-
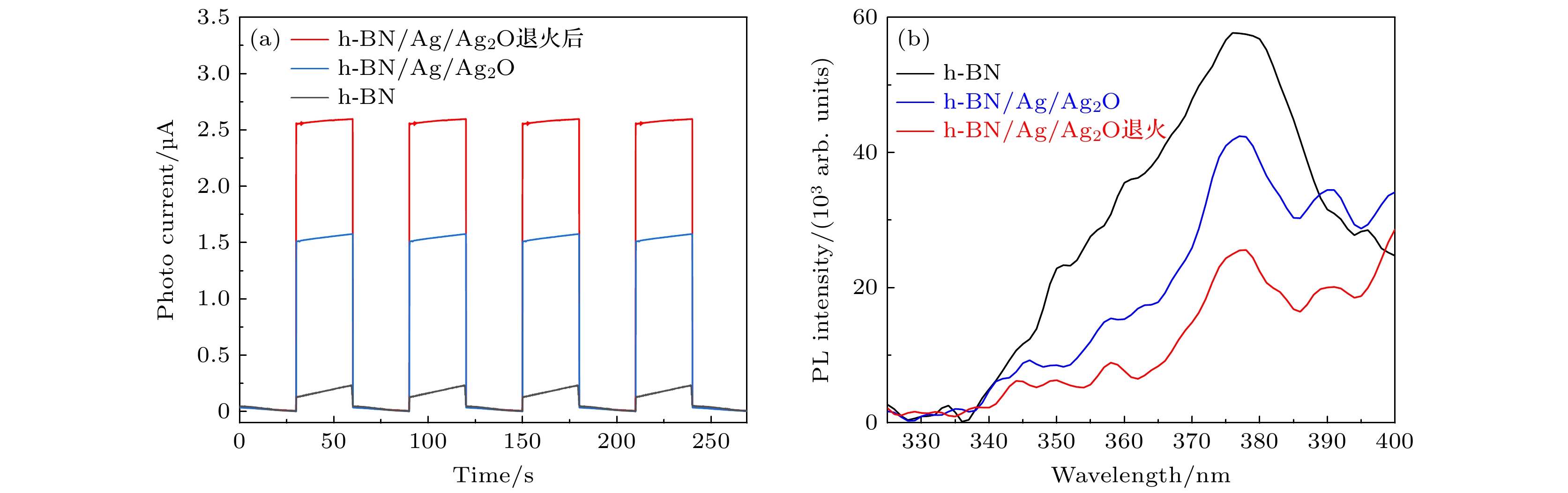
由等离激元金属和半导体结合形成的金属/半导体异质结构型有利于光诱导电荷转移(PICT)效率的提高, 在表面增强拉曼散射(SERS)研究中具有明显优势. 本文通过对所制备的杂化底物进行热退火处理进一步提高了其SERS活性. 首先, 在二维六方氮化硼(h-BN)纳米片表面成功负载生长了高密度和单分散的Ag/Ag2O纳米颗粒. 在此基础上, 通过进一步高温退火处理所得复合体系, 构建出高效的电荷转移通道, 从而大幅度提高了PICT效率, 使化学增强得到显著提高. 实验结果表明, 相比于退火前, 经320 ℃高温退火处理所得到的h-BN/Ag/Ag2O复合材料作为基底, 可以使结晶紫分子的SERS信号强度显著增强18倍, 增强因子高达1.63145 × 107. 最后, 基于h-BN/Ag/Ag2O 320 ℃退火复合材料优异的SERS性能, 实现了对食品添加剂专利蓝V的超灵敏SERS检测, 其检测极限低至10–12 M. 本文构建的h-BN/Ag/Ag2O 320 ℃退火复合材料兼具物理增强和化学增强, 在食品添加剂的痕量分析中具有显著优势.As a commonly used food additive, synthetic dyes are widely used in foods such as beverages, sweets and dairy products, and in the food processing process. Excessive use of synthetic dyes may induce cell mutations, children’s hyperactivity, genetic diseases and even cancer, which greatly threatens human health. As a fast, cheap, stable, ultra-sensitive and accurate detection method, SERS detection can reflect rich information about molecular fingerprint through the vibration of the analyte, and accurately determine the trace quantity of synthetic dyes in the actual environment. The metal/semiconductor heterostructure formed by the combination of plasmon metal and semiconductor is advantageous for the enhancement of photoinduced charge transfer (PICT) efficiency, and has obvious advantages in the study of surface-enhanced Raman scattering. In this work, the prepared hybrid substrate is annealed to further improve the surface-enhanced Raman scattering activity. Initially, high-density and monodisperse silver/silver oxide (Ag/Ag2O) nanoparticles are loaded onto two-dimensional hexagonal boron nitride (h-BN) nanosheets. On this basis, by annealing the system at high temperature, an efficient charge transfer channel is constructed, which greatly improves the PICT efficiency and chemical enhancement (CM). It is confirmed experimentally that the SERS signal intensity of h-BN/Ag/Ag2O material annealed at 320 ℃ with crystal violet (CV) as probe molecule is significantly increased 18 times compared with that of unannealed material, enhancement factor (EF) reaches 16.3145 ×107. Finally, based on the excellent SERS property of h-BN/Ag/Ag2O annealed at 320 ℃, the SERS detection of food additive Patent Blue V (PBV) is conducted in this work. The results show that the lowest detection concentration can reach 10–12 M, and the trace detection of synthetic dye PBV is realized. It has excellent spatial uniformity and anti-interference capability, which is of great significance in implementing the actual scene detection of PBV. The h-BN/Ag/Ag2O annealed at 320 ℃, the composite which is constructed in this paper, has both physical enhancement and chemical enhancement, and possess significant advantages in ultra-low sensitive SERS detection of food additives.
[1] Zhang Y, Hu L, Liu X, Liu B, Wu K 2015 Food Chem. 166 352
 Google Scholar
Google Scholar
[2] Nevado J J B, Flores J R, Llerena M J V, Fariñas N R 1999 Talanta 48 895
 Google Scholar
Google Scholar
[3] Akbarian Y, Shabani-Nooshabadi M, Karimi-Maleh H 2018 Sens. Actuators, B 273 228
 Google Scholar
Google Scholar
[4] Bijad M, Karimi-Maleh H, Farsi M, Shahidi S A 2017 J. Food Meas. Charact. 12 634
 Google Scholar
Google Scholar
[5] Darabi R, Shabani-Nooshabadi M 2021 Food Chem. 339 127841
 Google Scholar
Google Scholar
[6] Ntrallou K, Gika H, Tsochatzis E 2020 Foods 9 58
 Google Scholar
Google Scholar
[7] Tahernejad-Javazmi F, Shabani-Nooshabadi M, Karimi-Maleh H 2018 Talanta 176 208
 Google Scholar
Google Scholar
[8] Tahernejad-Javazmi F, Shabani-Nooshabadi M, Karimi-Maleh H 2019 Composites Part B 172 666
 Google Scholar
Google Scholar
[9] Zhang J, Wang X, Zhang S, Wang W, Hojo M, Chen Z 2014 J. Electrochem. Soc. 161 H453
 Google Scholar
Google Scholar
[10] Duman S, Erbas Z, Soylak M 2020 Microchem. J. 159 105468
 Google Scholar
Google Scholar
[11] EFSA Panel on Food Additives and Nutrient Sources added to Food (ANS) 2013 EFSA J. 11 2818
[12] Perez-Urquiza M, Beltran J L 2000 J. Chromatogr. A 898 271
 Google Scholar
Google Scholar
[13] Ozdemir Y, Akkan A A 1999 Turk. J. Chem. 23 221
[14] Unsal Y E, Soylak M, Tuzen M 2015 Environ. Monit. Assess. 187 203
 Google Scholar
Google Scholar
[15] Yoshioka N, Ichihashi K 2008 Talanta 74 1408
 Google Scholar
Google Scholar
[16] Liu E, Fan X, Yang Z, Han L, Li S, Huang Y, Liao K, Cai L 2022 Spectrochim. Acta, Part A 276 121229
 Google Scholar
Google Scholar
[17] Zhang Y, Yang Z, Zou Y, Farooq S, Li Y, Zhang H 2023 Food Chem. 412 135563
 Google Scholar
Google Scholar
[18] Wang C M, Roy P K, Juluri B K, Chattopadhyay S 2018 Sens. Actuators, B 261 218
 Google Scholar
Google Scholar
[19] Koberling F, Gryczynski Z K, Gregor I, Berer T, Klar T A, Jacak J, Buchegger B, Langer G 2019 Proc. SPIE 10884 18840U
 Google Scholar
Google Scholar
[20] Campion A, Kambhampati P 1998 Chem. Soc. Rev. 27 241
 Google Scholar
Google Scholar
[21] Schlucker S 2014 Angew. Chem. Int. Ed. 53 4756
 Google Scholar
Google Scholar
[22] Kanipe K N, Chidester P P, Stucky G D, Moskovits M 2016 ACS Nano 10 7566
 Google Scholar
Google Scholar
[23] Cho W J, Kim Y, Kim J K 2012 ACS Nano 6 249
 Google Scholar
Google Scholar
[24] Xie S, Chen D, Gu C, Jiang T, Zeng S, Wang Y Y, Ni Z, Shen X, Zhou J 2021 ACS Appl. Mater. Interfaces 13 33345
 Google Scholar
Google Scholar
[25] Wang Y, Zhang M, Yu H, Zuo Y, Gao J, He G, Sun Z 2019 Appl. Catal. B 252 174
 Google Scholar
Google Scholar
[26] Zhou L, Zhou J, Lai W, Yang X, Meng J, Su L, Gu C, Jiang T, Pun E Y B, Shao L, Petti L, Sun X W, Jia Z, Li Q, Han J, Mormile P 2020 Nat. Commun. 11 1785
 Google Scholar
Google Scholar
[27] Ling X, Xie L, Fang Y, Xu H, Zhang H, Kong J, Dresselhaus M S, Zhang J, Liu Z 2010 Nano Lett. 10 553
 Google Scholar
Google Scholar
[28] Jing X X, Li D Q, Zhang Y, Hou X Y, Jiang J, Fan X C, Wang M C, Feng S P, Yu Y F, Lu J P, Hu Z L, Ni Z H 2021 Chin. Phys. Lett. 38 074203
 Google Scholar
Google Scholar
[29] Jiao A, Cui Q, Li S, Tian Y, Ma H, Wang C, Zhang M, Chen M, Li G, Liu X 2022 Opt. Express 30 588
 Google Scholar
Google Scholar
[30] Yu D, Xu L, Zhang H, Li J, Wang W, Yang L, Jiang X, Zhao B 2023 Chin. Chem. Lett. 34 107771
 Google Scholar
Google Scholar
[31] Xue W, Han B, Ma H, Hou X, Wang J J, Li R, Zhang Y 2023 Chem. Eng. J. Adv. 13 100440
 Google Scholar
Google Scholar
[32] Ji S, Kou S, Wang M, Qiu H, Sun X, Dou J, Yang Z 2019 Appl. Surf. Sci. 489 1002
 Google Scholar
Google Scholar
[33] Xu L, Zhang H, Tian Y, Jiao A, Li S, Tan Y, Chen M, Chen F 2020 Talanta 209 120535
 Google Scholar
Google Scholar
[34] Zhang H, Li G, Li S, Xu L, Tian Y, Jiao A, Liu X, Chen F, Chen M 2018 Appl. Surf. Sci. 457 684
 Google Scholar
Google Scholar
[35] Peng Q, Ji W, De S 2012 Comput. Mater. Sci. 56 11
 Google Scholar
Google Scholar
[36] Cai Q R, Scullion D, Gan W, Falin A, Zhang S Y, Watanabe K, Taniguchi T, Chen Y, Santos E J G, Li L H 2019 Science Advances 5 eaav0129
 Google Scholar
Google Scholar
[37] Zhou H, Zhu J, Liu Z, Yan Z, Fan X, Lin J, Wang G, Yan Q, Yu T, Ajayan P M, Tour J M 2014 Nano Res. 7 1232
 Google Scholar
Google Scholar
[38] Cai Q R, Scullion D, Gan W, Falin A, Cizek P, Liu S, Edgar J H, Liu R, Cowie B C C, Santos E J G, Li L H 2020 Phys. Rev. Lett. 125 085902
 Google Scholar
Google Scholar
[39] Li L H, Xing T, Chen Y, Jones R 2014 Adv. Mater. Interfaces 1 1300132
 Google Scholar
Google Scholar
[40] Shen L, Zhao Y, Wang Y, Song R, Yao Q, Chen S, Chai Y 2016 J. Mater. Chem. A 4 5044
 Google Scholar
Google Scholar
[41] Cai Q, Du A, Gao G, et al. 2016 Adv. Funct. Mater. 26 8202
 Google Scholar
Google Scholar
[42] Li L H, Cervenka J, Watanabe K, Taniguchi T, Chen Y 2014 ACS Nano 8 1457
 Google Scholar
Google Scholar
[43] Dai P, Xue Y, Wang X, Weng Q, Zhang C, Jiang X, Tang D, Wang X, Kawamoto N, Ide Y, Mitome M, Golberg D, Bando Y 2015 Nanoscale 7 18992
 Google Scholar
Google Scholar
[44] Ling X, Fang W, Lee Y H, Araujo P T, Zhang X, Rodriguez-Nieva J F, Lin Y, Zhang J, Kong J, Dresselhaus M S 2014 Nano Lett. 14 3033
 Google Scholar
Google Scholar
[45] Blase X, Rubio A, Louie S G, Cohen M L 1995 Phys. Rev. B: Condens. Matter Mater. Phys. 51 6868
 Google Scholar
Google Scholar
[46] Han W Q, Wu L, Zhu Y, Watanabe K, Taniguchi T 2008 Appl. Phys. Lett. 93 223103
 Google Scholar
Google Scholar
[47] Cai Q, Gan W, Falin A, Watanabe K, Taniguchi T, Zhuang J, Hao W, Huang S, Tao T, Chen Y, Li L H 2020 ACS Appl. Mater. Interfaces 12 21985
 Google Scholar
Google Scholar
[48] Madasu M, Hsieh P L, Chen Y J, Huang M H 2019 ACS Appl. Mater. Interfaces 11 38039
 Google Scholar
Google Scholar
[49] Xu L, Cui Q, Tian Y, Jiao A, Zhang M, Li S, Li H, Chen M 2021 Appl. Surf. Sci. 545 149054
 Google Scholar
Google Scholar
[50] Tian Y, Cui Q, Ma H, Jiao A, Wang C, Zhang M, Zheng L, Wang X, Li S, Li G, Chen M 2022 Appl. Surf. Sci. 578 152100
 Google Scholar
Google Scholar
[51] Li S, Zhao J, Liu G, Xu L, Tian Y, Jiao A, Chen M 2021 Adv. Nanotechnol. 32 125401
 Google Scholar
Google Scholar
[52] Tian Y, Zhang H, Xu L, Chen M, Chen F 2018 Opt. Lett. 43 635
 Google Scholar
Google Scholar
[53] Tian Y, Li X, Wang F, Gu C, Zhao Z, Si H, Jiang T 2021 J. Hazard. Mater. 403 124009
 Google Scholar
Google Scholar
[54] Chen Y, Liu H, Li X, Tang S, Gu C, Wei G, Jiang T, Zhou X 2021 Sens. Actuators, B 339 129856
 Google Scholar
Google Scholar
[55] Li X, Liu H, Chen Y, Gu C, Wei G, Zhou J, Jiang T 2021 ACS Sustainable Chem. Eng. 9 12885
 Google Scholar
Google Scholar
[56] Lin S, Lin X, Han S, He L, Zhao H, Zhang J, Hasi W, Wang L 2019 J. Alloys Compd. 805 318
 Google Scholar
Google Scholar
[57] Mei R, Wang Y, Yu Q, Yin Y, Zhao R, Chen L 2020 ACS Appl. Mater. Interfaces 12 2059
 Google Scholar
Google Scholar
[58] Yang Y, Liu J, Fu Z W, Qin D 2014 J. Am. Chem. Soc. 136 8153
 Google Scholar
Google Scholar
[59] Gu H, Tang M, Qin L, Kang S Z, Li X 2022 Environ. Res. 204 112289
 Google Scholar
Google Scholar
[60] Cui X, Li J, Li Y, Liu M, Qiao J, Wang D, Cao H, He W, Feng Y, Yang Z 2022 Spectrochim. Acta, Part A 266 120432
 Google Scholar
Google Scholar
[61] Natan M J 2006 Faraday Discuss. 132 321
 Google Scholar
Google Scholar
[62] Wei W, Wang Y, Ji J, Zuo S, Li W, Bai F, Fan H 2018 Nano Lett. 18 4467
 Google Scholar
Google Scholar
-
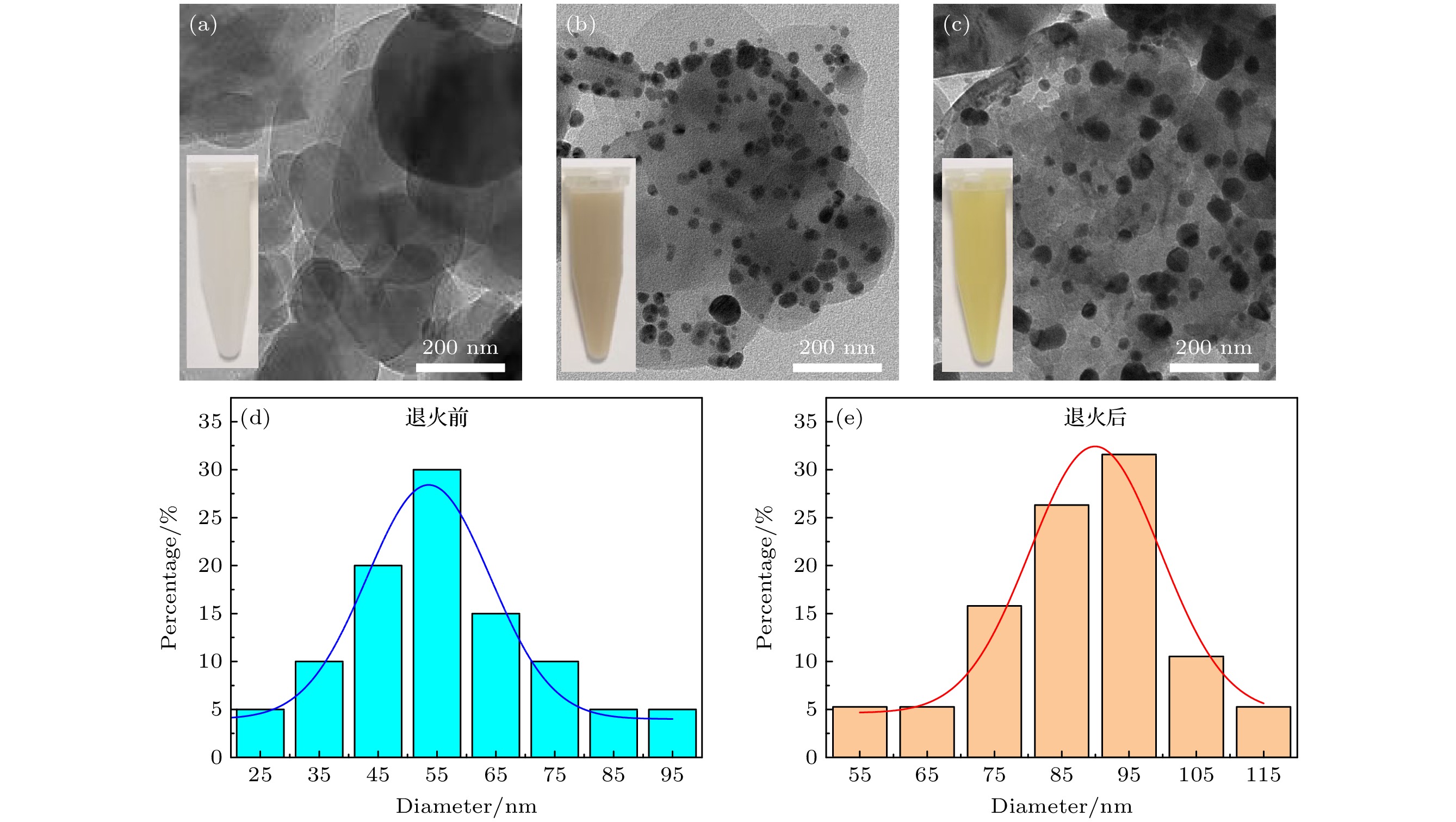
图 1 (a)—(c) 原始h-BN纳米片, h-BN/Ag/Ag2O, h-BN/Ag/Ag2O 320 ℃退火产物的TEM图像(插图为对应纳米材料的溶液); (d), (e) 负载在h-BN上的Ag/Ag2O纳米颗粒在320 ℃退火前和退火后的粒径分布图
Fig. 1. (a)–(c) TEM images of the original h-BN nanosheets, h-BN/Ag/Ag2O, and h-BN/Ag/Ag2O annealing products at 320 ℃ (Illustration is the solution of the corresponding nanomaterial); (d), (e) particle size distributions of Ag/Ag2O nanoparticles loaded on h-BN before and after annealing at 320 ℃.
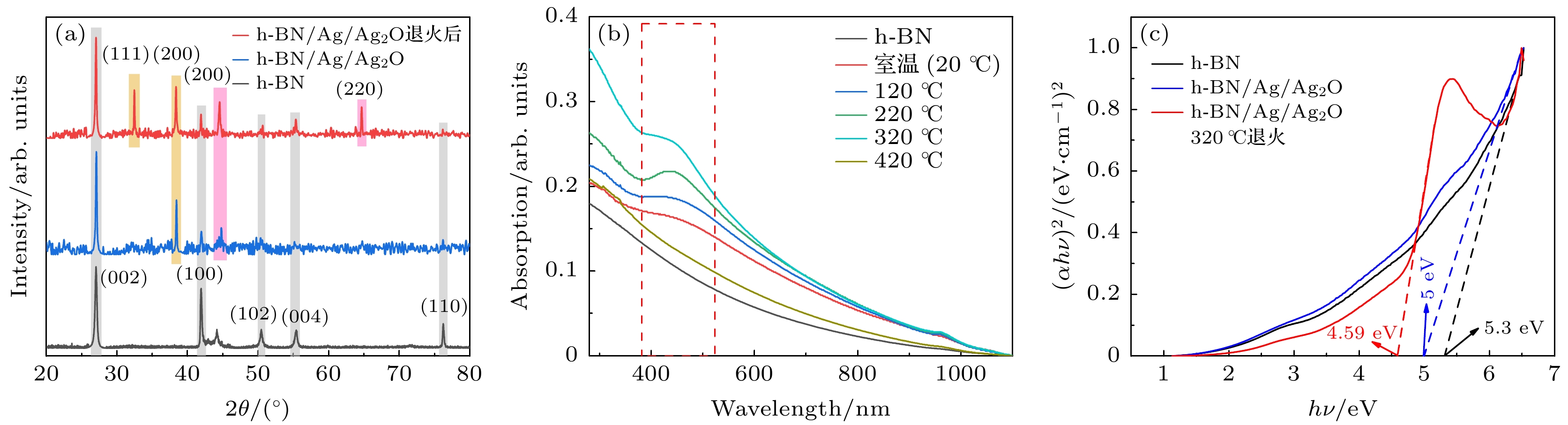
图 4 (a) 原始h-BN, h-BN/Ag/Ag2O和h-BN/Ag/Ag2O 320 ℃退火复合材料的XRD图谱; (b) h-BN/Ag/Ag2O在不同温度下退火所得产物的紫外-可见光-近红外吸收光谱; (c) h-BN, h-BN/Ag/Ag2O和h-BN/Ag/Ag2O 320 ℃退火复合材料的带隙
Fig. 4. (a) XRD spectrum of original h-BN nanosheets, h-BN/Ag/Ag2O and h-BN/Ag/Ag2O annealed 320 ℃; (b) ultraviolet-visible-near-infrared absorption spectra of materials annealed by h-BN/Ag/Ag2O at different temperatures; (c) band gaps of h-BN, h-BN/Ag/Ag2O and h-BN/Ag/Ag2O annealed at 320 ℃.
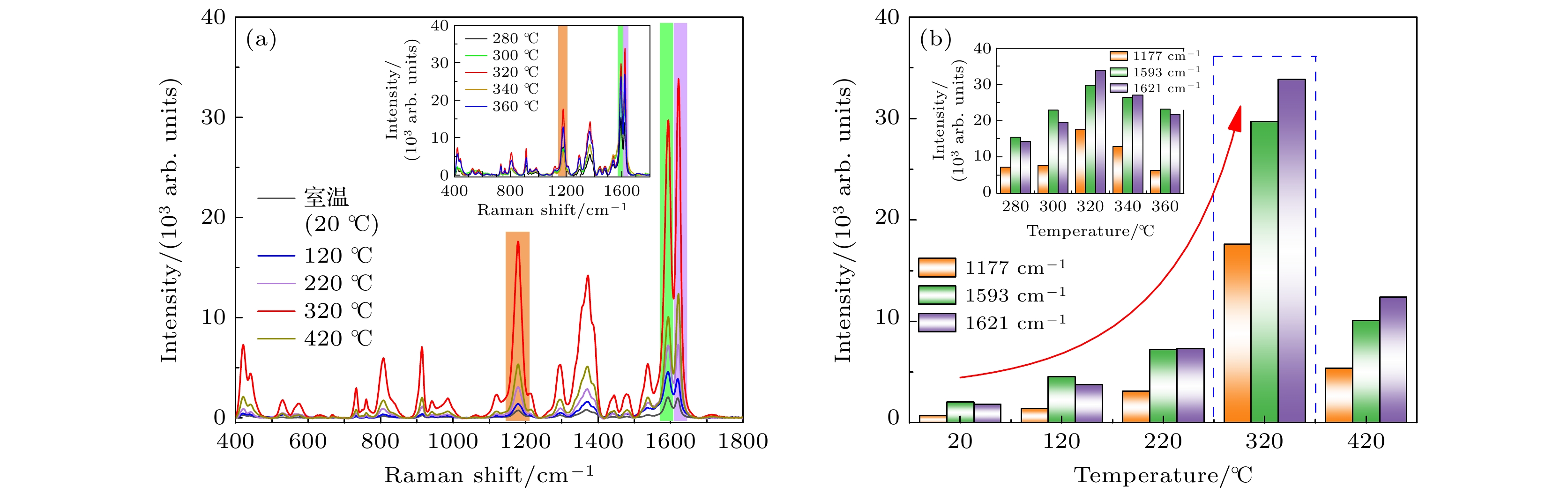
图 6 (a) 不同退火温度下获得的h-BN/Ag/Ag2O复合材料作为基底, 对CV (10–6 M)分子进行拉曼测试得到的SERS光谱, 插图为在320 ℃附近退火后得到的SERS光谱. (b) 不同退火温度的基底对应的CV分子的SERS光谱在1177, 1593, 1621 cm–1处特征峰的强度分布, 插图为在320 ℃附近退火后的基底所对应的特征峰强度分布
Fig. 6. (a) SERS spectrum of CV (10–6 M) molecules was obtained by Raman test on h-BN/Ag/Ag2O composites at different annealing temperatures, the inset shows the SERS spectrum obtained after annealing near 320 ℃. (b) Intensity distribution of characteristic peaks of CV molecules corresponding to substrates with different annealing temperatures at 1177, 1593 and 1621 cm–1, the inset shows the characteristic peak intensity distribution corresponding to the substrate after annealing near 320 ℃.
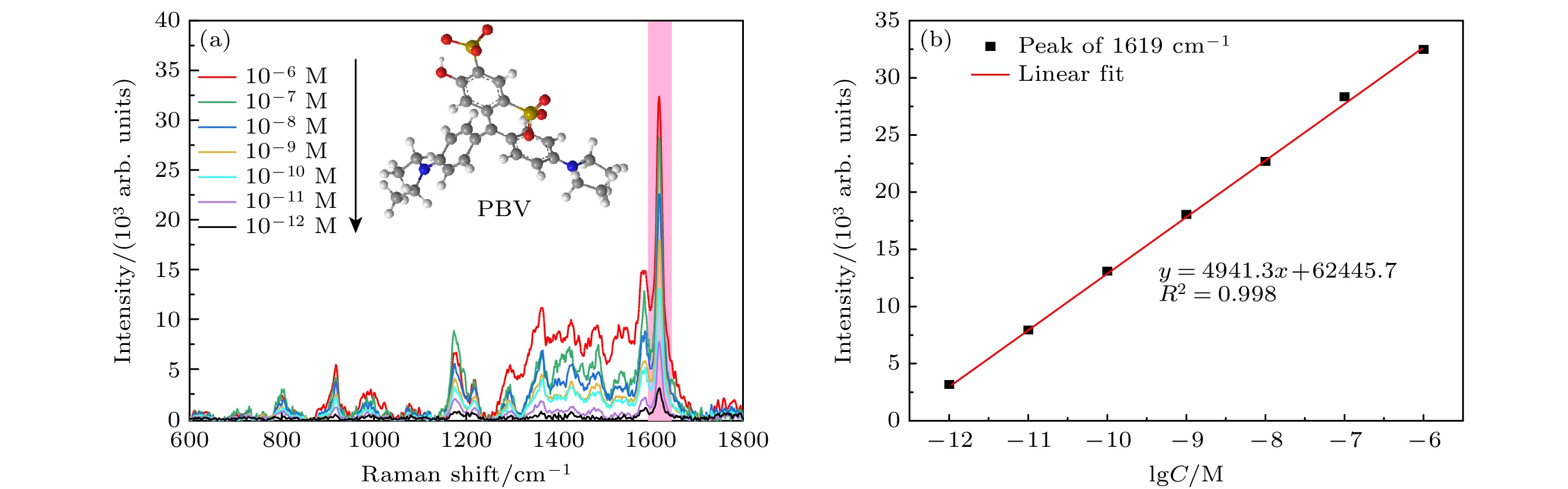
图 7 (a) 不同浓度的CV的SERS光谱; (b) CV分子在1621 cm–1处的SERS强度和分子浓度的对数之间的关系; (c) 不同浓度的R6G的SERS光谱; (d) R6G分子在1502 cm–1的SERS强度和分子浓度的对数之间的关系
Fig. 7. (a) SERS spectra of CV with different concentrations; (b) relationship between SERS intensity and logarithm of molecular concentration of CV molecule at 1621 cm–1; (c) SERS spectrum of R6G with different concentrations; (d) relationship between SERS intensity and logarithm of molecular concentration of R6G at 1502 cm–1.
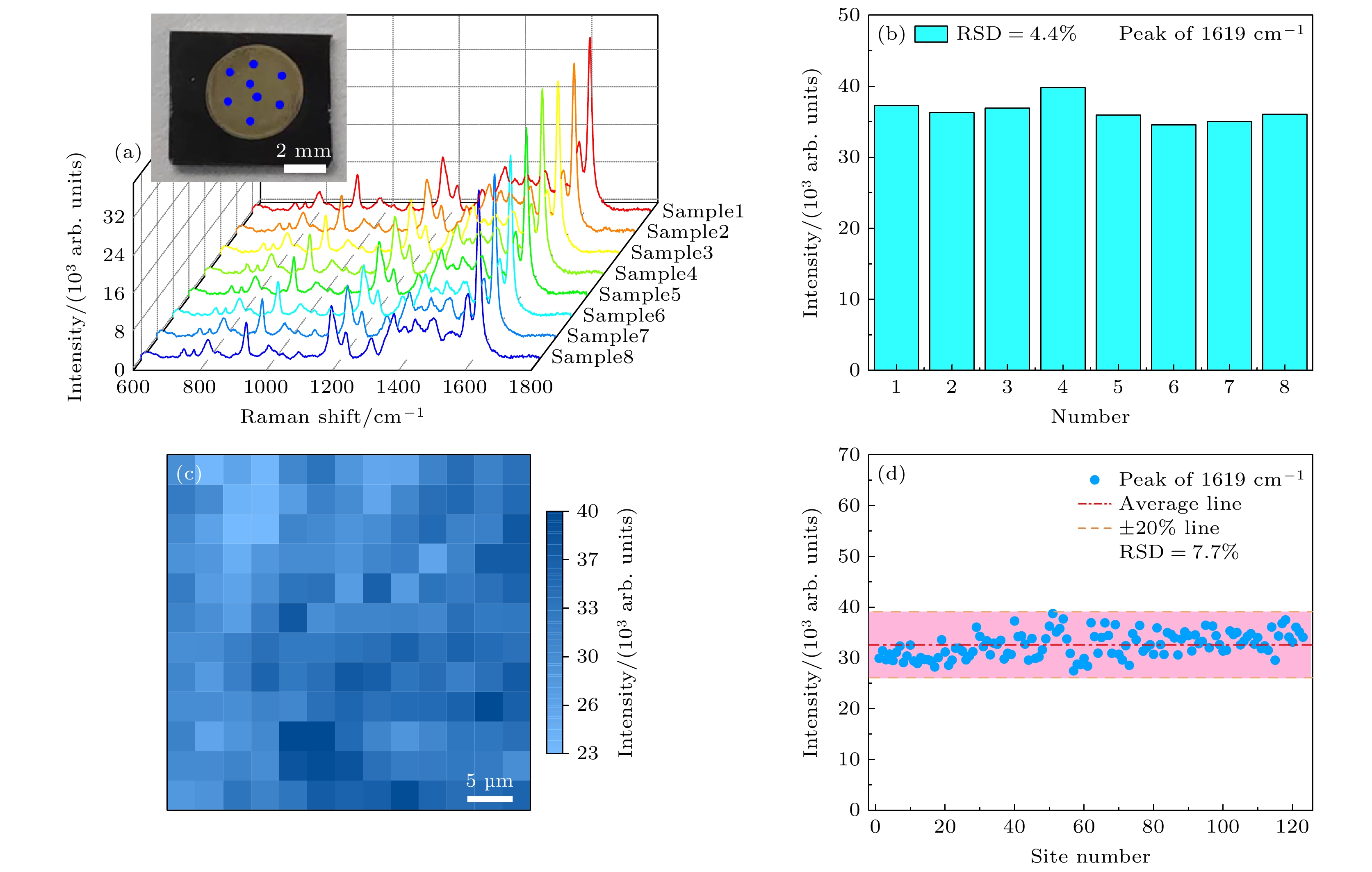
图 9 (a) 以PBV (10–6 M)为探针分子, 在h-BN/Ag/Ag2O 320 ℃退火复合材料上随机选取8个不同位置的点进行检测, 得到的SERS光谱; (b) PBV分子位于1619 cm–1处的拉曼特征峰强度的变化情况; (c) 在39 μm×39 μm的正方形区域内等距离取得169个点位得到SERS光谱, 峰位在1619 cm–1处的强度变化情况; (d) 拉曼信号峰位在1619 cm–1处强度的RSD
Fig. 9. (a) Using PBV (10–6 M) as probe molecule, the SERS spectrum was obtained by randomly selecting 8 points at different positions on the h-BN/Ag/Ag2O annealed composite at 320 ℃; (b) change of Raman characteristic peak intensity of PBV at 1619 cm–1; (c) 169 sites were obtained equidistant in a square area of 39 μm×39 μm to obtain SERS spectrum, with the intensity change at 1619 cm–1; (d) Raman signal peak at 1619 cm–1 intensity RSD.
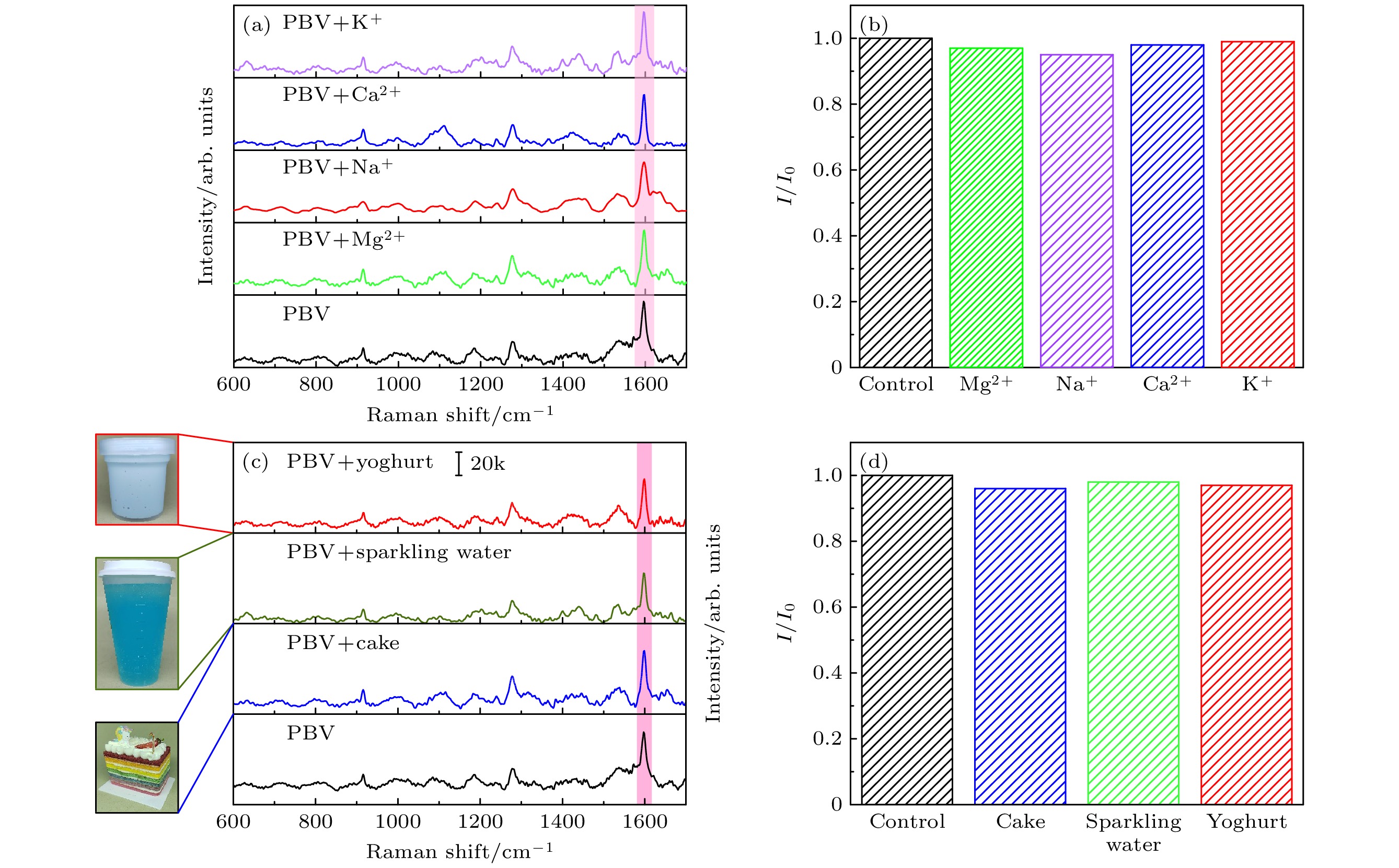
图 10 (a) 浓度为10–6 M的PBV通过单独添加K+, Ca2+, Na+和Mg2+离子(10–3 M)后的拉曼光谱; (b) 在离子干扰的情况下PBV分子在1619 cm–1处的拉曼峰强度的波动; (c) 浓度为10–6 M的PBV与酸奶、气泡水和彩色蛋糕提取液混合的拉曼光谱; (d) 在添加三种干扰物质后1619 cm–1 处拉曼峰强度的变化
Fig. 10. (a) Raman spectra of PBV with a concentration of 10–6 M by adding K+, Ca2+, Na+ and Mg2+ (10–3 M) alone; (b) the fluctuation of Raman peak intensity of PBV molecule at 1619 cm–1 in the presence of ion interference; (c) Raman spectra of PBV at a concentration of 10–6 M mixed with yogurt, sparkling water and colored cake extract; (d) change in Raman peak intensity at 1619 cm–1 after the addition of three interfering substances.
-
[1] Zhang Y, Hu L, Liu X, Liu B, Wu K 2015 Food Chem. 166 352
 Google Scholar
Google Scholar
[2] Nevado J J B, Flores J R, Llerena M J V, Fariñas N R 1999 Talanta 48 895
 Google Scholar
Google Scholar
[3] Akbarian Y, Shabani-Nooshabadi M, Karimi-Maleh H 2018 Sens. Actuators, B 273 228
 Google Scholar
Google Scholar
[4] Bijad M, Karimi-Maleh H, Farsi M, Shahidi S A 2017 J. Food Meas. Charact. 12 634
 Google Scholar
Google Scholar
[5] Darabi R, Shabani-Nooshabadi M 2021 Food Chem. 339 127841
 Google Scholar
Google Scholar
[6] Ntrallou K, Gika H, Tsochatzis E 2020 Foods 9 58
 Google Scholar
Google Scholar
[7] Tahernejad-Javazmi F, Shabani-Nooshabadi M, Karimi-Maleh H 2018 Talanta 176 208
 Google Scholar
Google Scholar
[8] Tahernejad-Javazmi F, Shabani-Nooshabadi M, Karimi-Maleh H 2019 Composites Part B 172 666
 Google Scholar
Google Scholar
[9] Zhang J, Wang X, Zhang S, Wang W, Hojo M, Chen Z 2014 J. Electrochem. Soc. 161 H453
 Google Scholar
Google Scholar
[10] Duman S, Erbas Z, Soylak M 2020 Microchem. J. 159 105468
 Google Scholar
Google Scholar
[11] EFSA Panel on Food Additives and Nutrient Sources added to Food (ANS) 2013 EFSA J. 11 2818
[12] Perez-Urquiza M, Beltran J L 2000 J. Chromatogr. A 898 271
 Google Scholar
Google Scholar
[13] Ozdemir Y, Akkan A A 1999 Turk. J. Chem. 23 221
[14] Unsal Y E, Soylak M, Tuzen M 2015 Environ. Monit. Assess. 187 203
 Google Scholar
Google Scholar
[15] Yoshioka N, Ichihashi K 2008 Talanta 74 1408
 Google Scholar
Google Scholar
[16] Liu E, Fan X, Yang Z, Han L, Li S, Huang Y, Liao K, Cai L 2022 Spectrochim. Acta, Part A 276 121229
 Google Scholar
Google Scholar
[17] Zhang Y, Yang Z, Zou Y, Farooq S, Li Y, Zhang H 2023 Food Chem. 412 135563
 Google Scholar
Google Scholar
[18] Wang C M, Roy P K, Juluri B K, Chattopadhyay S 2018 Sens. Actuators, B 261 218
 Google Scholar
Google Scholar
[19] Koberling F, Gryczynski Z K, Gregor I, Berer T, Klar T A, Jacak J, Buchegger B, Langer G 2019 Proc. SPIE 10884 18840U
 Google Scholar
Google Scholar
[20] Campion A, Kambhampati P 1998 Chem. Soc. Rev. 27 241
 Google Scholar
Google Scholar
[21] Schlucker S 2014 Angew. Chem. Int. Ed. 53 4756
 Google Scholar
Google Scholar
[22] Kanipe K N, Chidester P P, Stucky G D, Moskovits M 2016 ACS Nano 10 7566
 Google Scholar
Google Scholar
[23] Cho W J, Kim Y, Kim J K 2012 ACS Nano 6 249
 Google Scholar
Google Scholar
[24] Xie S, Chen D, Gu C, Jiang T, Zeng S, Wang Y Y, Ni Z, Shen X, Zhou J 2021 ACS Appl. Mater. Interfaces 13 33345
 Google Scholar
Google Scholar
[25] Wang Y, Zhang M, Yu H, Zuo Y, Gao J, He G, Sun Z 2019 Appl. Catal. B 252 174
 Google Scholar
Google Scholar
[26] Zhou L, Zhou J, Lai W, Yang X, Meng J, Su L, Gu C, Jiang T, Pun E Y B, Shao L, Petti L, Sun X W, Jia Z, Li Q, Han J, Mormile P 2020 Nat. Commun. 11 1785
 Google Scholar
Google Scholar
[27] Ling X, Xie L, Fang Y, Xu H, Zhang H, Kong J, Dresselhaus M S, Zhang J, Liu Z 2010 Nano Lett. 10 553
 Google Scholar
Google Scholar
[28] Jing X X, Li D Q, Zhang Y, Hou X Y, Jiang J, Fan X C, Wang M C, Feng S P, Yu Y F, Lu J P, Hu Z L, Ni Z H 2021 Chin. Phys. Lett. 38 074203
 Google Scholar
Google Scholar
[29] Jiao A, Cui Q, Li S, Tian Y, Ma H, Wang C, Zhang M, Chen M, Li G, Liu X 2022 Opt. Express 30 588
 Google Scholar
Google Scholar
[30] Yu D, Xu L, Zhang H, Li J, Wang W, Yang L, Jiang X, Zhao B 2023 Chin. Chem. Lett. 34 107771
 Google Scholar
Google Scholar
[31] Xue W, Han B, Ma H, Hou X, Wang J J, Li R, Zhang Y 2023 Chem. Eng. J. Adv. 13 100440
 Google Scholar
Google Scholar
[32] Ji S, Kou S, Wang M, Qiu H, Sun X, Dou J, Yang Z 2019 Appl. Surf. Sci. 489 1002
 Google Scholar
Google Scholar
[33] Xu L, Zhang H, Tian Y, Jiao A, Li S, Tan Y, Chen M, Chen F 2020 Talanta 209 120535
 Google Scholar
Google Scholar
[34] Zhang H, Li G, Li S, Xu L, Tian Y, Jiao A, Liu X, Chen F, Chen M 2018 Appl. Surf. Sci. 457 684
 Google Scholar
Google Scholar
[35] Peng Q, Ji W, De S 2012 Comput. Mater. Sci. 56 11
 Google Scholar
Google Scholar
[36] Cai Q R, Scullion D, Gan W, Falin A, Zhang S Y, Watanabe K, Taniguchi T, Chen Y, Santos E J G, Li L H 2019 Science Advances 5 eaav0129
 Google Scholar
Google Scholar
[37] Zhou H, Zhu J, Liu Z, Yan Z, Fan X, Lin J, Wang G, Yan Q, Yu T, Ajayan P M, Tour J M 2014 Nano Res. 7 1232
 Google Scholar
Google Scholar
[38] Cai Q R, Scullion D, Gan W, Falin A, Cizek P, Liu S, Edgar J H, Liu R, Cowie B C C, Santos E J G, Li L H 2020 Phys. Rev. Lett. 125 085902
 Google Scholar
Google Scholar
[39] Li L H, Xing T, Chen Y, Jones R 2014 Adv. Mater. Interfaces 1 1300132
 Google Scholar
Google Scholar
[40] Shen L, Zhao Y, Wang Y, Song R, Yao Q, Chen S, Chai Y 2016 J. Mater. Chem. A 4 5044
 Google Scholar
Google Scholar
[41] Cai Q, Du A, Gao G, et al. 2016 Adv. Funct. Mater. 26 8202
 Google Scholar
Google Scholar
[42] Li L H, Cervenka J, Watanabe K, Taniguchi T, Chen Y 2014 ACS Nano 8 1457
 Google Scholar
Google Scholar
[43] Dai P, Xue Y, Wang X, Weng Q, Zhang C, Jiang X, Tang D, Wang X, Kawamoto N, Ide Y, Mitome M, Golberg D, Bando Y 2015 Nanoscale 7 18992
 Google Scholar
Google Scholar
[44] Ling X, Fang W, Lee Y H, Araujo P T, Zhang X, Rodriguez-Nieva J F, Lin Y, Zhang J, Kong J, Dresselhaus M S 2014 Nano Lett. 14 3033
 Google Scholar
Google Scholar
[45] Blase X, Rubio A, Louie S G, Cohen M L 1995 Phys. Rev. B: Condens. Matter Mater. Phys. 51 6868
 Google Scholar
Google Scholar
[46] Han W Q, Wu L, Zhu Y, Watanabe K, Taniguchi T 2008 Appl. Phys. Lett. 93 223103
 Google Scholar
Google Scholar
[47] Cai Q, Gan W, Falin A, Watanabe K, Taniguchi T, Zhuang J, Hao W, Huang S, Tao T, Chen Y, Li L H 2020 ACS Appl. Mater. Interfaces 12 21985
 Google Scholar
Google Scholar
[48] Madasu M, Hsieh P L, Chen Y J, Huang M H 2019 ACS Appl. Mater. Interfaces 11 38039
 Google Scholar
Google Scholar
[49] Xu L, Cui Q, Tian Y, Jiao A, Zhang M, Li S, Li H, Chen M 2021 Appl. Surf. Sci. 545 149054
 Google Scholar
Google Scholar
[50] Tian Y, Cui Q, Ma H, Jiao A, Wang C, Zhang M, Zheng L, Wang X, Li S, Li G, Chen M 2022 Appl. Surf. Sci. 578 152100
 Google Scholar
Google Scholar
[51] Li S, Zhao J, Liu G, Xu L, Tian Y, Jiao A, Chen M 2021 Adv. Nanotechnol. 32 125401
 Google Scholar
Google Scholar
[52] Tian Y, Zhang H, Xu L, Chen M, Chen F 2018 Opt. Lett. 43 635
 Google Scholar
Google Scholar
[53] Tian Y, Li X, Wang F, Gu C, Zhao Z, Si H, Jiang T 2021 J. Hazard. Mater. 403 124009
 Google Scholar
Google Scholar
[54] Chen Y, Liu H, Li X, Tang S, Gu C, Wei G, Jiang T, Zhou X 2021 Sens. Actuators, B 339 129856
 Google Scholar
Google Scholar
[55] Li X, Liu H, Chen Y, Gu C, Wei G, Zhou J, Jiang T 2021 ACS Sustainable Chem. Eng. 9 12885
 Google Scholar
Google Scholar
[56] Lin S, Lin X, Han S, He L, Zhao H, Zhang J, Hasi W, Wang L 2019 J. Alloys Compd. 805 318
 Google Scholar
Google Scholar
[57] Mei R, Wang Y, Yu Q, Yin Y, Zhao R, Chen L 2020 ACS Appl. Mater. Interfaces 12 2059
 Google Scholar
Google Scholar
[58] Yang Y, Liu J, Fu Z W, Qin D 2014 J. Am. Chem. Soc. 136 8153
 Google Scholar
Google Scholar
[59] Gu H, Tang M, Qin L, Kang S Z, Li X 2022 Environ. Res. 204 112289
 Google Scholar
Google Scholar
[60] Cui X, Li J, Li Y, Liu M, Qiao J, Wang D, Cao H, He W, Feng Y, Yang Z 2022 Spectrochim. Acta, Part A 266 120432
 Google Scholar
Google Scholar
[61] Natan M J 2006 Faraday Discuss. 132 321
 Google Scholar
Google Scholar
[62] Wei W, Wang Y, Ji J, Zuo S, Li W, Bai F, Fan H 2018 Nano Lett. 18 4467
 Google Scholar
Google Scholar
计量
- 文章访问数: 5661
- PDF下载量: 92
- 被引次数: 0













 下载:
下载: