc. Rev. 40 3336
 Google Scholar
Google Scholar
[27] Žutić I, Fabian J, Das Sarma S 2004 Rev. Mod. Phys. 76 323
 Google Scholar
Google Scholar
[28] Sun X N, Velez S, Atxabal A, Bedoya-Pinto A, Parui S, Zhu X W, Llopis R, Casanova F, Hueso L E 2017 Science 357 677
 Google Scholar
Google Scholar
[29] Koplovitz G, Primc D, Ben Dor O, Yochelis S, Rotem D, Porath D, Paltiel Y 2017 Adv. Mater. 29 1606748
 Google Scholar
Google Scholar
[30] Jang H J, Richter C A 2017 Adv. Mater. 29 1602739
 Google Scholar
Google Scholar
[31] Wang Y, Yao J, Ding S, Guo S, Cui D, Wang X, Yang S, Zhang L, Tian X, Wu D, Jin C, Li R, Hu W 2021 Sci. China Mater. 64 2795
 Google Scholar
Google Scholar
[32] Kang J, Sangwan V K, Wood J D, Hersam M C 2017 Accounts. Chem. Res. 50 943
 Google Scholar
Google Scholar
[33] Sun X, Bedoya-Pinto A, Mao Z, Gobbi M, Yan W, Guo Y, Atxabal A, Llopis R, Yu G, Liu Y, Chuvilin A, Casanova F, Hueso L E 2016 Adv. Mater. 28 2609
 Google Scholar
Google Scholar
[34] Prezioso M, Riminucci A, Graziosi P, Bergenti I, Rakshit R, Cecchini R, Vianelli A, Borgatti F, Haag N, Willis M, Drew A J, Gillin W P, Dediu V A 2013 Adv. Mater. 25 534
 Google Scholar
Google Scholar
[35] Drew A J, Hoppler J, Schulz L, et al. 2009 Nat. Mater. 8 109
 Google Scholar
Google Scholar
[36] Cinchetti M, Heimer K, Wüstenberg J P, Andreyev O, Bauer M, Lach S, Ziegler C, Gao Y, Aeschlimann M 2009 Nat. Mater. 8 115
 Google Scholar
Google Scholar
[37] Lach S, Altenhof A, Tarafder K, Schmitt F, Ali M E, Vogel M, Sauther J, Oppeneer P M, Ziegler C 2012 Adv. Funct. Mater. 22 989
 Google Scholar
Google Scholar
[38] Prezioso M, Riminucci A, Bergenti I, Graziosi P, Brunel D, Dediu V A 2011 Adv. Mater. 23 1371
 Google Scholar
Google Scholar
[39] Hu B, Wu Y 2007 Nat. Mater. 6 985
 Google Scholar
Google Scholar
[40] Li T, Xu L, Xiao X, Chen F, Cao L, Wu W, Tong W, Zhang F 2020 ACS Appl. Mater. Interf. 12 2708
 Google Scholar
Google Scholar
[41] Dediu V, Hueso L E, Bergenti I, Riminucci A, Borgatti F, Graziosi P, Newby C, Casoli F, De Jong M P, Taliani C, Zhan Y 2008 Phys. Rev. B 78 115203
 Google Scholar
Google Scholar
[42] Heimel G, Romaner L, Zojer E, Bredas J-L 2008 Accounts. Chem. Res. 41 721
 Google Scholar
Google Scholar
[43] Liu C, Xu Y, Noh Y Y 2015 Mater. Today 18 79
 Google Scholar
Google Scholar
[44] Barraud C, Seneor P, Mattana R, Fusil S, Bouzehouane K, Deranlot C, Graziosi P, Hueso L, Bergenti I, Dediu V, Petroff F, Fert A 2010 Nat. Phys. 6 615
 Google Scholar
Google Scholar
[45] Bergenti I, Dediu V 2019 Nano Mater. Sci. 1 149
 Google Scholar
Google Scholar
[46] Galbiati M, Tatay S, Barraud C, Dediu A V, Petroff F, Mattana R, Seneor P 2014 MRS Bull. 39 602
 Google Scholar
Google Scholar
[47] Brede J, Wiesendanger R 2012 Phys. Rev. B 86 184423
 Google Scholar
Google Scholar
[48] Ding S, Tian Y, Li Y, Mi W, Dong H, Zhang X, Hu W, Zhu D 2017 ACS Appl. Mater. Inter. 9 15644
 Google Scholar
Google Scholar
[49] Ciudad D, Gobbi M, Kinane C J, Eich M, Moodera J S, Hueso L E 2014 Adv. Mater. 26 7561
 Google Scholar
Google Scholar
[50] Han X, Mi W, Wang X 2019 J. Mater. Chem. C 7 4079
 Google Scholar
Google Scholar
[51] Liang S, Yang H, Yang H, et al. 2016 Adv. Mater. 28 10204
 Google Scholar
Google Scholar
[52] Naber W J M, Faez S, van der Wiel W G 2007 J. Phys. D. Appl. Phys. 40 R205
 Google Scholar
Google Scholar
[53] Julliere M 1975 Phys. Lett. A 54 225
 Google Scholar
Google Scholar
[54] Vardeny Z V 2010 Organic Spintronics (CRC Press) pp112, 140
[55] Schmidt G, Molenkamp L W 2002 Semicond. Sci. Tech. 17 310
 Google Scholar
Google Scholar
[56] Köhler A, Bässler H 2015 Electronic Processes in Organic Semiconductors (Wiley-VCH) p117
[57] Marta G 2016 Molecular Spintronics (Cham: Springer) pp7–9, 20–21, 32–36
[58] Ji D Y, Li T, Liu J, Amirjalayer S, Zhong M Z, Zhang Z Y, Huang X H, Wei Z M, Dong H L, Hu W P, Fuchs H 2019 Nat. Commun. 10 1
 Google Scholar
Google Scholar
[59] Fratini S, Nikolka M, Salleo A, Schweicher G, Sirringhaus H 2020 Nat. Mater. 19 491
 Google Scholar
Google Scholar
[60] Cohen M J, Coleman L B, Garito A F, Heeger A J 1974 Phys. Rev. B 10 1298
 Google Scholar
Google Scholar
[61] Berleb S, Brütting W 2002 Phys. Rev. Lett. 89 286601
 Google Scholar
Google Scholar
[62] Vázquez H, Oszwaldowski R, Pou P, Ortega J, Pérez R, Flores F, Kahn A 2004 Europhys. Lett. 65 802
 Google Scholar
Google Scholar
[63] Perrin M L, Verzijl C J O, Martin C A, Shaikh A J, Eelkema R, van Esch J H, van Ruitenbeek J M, Thijssen J M, van der Zant H S J, Dulić D 2013 Nat. Nanotechnol. 8 282
 Google Scholar
Google Scholar
[64] Atodiresei N, Brede J, Lazić P, Caciuc V, Hoffmann G, Wiesendanger R, Blügel S 2010 Phys. Rev. Lett. 105 066601
 Google Scholar
Google Scholar
[65] Kawahara S L, Lagoute J, Repain V, Chacon C, Girard Y, Rousset S, Smogunov A, Barreteau C 2012 Nano Lett. 12 4558
 Google Scholar
Google Scholar
[66] Shi S, Sun Z, Bedoya-Pinto A, Graziosi P, Li X, Liu X, Hueso L, Dediu V A, Luo Y, Fahlman M 2014 Adv. Funct. Mater. 24 4812
 Google Scholar
Google Scholar
[67] Iacovita C, Rastei M V, Heinrich B W, Brumme T, Kortus J, Limot L, Bucher J P 2008 Phys. Rev. Lett. 101 116602
 Google Scholar
Google Scholar
[68] Raman K V, Kamerbeek A M, Mukherjee A, Atodiresei N, Sen T K, Lazić P, Caciuc V, Michel R, Stalke D, Mandal S K, Blügel S, Münzenberg M, Moodera J S 2013 Nature 493 509
 Google Scholar
Google Scholar
[69] Ding S S, Tian Y, Wang H L, Zhou Z, Mi W B, Ni Z J, Zou Y, Dong H L, Gao H J, Zhu D B, Hu W P 2018 ACS Nano 12 12657
 Google Scholar
Google Scholar
[70] Ding S S, Tian Y, Liu X, Zou Y, Dong H L, Mi W B, Hu W P 2021 Nano Res. 14 304
 Google Scholar
Google Scholar
[71] Sun D, Fang M, Xu X, Jiang L, Guo H, Wang Y, Yang W, Yin L, Snijders P C, Ward T Z, Gai Z, Zhang X G, Lee H N, Shen J 2014 Nat. Commun. 5 4396
 Google Scholar
Google Scholar
[72] Yang W, Shi Q, Miao T, Li Q, Cai P, Liu H, Lin H, Bai Y, Zhu Y, Yu Y, Deng L, Wang W, Yin L, Sun D, Zhang X G, Shen J 2019 Nat. Commun. 10 3877
 Google Scholar
Google Scholar
[73] Raman K V 2014 Appl. Phys. Rev. 1 031101
 Google Scholar
Google Scholar
-
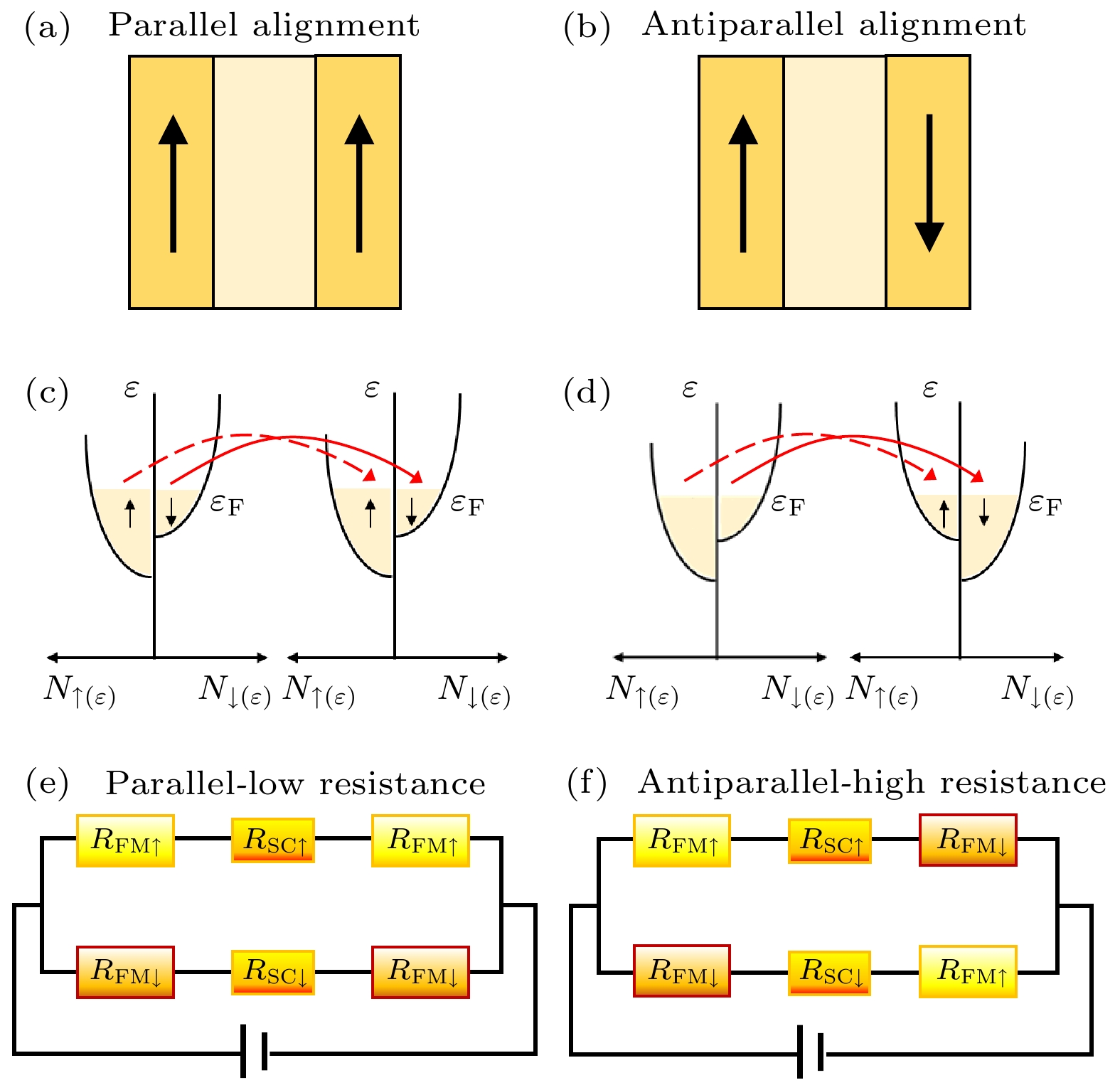
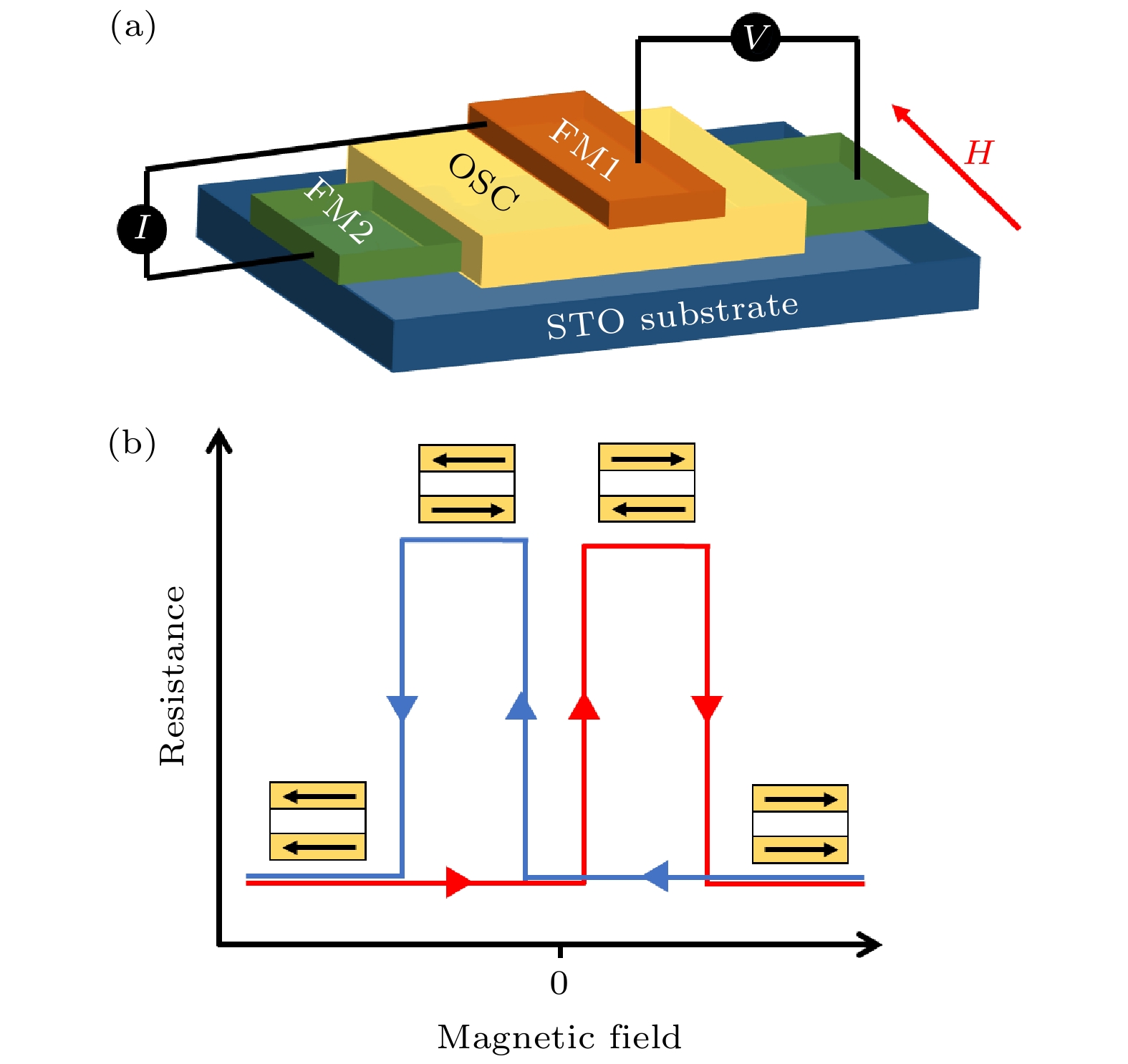
图 2 (a)和(b)磁性隧道结中铁磁电极在平行和反平行磁化状态的结构示意图; (c)和(d)在平行和反平行磁化状态下铁磁层的能带结构; (e)和(f)在磁化方向平行和反平行状态下的双电阻网络模型[54]
Fig. 2. (a) and (b) Schematics shows different states of ferromagnetic electrode with parallel and antiparallel magnetization in MTJ; (c) and (d) band structure of ferromagnetic layer for parallel and antiparallel magnetization; (e) and (f) two-resistor network model for magnetization of parallel and antiparallel alignment[54].
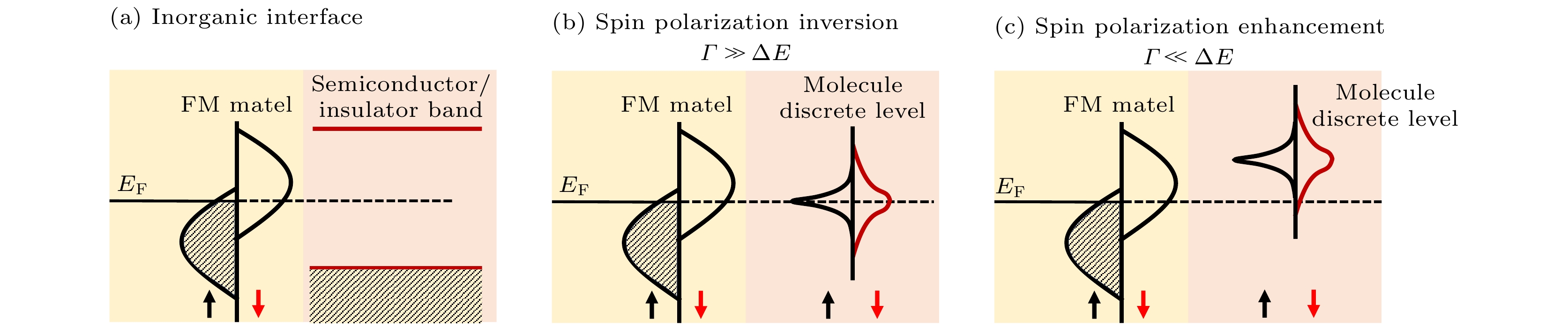
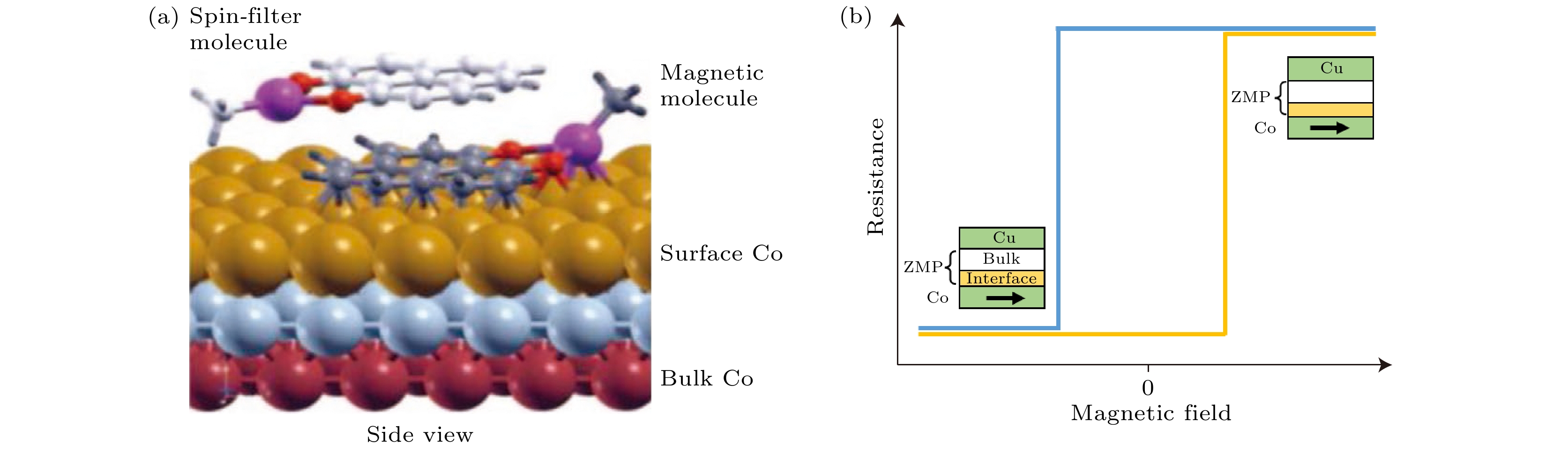
图 4 自旋界面的示意图 (a)无机物和铁磁电极接触界面的导带和价带示意图; (b)自旋界面处当
$\varGamma \gg \Delta E$ 时会诱导自旋极化的反转; (c)$\varGamma \ll \Delta E$ 时会造成自旋极化增强[46]Fig. 4. Schematics of spinterface[46]: (a) Schematics of conduction and valence band structure at inorganic/FM interface; (b) inversed spin-polarization case of
$\varGamma \gg \Delta E$ at the spinterface; (c) enhanced spin-polarization case of$\varGamma \ll \Delta E$ at the interface[46].图 5 (a) H2Pc吸附在Fe上的SP-STM图[64]; (b) 两个吸附在Cr (001)表面的C60分子上的电导图[65]; 吸附于Co基底上不同厚度TNAP的UPS图, 其中(c)—(e)分别对应(c)二次电子截止边、(d)价带边、(e)价带的细节谱图; (f)在Co上TNAP吸附前后Co的L边XMCD图; (g) 单层和多层TNAP在Co上N元素的K边NEXAFS图[66]
Fig. 5. (a) SP-STM image of H2Pc absorbed on Fe[64]. (b) conductance maps measured over two C60 molecules absorbed on Cr (001) surface[65]. UPS spectra of TNAP with different thickness deposited on Co substrate: (c) Secondary electron cutoff; (d) valence band; (e) detail spectral features of valence band. (f) Co L-edge XMCD results before and after adsorption of TNAP on Co; (g) NEXAFS N K-edge spectra of monolayer and multilayer TNAP on Co[66].
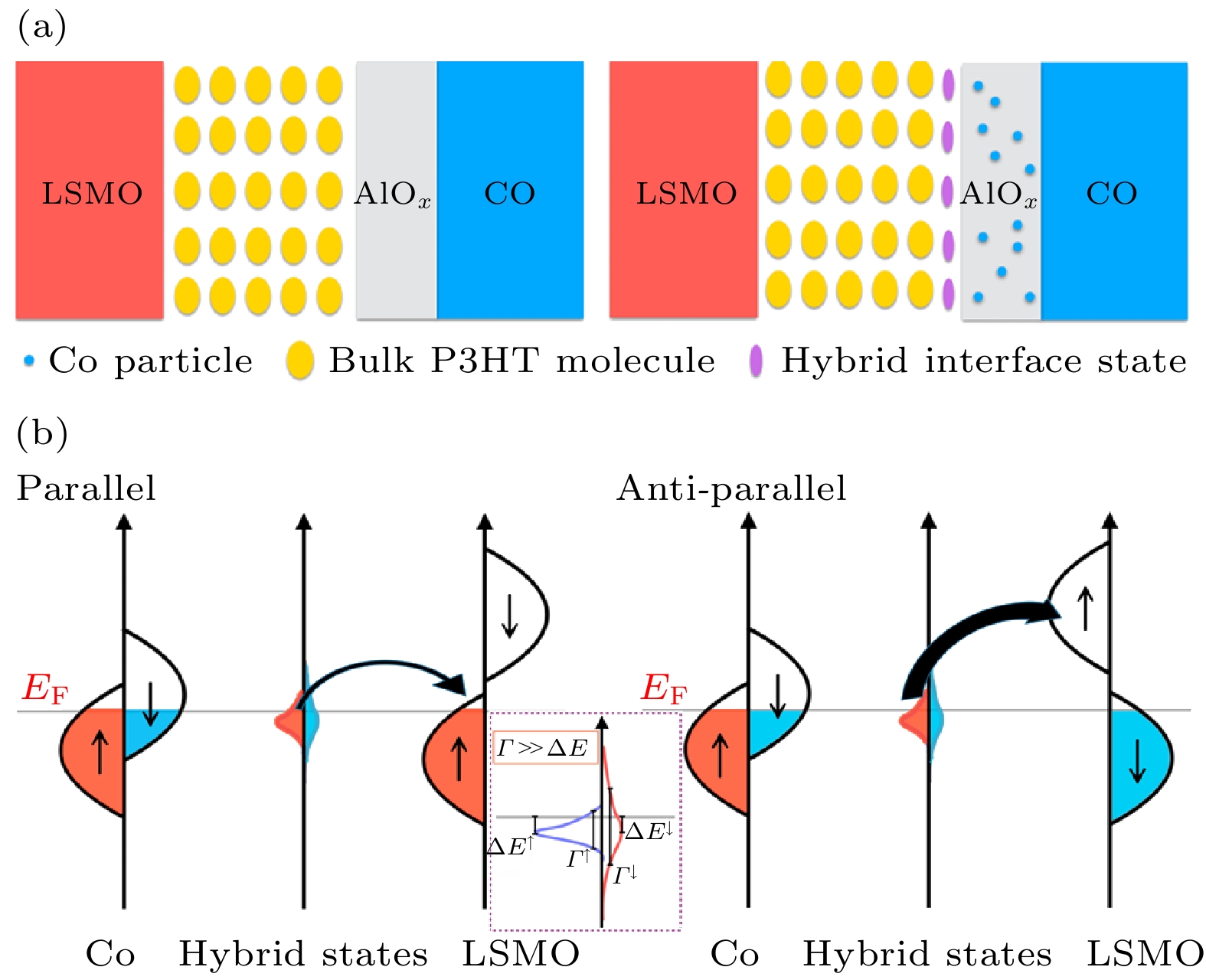
图 6 (a) AlOx绝缘层对Co渗透的阻挡作用及渗透的Co和P3HT间形成自旋界面的示意图; (b) LSMO/P3HT/AlOx/Co器件中自旋依赖电子隧穿过程示意图[48]
Fig. 6. (a) Schematic drawing of blocking effect for the insulated AlOx to penetrated Co, and the formation of spinterface between penetrated Co and P3HT molecular; (b) schematics of spin-dependent electron tunneling in LSMO/P3HT/AlOx/Co junction[48].
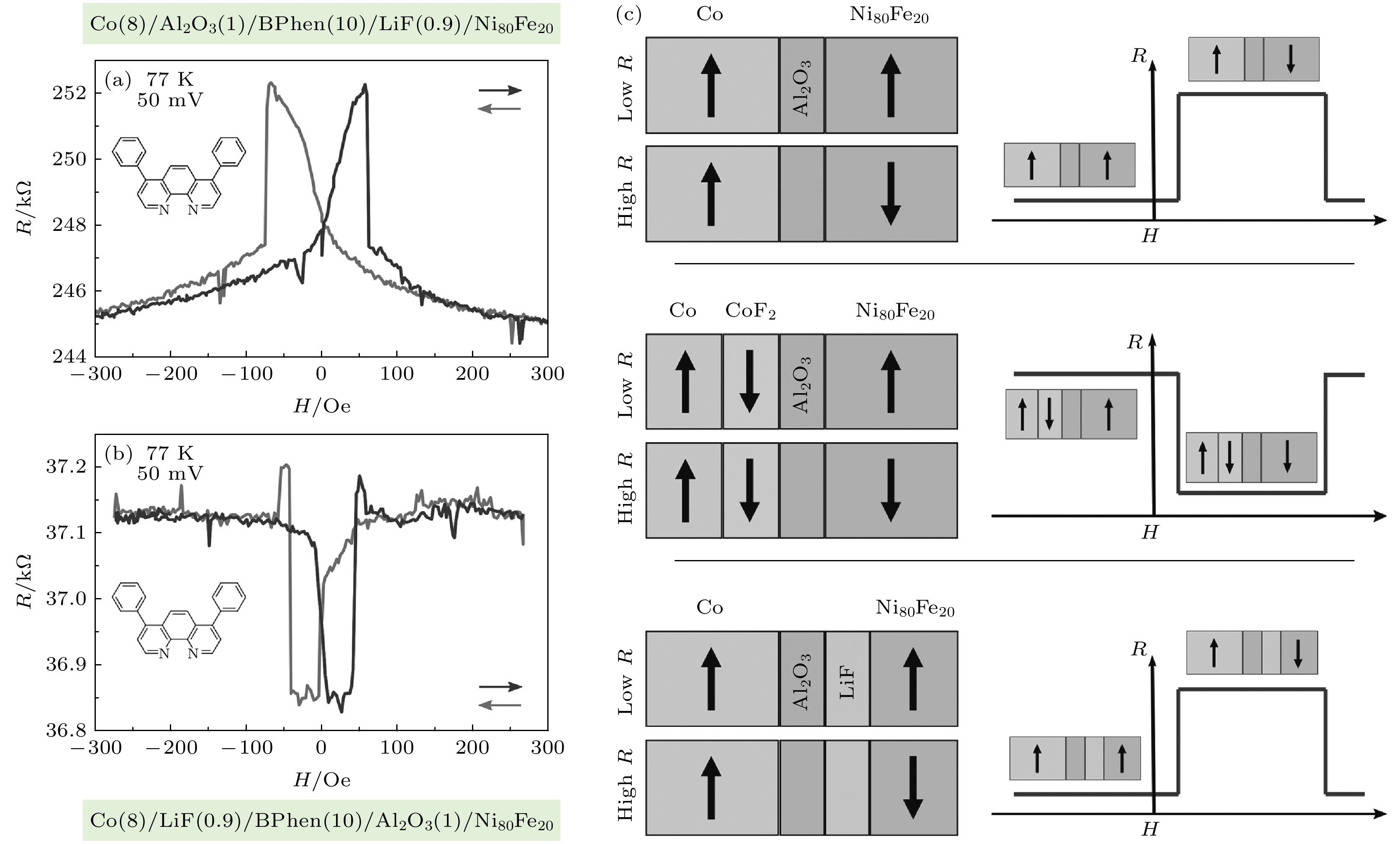
图 7 (a)和(b)分别为器件A和器件B的磁输运测试; (c)无LiF层、具有反铁磁双氟层和LiF沉积在氧化铝上的器件结构以及对应的磁电阻信号示意图[49]
Fig. 7. (a) and (b) Magnetotransport measurements of device A and device B, respectively; (c) schematics of devices with no LiF layer, an anti-ferromagnetic difluoride layer and LiF deposited on an alumina and their respective MR curves [49].
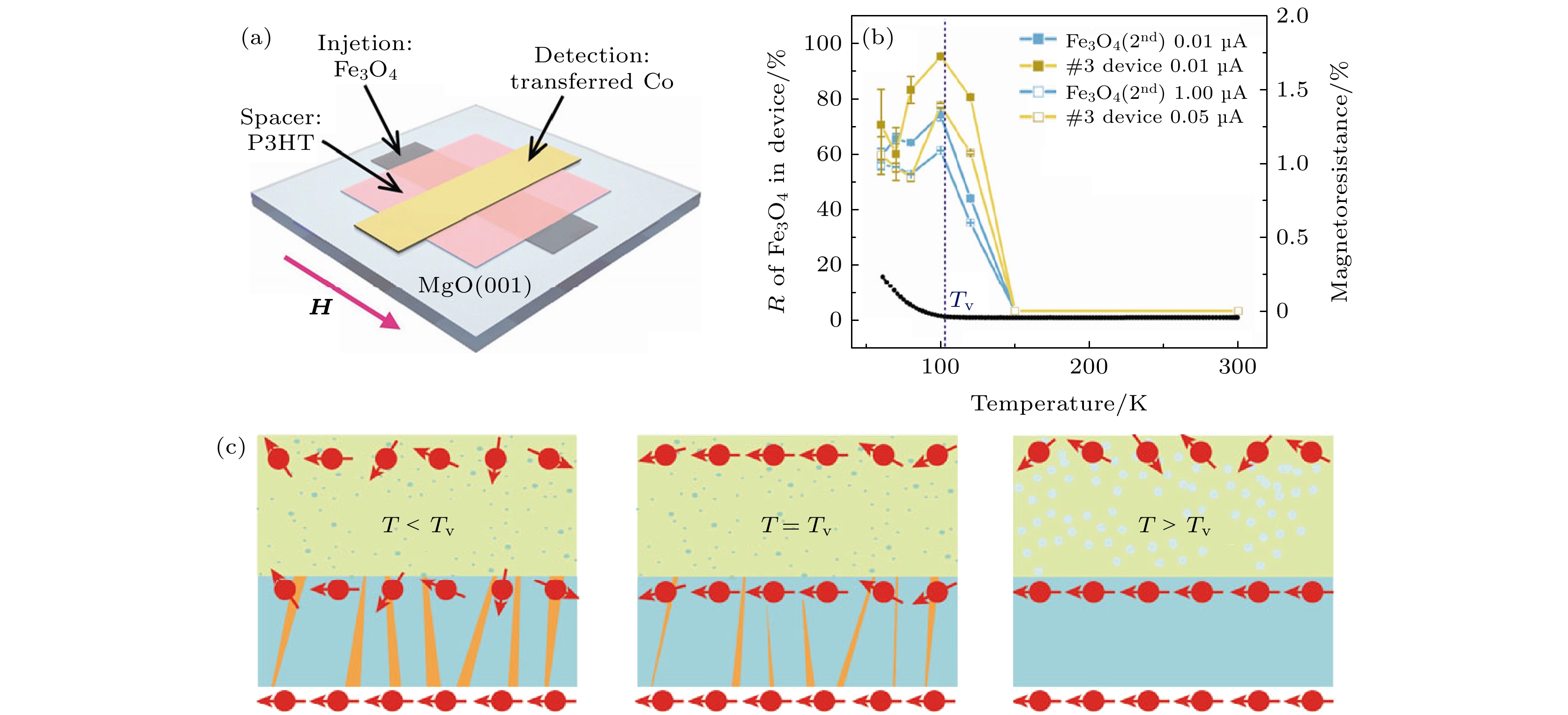
图 8 (a) Fe3O4/P3HT/Co有机自旋阀器件示意图; (b) 不同电流下Fe3O4/P3HT/Co有机自旋阀器件和Fe3O4电极MR值与温度的关系; (c) 不同温度下孪晶界对自旋注入调制过程的模型图[70]
Fig. 8. (a) Schematic of organic spin valve device of Fe3O4/P3HT/Co; (b) relationship between MR ratio and temperature for Fe3O4/P3HT/Co OSV device and Fe3O4 electrode at different bias current; (c) model of twin boundary-modulated spin injection at different temperature[70].
图 9 (a) LSMO/PVDF/Co器件示意图; (b)在PVDF表面测得的PFM相图; (c) 极化后器件所测得的隧穿磁电阻信号; (d)在10 mV, 10 K条件下测得LSMO/PVDF/Co器件的隧穿磁电阻; (e)在10 mV, 10 K条件下测得LSMO/PVDF/MgO/Co器件的隧穿磁电阻[51]
Fig. 9. (a) Schematic of LSMO/PVDF/Co device; (b) PFM phase image measured on the PVDF surface; (c) tunneling magnetoresistance measured after polarizing the device; (d) tunneling magneto resistance of a LSMO/PVDF/Co device measured under 10 mV at 10 K; (e) tunneling magneto resistance of a LSMO/PVDF/MgO/Co device measured under 10 mV at 10 K[51].
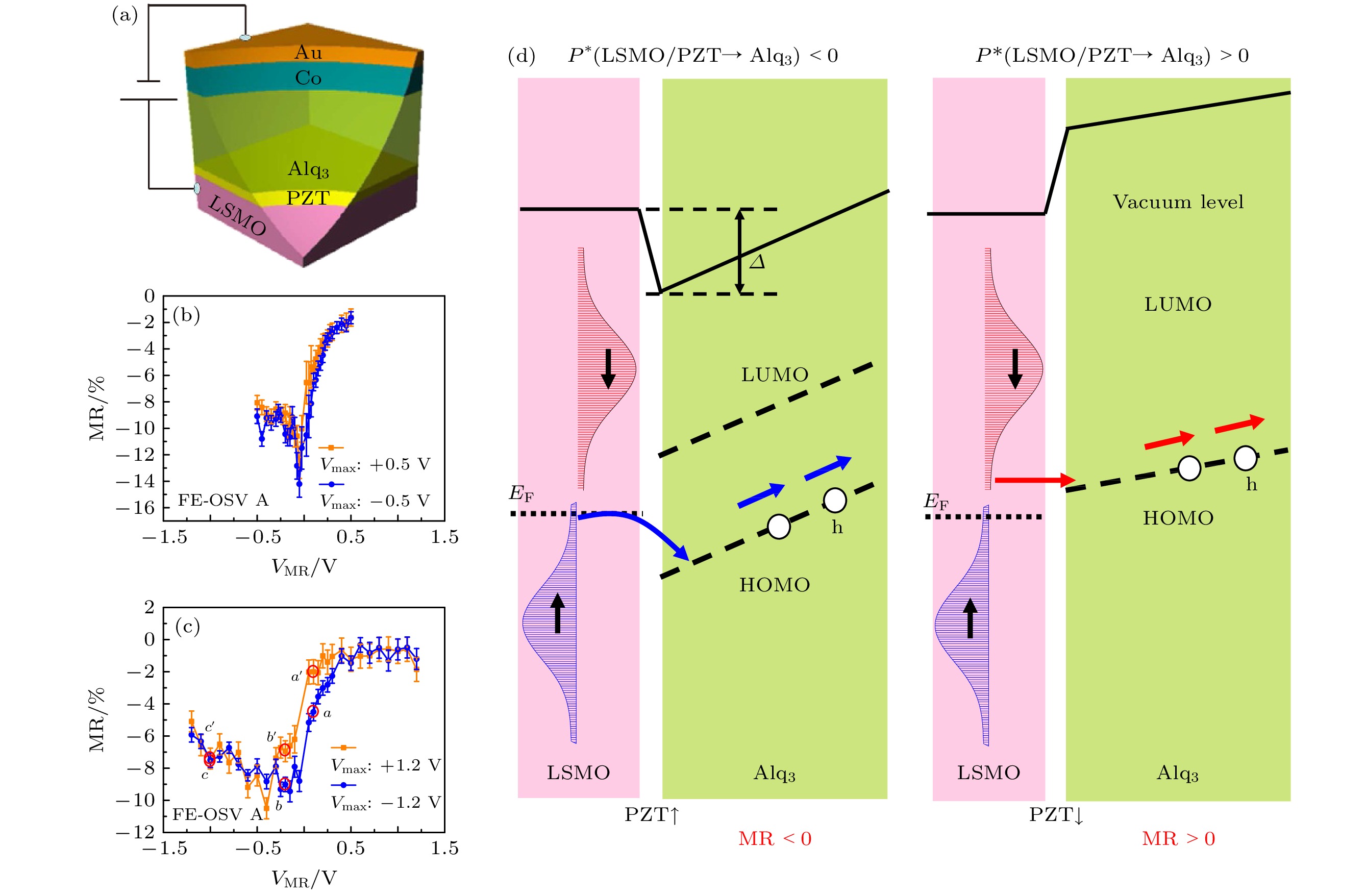
图 10 (a) Au/Co/Alq3/PZT/LSMO有机自旋阀的器件示意图; (b)和(c)施加不同预设电压后MR的偏移; (d)当PZT的电极化向上和向下时器件的能级关系示意图[71]
Fig. 10. (a) Schematic of a Au/Co/Alq3/PZT/LSMO organic spin valve device; (b) and (c) MR shift after applying different ramping voltage; (d) the energy relationship schematic of device when the electric polarization of the PZT is “up” and “down”[71].
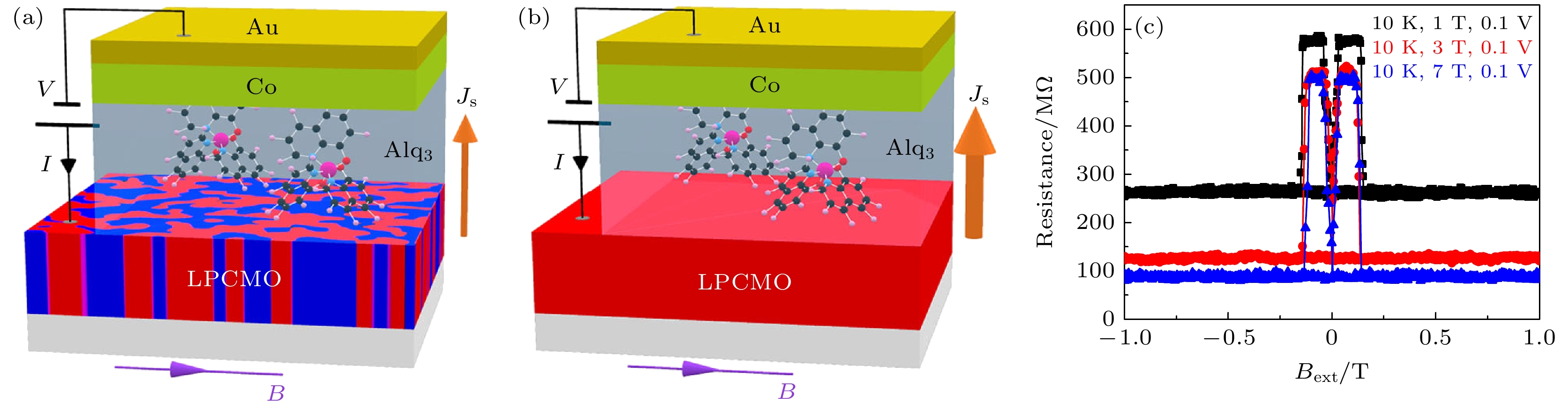
图 11 在LPCMO有机自旋阀中(a) FMM和COI相共存和(b)全FE相时的EPS调制自旋注入示意图; (c)在LPCMO有机自旋阀中不同预设磁场强度下的MR信号[72]
Fig. 11. Illustration of EPS-modulated spin current injection in the LPCMO-OSVs under the co-existed FM/COI phase (a) and fully FM phase (b) of the LPCMO thin film; (c) MR loops of the LPCMO-OSV device under different pre-set magnetic field strength[72].
-
[1] Chiang C K, Fincher C R, Park Y W, Heeger A J, Shirakawa H, Louis E J, Gau S C, MacDiarmid A G 1977 Phys. Rev. Lett. 39 1098
 Google Scholar
Google Scholar
[2] Aviram A, Ratner M A 1974 Chem. Phys. Lett. 29 277
 Google Scholar
Google Scholar
[3] Aragonès A C, Aravena D, Cerdá J I, Acís-Castillo Z, Li H, Real J A, Sanz F, Hihath J, Ruiz E, Díez-Pérez I 2016 Nano Lett. 16 218
 Google Scholar
Google Scholar
[4] Guo L, Gu X, Zhu X, Sun X 2019 Adv. Mater. 31 1805355
 Google Scholar
Google Scholar
[5] Devkota J, Geng R G, Subedi R C, Nguyen T D 2016 Adv. Funct. Mater. 26 3881
 Google Scholar
Google Scholar
[6] Xin N, Guan J X, Zhou C G, Chen X J N, Gu C H, Li Y, Ratner M A, Nitzan A, Stoddart J F, Guo X F 2019 Nat. Rev. Phys. 1 211
 Google Scholar
Google Scholar
[7] Gehring P, Thijssen J M, van der Zant H S J 2019 Nat. Rev. Phys. 1 381
 Google Scholar
Google Scholar
[8] Ding S S, Tian Y, Li Y, Zhang H T, Zhou K, Liu J Y, Qin L, Zhang X X, Qiu X H, Dong H L, Zhu D B, Hu W P 2019 ACS Nano 13 9491
 Google Scholar
Google Scholar
[9] Sun D, Ehrenfreund E, Valy Vardeny Z 2014 Chem. Commun. 50 1781
 Google Scholar
Google Scholar
[10] Sun X, Gobbi M, Bedoya-Pinto A, Txoperena O, Golmar F, Llopis R, Chuvilin A, Casanova F, Hueso L E 2013 Nat. Commun. 4 2794
 Google Scholar
Google Scholar
[11] Gobbi M, Golmar F, Llopis R, Casanova F, Hueso L E 2011 Adv. Mater. 23 1609
 Google Scholar
Google Scholar
[12] Tran T L A, Le T Q, Sanderink J G M, van der Wiel W G, de Jong M P 2012 Adv. Funct. Mater. 22 1180
 Google Scholar
Google Scholar
[13] Dediu V A, Hueso L E, Bergenti I, Taliani C 2009 Nat. Mater. 8 707
 Google Scholar
Google Scholar
[14] Ding S, Tian Y, Hu W 2021 Nano Res. 14 3653
 Google Scholar
Google Scholar
[15] Xiong Z H, Wu D, Valy Vardeny Z, Shi J 2004 Nature 427 821
 Google Scholar
Google Scholar
[16] Nguyen T D, Ehrenfreund E, Vardeny Z V 2012 Science 337 204
 Google Scholar
Google Scholar
[17] Wang C, Fu B, Zhang X, Li R, Dong H, Hu W 2020 ACS. Central. Sci. 6 636
 Google Scholar
Google Scholar
[18] Zhou K, Dai K, Liu C, Shen C 2020 SmartMat 1 e1010
[19] Yao Y, Chen Y, Wang H, Samorì P 2020 SmartMat 1 e1009
[20] Huang Y, Gong X, Meng Y, Wang Z, Chen X, Li J, Ji D, Wei Z, Li L, Hu W 2021 Nat. Commun. 12 21
 Google Scholar
Google Scholar
[21] Chow P C Y, Someya T 2020 Adv. Mater. 32 1902045
 Google Scholar
Google Scholar
[22] Krinichnyi V I, Chemerisov S D, Lebedev Y S 1997 Phy. Rev. B 55 16233
 Google Scholar
Google Scholar
[23] Zhang X, Tong J, Ruan L, Yao X, Zhou L, Tian F, Qin G 2020 Phys. Chem. Chem. Phys. 22 11663
 Google Scholar
Google Scholar
[24] Boehme C, Lupton J M 2013 Nat. Nanotechnol. 8 612
 Google Scholar
Google Scholar
[25] Tsurumi J, Matsui H, Kubo T, Häusermann R, Mitsui C, Okamoto T, Watanabe S, Takeya J 2017 Nat. Phys. 13 994
 Google Scholar
Google Scholar
[26] Sanvito S 2011 Chem. Soc. Rev. 40 3336
 Google Scholar
Google Scholar
[27] Žutić I, Fabian J, Das Sarma S 2004 Rev. Mod. Phys. 76 323
 Google Scholar
Google Scholar
[28] Sun X N, Velez S, Atxabal A, Bedoya-Pinto A, Parui S, Zhu X W, Llopis R, Casanova F, Hueso L E 2017 Science 357 677
 Google Scholar
Google Scholar
[29] Koplovitz G, Primc D, Ben Dor O, Yochelis S, Rotem D, Porath D, Paltiel Y 2017 Adv. Mater. 29 1606748
 Google Scholar
Google Scholar
[30] Jang H J, Richter C A 2017 Adv. Mater. 29 1602739
 Google Scholar
Google Scholar
[31] Wang Y, Yao J, Ding S, Guo S, Cui D, Wang X, Yang S, Zhang L, Tian X, Wu D, Jin C, Li R, Hu W 2021 Sci. China Mater. 64 2795
 Google Scholar
Google Scholar
[32] Kang J, Sangwan V K, Wood J D, Hersam M C 2017 Accounts. Chem. Res. 50 943
 Google Scholar
Google Scholar
[33] Sun X, Bedoya-Pinto A, Mao Z, Gobbi M, Yan W, Guo Y, Atxabal A, Llopis R, Yu G, Liu Y, Chuvilin A, Casanova F, Hueso L E 2016 Adv. Mater. 28 2609
 Google Scholar
Google Scholar
[34] Prezioso M, Riminucci A, Graziosi P, Bergenti I, Rakshit R, Cecchini R, Vianelli A, Borgatti F, Haag N, Willis M, Drew A J, Gillin W P, Dediu V A 2013 Adv. Mater. 25 534
 Google Scholar
Google Scholar
[35] Drew A J, Hoppler J, Schulz L, et al. 2009 Nat. Mater. 8 109
 Google Scholar
Google Scholar
[36] Cinchetti M, Heimer K, Wüstenberg J P, Andreyev O, Bauer M, Lach S, Ziegler C, Gao Y, Aeschlimann M 2009 Nat. Mater. 8 115
 Google Scholar
Google Scholar
[37] Lach S, Altenhof A, Tarafder K, Schmitt F, Ali M E, Vogel M, Sauther J, Oppeneer P M, Ziegler C 2012 Adv. Funct. Mater. 22 989
 Google Scholar
Google Scholar
[38] Prezioso M, Riminucci A, Bergenti I, Graziosi P, Brunel D, Dediu V A 2011 Adv. Mater. 23 1371
 Google Scholar
Google Scholar
[39] Hu B, Wu Y 2007 Nat. Mater. 6 985
 Google Scholar
Google Scholar
[40] Li T, Xu L, Xiao X, Chen F, Cao L, Wu W, Tong W, Zhang F 2020 ACS Appl. Mater. Interf. 12 2708
 Google Scholar
Google Scholar
[41] Dediu V, Hueso L E, Bergenti I, Riminucci A, Borgatti F, Graziosi P, Newby C, Casoli F, De Jong M P, Taliani C, Zhan Y 2008 Phys. Rev. B 78 115203
 Google Scholar
Google Scholar
[42] Heimel G, Romaner L, Zojer E, Bredas J-L 2008 Accounts. Chem. Res. 41 721
 Google Scholar
Google Scholar
[43] Liu C, Xu Y, Noh Y Y 2015 Mater. Today 18 79
 Google Scholar
Google Scholar
[44] Barraud C, Seneor P, Mattana R, Fusil S, Bouzehouane K, Deranlot C, Graziosi P, Hueso L, Bergenti I, Dediu V, Petroff F, Fert A 2010 Nat. Phys. 6 615
 Google Scholar
Google Scholar
[45] Bergenti I, Dediu V 2019 Nano Mater. Sci. 1 149
 Google Scholar
Google Scholar
[46] Galbiati M, Tatay S, Barraud C, Dediu A V, Petroff F, Mattana R, Seneor P 2014 MRS Bull. 39 602
 Google Scholar
Google Scholar
[47] Brede J, Wiesendanger R 2012 Phys. Rev. B 86 184423
 Google Scholar
Google Scholar
[48] Ding S, Tian Y, Li Y, Mi W, Dong H, Zhang X, Hu W, Zhu D 2017 ACS Appl. Mater. Inter. 9 15644
 Google Scholar
Google Scholar
[49] Ciudad D, Gobbi M, Kinane C J, Eich M, Moodera J S, Hueso L E 2014 Adv. Mater. 26 7561
 Google Scholar
Google Scholar
[50] Han X, Mi W, Wang X 2019 J. Mater. Chem. C 7 4079
 Google Scholar
Google Scholar
[51] Liang S, Yang H, Yang H, et al. 2016 Adv. Mater. 28 10204
 Google Scholar
Google Scholar
[52] Naber W J M, Faez S, van der Wiel W G 2007 J. Phys. D. Appl. Phys. 40 R205
 Google Scholar
Google Scholar
[53] Julliere M 1975 Phys. Lett. A 54 225
 Google Scholar
Google Scholar
[54] Vardeny Z V 2010 Organic Spintronics (CRC Press) pp112, 140
[55] Schmidt G, Molenkamp L W 2002 Semicond. Sci. Tech. 17 310
 Google Scholar
Google Scholar
[56] Köhler A, Bässler H 2015 Electronic Processes in Organic Semiconductors (Wiley-VCH) p117
[57] Marta G 2016 Molecular Spintronics (Cham: Springer) pp7–9, 20–21, 32–36
[58] Ji D Y, Li T, Liu J, Amirjalayer S, Zhong M Z, Zhang Z Y, Huang X H, Wei Z M, Dong H L, Hu W P, Fuchs H 2019 Nat. Commun. 10 1
 Google Scholar
Google Scholar
[59] Fratini S, Nikolka M, Salleo A, Schweicher G, Sirringhaus H 2020 Nat. Mater. 19 491
 Google Scholar
Google Scholar
[60] Cohen M J, Coleman L B, Garito A F, Heeger A J 1974 Phys. Rev. B 10 1298
 Google Scholar
Google Scholar
[61] Berleb S, Brütting W 2002 Phys. Rev. Lett. 89 286601
 Google Scholar
Google Scholar
[62] Vázquez H, Oszwaldowski R, Pou P, Ortega J, Pérez R, Flores F, Kahn A 2004 Europhys. Lett. 65 802
 Google Scholar
Google Scholar
[63] Perrin M L, Verzijl C J O, Martin C A, Shaikh A J, Eelkema R, van Esch J H, van Ruitenbeek J M, Thijssen J M, van der Zant H S J, Dulić D 2013 Nat. Nanotechnol. 8 282
 Google Scholar
Google Scholar
[64] Atodiresei N, Brede J, Lazić P, Caciuc V, Hoffmann G, Wiesendanger R, Blügel S 2010 Phys. Rev. Lett. 105 066601
 Google Scholar
Google Scholar
[65] Kawahara S L, Lagoute J, Repain V, Chacon C, Girard Y, Rousset S, Smogunov A, Barreteau C 2012 Nano Lett. 12 4558
 Google Scholar
Google Scholar
[66] Shi S, Sun Z, Bedoya-Pinto A, Graziosi P, Li X, Liu X, Hueso L, Dediu V A, Luo Y, Fahlman M 2014 Adv. Funct. Mater. 24 4812
 Google Scholar
Google Scholar
[67] Iacovita C, Rastei M V, Heinrich B W, Brumme T, Kortus J, Limot L, Bucher J P 2008 Phys. Rev. Lett. 101 116602
 Google Scholar
Google Scholar
[68] Raman K V, Kamerbeek A M, Mukherjee A, Atodiresei N, Sen T K, Lazić P, Caciuc V, Michel R, Stalke D, Mandal S K, Blügel S, Münzenberg M, Moodera J S 2013 Nature 493 509
 Google Scholar
Google Scholar
[69] Ding S S, Tian Y, Wang H L, Zhou Z, Mi W B, Ni Z J, Zou Y, Dong H L, Gao H J, Zhu D B, Hu W P 2018 ACS Nano 12 12657
 Google Scholar
Google Scholar
[70] Ding S S, Tian Y, Liu X, Zou Y, Dong H L, Mi W B, Hu W P 2021 Nano Res. 14 304
 Google Scholar
Google Scholar
[71] Sun D, Fang M, Xu X, Jiang L, Guo H, Wang Y, Yang W, Yin L, Snijders P C, Ward T Z, Gai Z, Zhang X G, Lee H N, Shen J 2014 Nat. Commun. 5 4396
 Google Scholar
Google Scholar
[72] Yang W, Shi Q, Miao T, Li Q, Cai P, Liu H, Lin H, Bai Y, Zhu Y, Yu Y, Deng L, Wang W, Yin L, Sun D, Zhang X G, Shen J 2019 Nat. Commun. 10 3877
 Google Scholar
Google Scholar
[73] Raman K V 2014 Appl. Phys. Rev. 1 031101
 Google Scholar
Google Scholar
计量
- 文章访问数: 15188
- PDF下载量: 527
- 被引次数: 0













 下载:
下载: