-
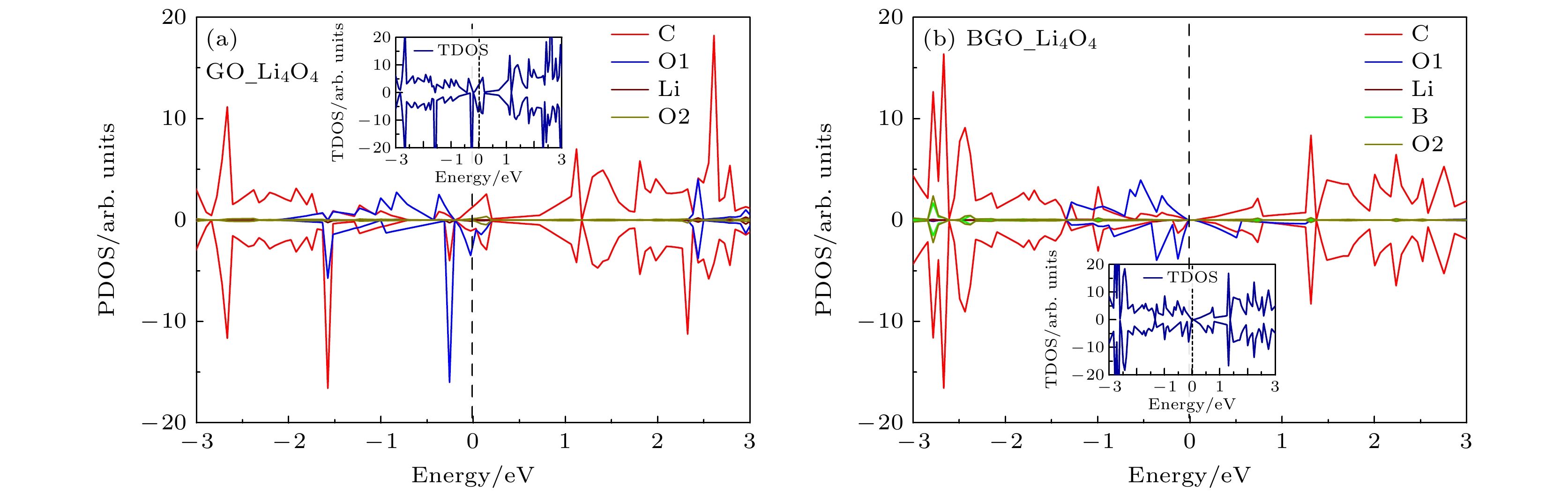
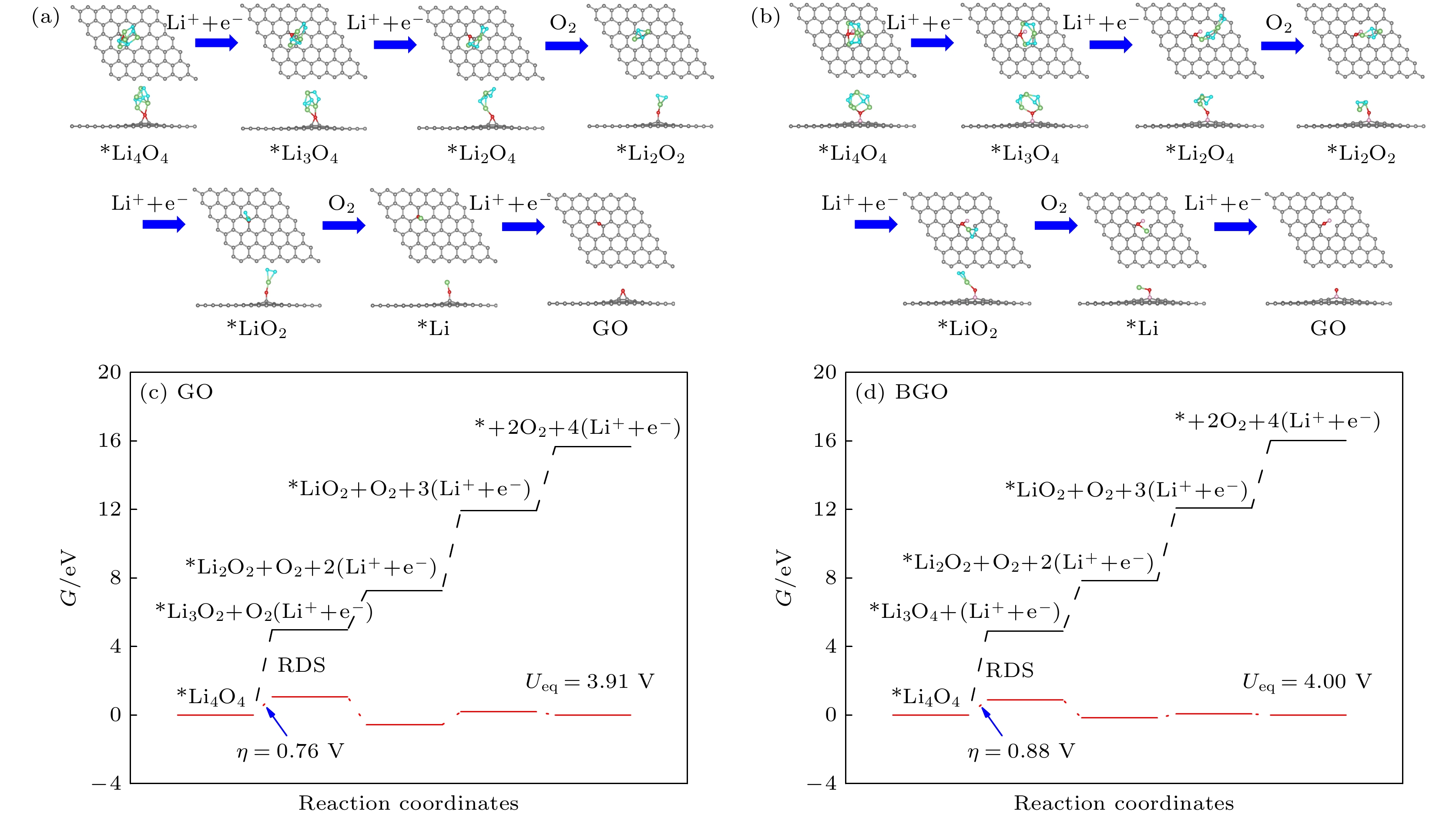
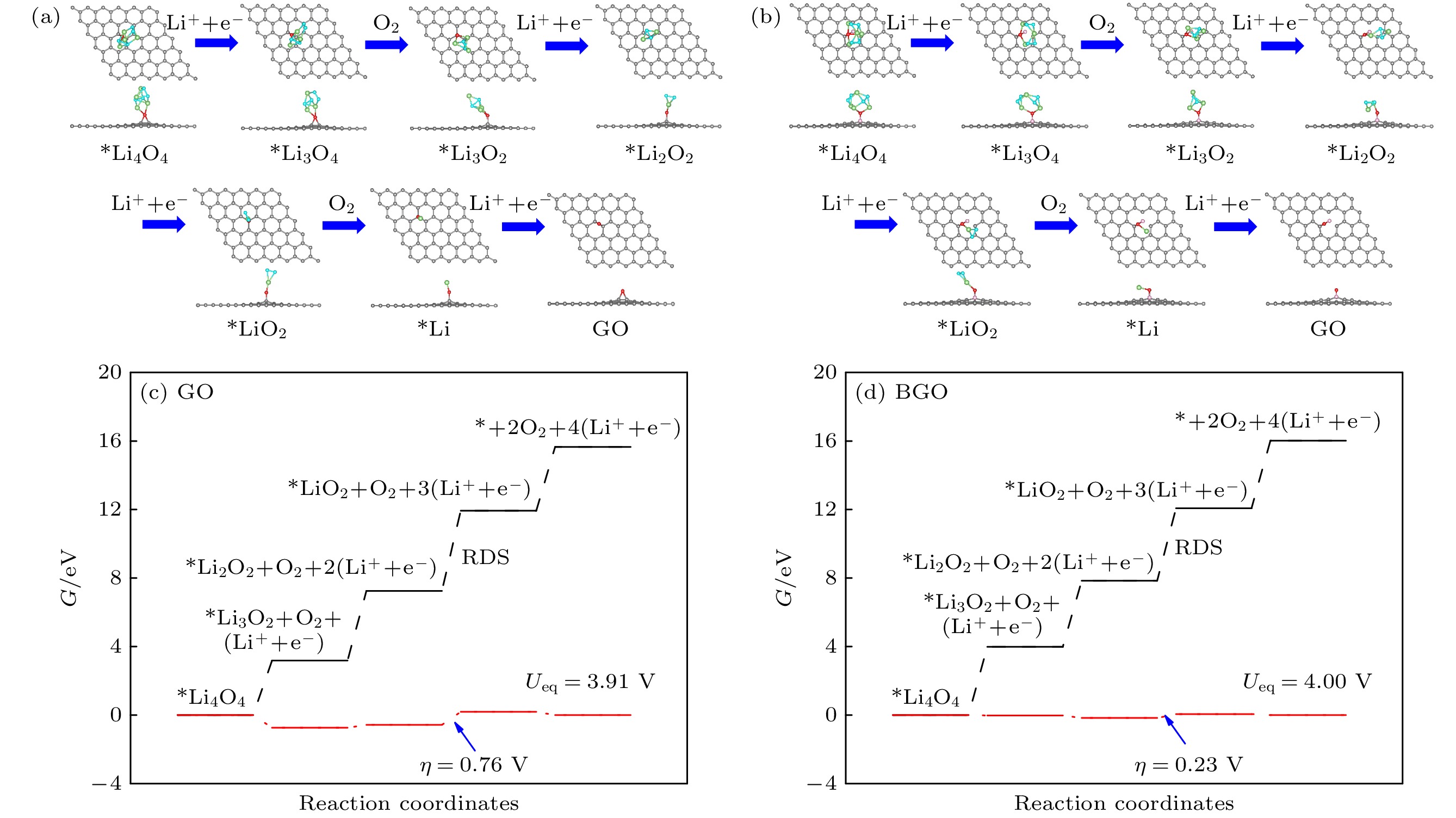
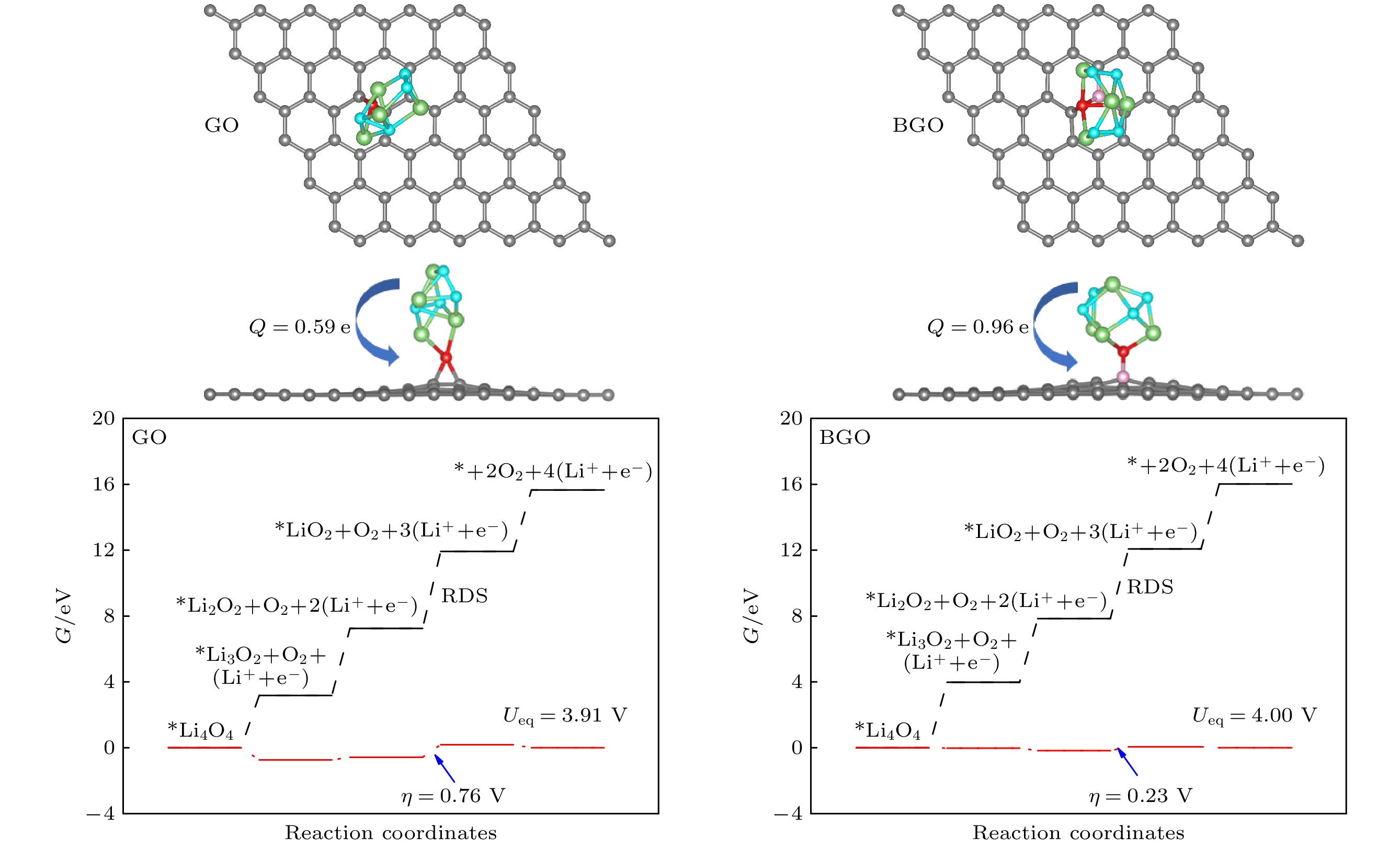
锂-氧电池由于高能量密度在后锂离子电池中脱颖而出, 而放电产物过氧化锂缓慢的氧化反应降低了电池的循环性能. 因此, 提高过氧化锂氧化反应动能、降低充电过电位对于实现高能量密度的可逆锂-氧电池具有重要意义. 本文通过第一性原理计算, 对比研究了氧化石墨烯(GO)和硼掺杂氧化石墨烯(BGO)对过氧化锂小团簇(Li2O2)2氧化反应的催化机理. 结果表明, 从(Li2O2)2团簇转移到GO和BGO上的电荷分别为0.59 e和0.96 e, B掺杂提高了电荷转移. 4电子反应过程表明, (Li2O2)2团簇倾向于Li-O2-Li分解路径, 在GO和BGO上反应的速率决定步均是第三步去锂. 在平衡电位下, GO和BGO的充电过电位分别是0.76 V和0.23 V, B掺杂大大降低了锂-氧电池充电过电位. 机理分析表明B与O对(Li2O2)2团簇起到了协同催化的作用.Lithium-oxygen batteries stand out among post-lithium-ion batteries due to their theoretically high energy density, while the sluggish reaction kinetics of lithium peroxide reduces the rate performance of the batteries. Therefore, improving the reaction kinetics of the lithium peroxide and then lowering the charge overpotential are of great importance for realizing reversible lithium-oxygen batteries with high energy density. In this work, the catalytic mechanism of graphene oxide (GO) and boron-doped graphene oxide (BGO) on the oxygen evolution reaction of (Li2O2)2 cluster is investigated by first-principles calculations. The results show that the charge transfer from (Li2O2)2 cluster to GO and from (Li2O2)2 cluster to BGO are 0.59 e and 0.96 e, respectively, suggests that B doping improves the charge transfer from the discharged product to the cathode material. The Gibbs free energy of the 4-electron decomposition process shows that the (Li2O2)2 cluster favors the Li-O2-Li decomposition pathway, and the rate-determining step for the reaction on both GO and BGO is the third step, that is, the removal of the third lithium. At the equilibrium potential, the charge overpotential of GO and BGO are 0.76 V and 0.23 V, respectively, showing that B doping greatly reduces the charging overpotential of lithium-oxygen batteries. Moreover, mechanistic analysis shows that B doping enhances the electronic conductance of GO and forms an electron-deficient active center, which facilitates charge transport in cathode and charge transfer from lithium peroxide to cathode materials, thereby reducing the charging overpotential of the lithium-oxygen batteries and improving its cycling performance. The B and O play a synergistic role in catalyzing the oxygen evolution reaction of (Li2O2)2 clusters.
-
Keywords:
- lithium-oxygen batteries /
- oxygen evolution reaction /
- B-doped graphene oxide /
- first-principles
[1] Liu T, Zhao S Y, Xiong Q, Yu J, Wang J, Huang G, Ni M, Zhang X B 2023 Adv. Mater. 35 2208925
 Google Scholar
Google Scholar
[2] Hou X Y, Rong X H, Lu Y X, Hu Y S 2022 Chin. Phys. B 31 098801
 Google Scholar
Google Scholar
[3] Guo Q, Han S, Lu Y X, Chen L, Hu Y S 2023 Chin. Phys. Lett. 40 028801
 Google Scholar
Google Scholar
[4] 丁飞翔, 容晓晖, 王海波, 杨佯, 胡紫霖, 党荣彬, 陆雅翔, 胡勇胜 2022 物理学报 71 108801
 Google Scholar
Google Scholar
Ding F X, Rong X H, Wang H B, Yang Y, Hu Z L, Dang R B, Lu Y X, Hu Y S 2022 Acta Phys. Sin. 71 108801
 Google Scholar
Google Scholar
[5] Zhao W Y, Xu L C, Guo Y H, Yang Z, Liu R P, Li X Y 2022 Chin. Phys. B 31 047101
 Google Scholar
Google Scholar
[6] Aurbach D, Mccloskey B D, Nazar L F, Bruce P G 2016 Nat. Energy 1 16128
 Google Scholar
Google Scholar
[7] Zhang X P, Mu X W, Yang S X, Wang P F, Guo S H, Han M, He P, Zhou H S 2018 Energy Environ. Mater. 1 61
 Google Scholar
Google Scholar
[8] Zhang T, Wu N, Zhao Y, Zhang X, Wu J, Weng J, Li S, Huo F W, Huang W 2022 Adv. Sci. 9 e2103954
 Google Scholar
Google Scholar
[9] Wu Z Z, Tian Y H, Chen H, Wang L G, Qian S S, Wu T P, Zhang S Q, Lu J 2022 Chem. Soc. Rev. 51 8045
 Google Scholar
Google Scholar
[10] Kwak W J, Rosy, Sharon D, Xia C, Kim H, Johnson L R, Bruce P G, Nazar L F, Sun Y K, Frimer A A, Noked M, Freunberger S A, Aurbach D 2020 Chem. Rev. 120 6626
 Google Scholar
Google Scholar
[11] Chen K, Yang D Y, Huang G, Zhang X B 2021 Acc. Chem. Res. 54 632
 Google Scholar
Google Scholar
[12] Peng Z, Freunberger S A, Chen Y, Bruce P G 2012 Science 337 563
 Google Scholar
Google Scholar
[13] Lyu Z Y, Zhou Y, Dai W R, Cui X H, Lai M, Wang L, Huo F W, Huang W, Hu Z, Chen W 2017 Chem. Soc. Rev. 46 6046
 Google Scholar
Google Scholar
[14] Kang J H, Lee J, Jung J W, Park J, Jang T, Kim H S, Nam J S, Lim H, Yoon K R, Ryu W H, Kim I D, Byon H R 2020 ACS Nano 14 14549
 Google Scholar
Google Scholar
[15] Hummelshoj J S, Blomqvist J, Datta S, Vegge T, Rossmeisl J, Thygesen K S, Luntz A C, Jacobsen K W, Norskov J K 2010 J. Chem. Phys. 132 071101
 Google Scholar
Google Scholar
[16] Kang J, Jung Y S, Wei S H, Dillon A C 2012 Phys. Rev. B 85 035210
 Google Scholar
Google Scholar
[17] Assary R S, Lau K C, Amine K, Sun Y K, Curtiss L A 2013 J. Phys. Chem. C 117 8041
 Google Scholar
Google Scholar
[18] Mccloskey B D, Scheffler R, Speidel A, Bethune D S, Shelby R M, Luntz A C 2011 J. Am. Chem. Soc. 133 18038
 Google Scholar
Google Scholar
[19] Freunberger S A, Chen Y, Peng Z, Griffin J M, Hardwick L J, Barde F, Novak P, Bruce P G 2011 J. Am. Chem. Soc. 133 8040
 Google Scholar
Google Scholar
[20] Mo Y, Ong S P, Ceder G 2011 Phys. Rev. B 84 205446
 Google Scholar
Google Scholar
[21] Debart A, Paterson A J, Bao J, Bruce P G 2008 Angew. Chem. Int. Ed. Engl. 47 4521
 Google Scholar
Google Scholar
[22] Yu Y, Zhang B, He Y B, Huang Z D, Oh S W, Kim J K 2013 J. Mater. Chem. A 1 1163
 Google Scholar
Google Scholar
[23] Debart A, Bao J, Armstrong G, Bruce P G 2007 J. Power Sources 174 1177
 Google Scholar
Google Scholar
[24] Black R, Lee J H, Adams B, Mims C A, Nazar L F 2013 Angew. Chem. Int. Ed. Engl. 52 392
 Google Scholar
Google Scholar
[25] Cui Y, Wen Z, Liu Y 2011 Energy Environ. Sci. 4 4727
 Google Scholar
Google Scholar
[26] Zhu J, Ren X, Liu J, Zhang W, Wen Z 2015 Acs Catalysis 5 73
 Google Scholar
Google Scholar
[27] Lim H D, Song H, Gwon H, Park K Y, Kim J, Bae Y, Kim H, Jung S K, Kim T, Kim Y H, Lepro X, Ovalle Robles R, Baughman R H, Kang K 2013 Energy Environ. Sci. 6 3570
 Google Scholar
Google Scholar
[28] Lu Y C, Xu Z, Gasteiger H A, Chen S, Hamad Schifferli K, Shao Horn Y 2010 J. Am. Chem. Soc. 132 12170
 Google Scholar
Google Scholar
[29] Li F J, Tang D M, Chen Y, Golberg D, Kitaura H, Zhang T, Yamada A, Zhou H S 2013 Nano Lett. 13 4702
 Google Scholar
Google Scholar
[30] Jung H G, Jeong Y S, Park J B, Sun Y K, Scrosati B, Lee Y J 2013 ACS Nano 7 3532
 Google Scholar
Google Scholar
[31] Cai F, Lei X L 2023 Appl. Surf. Sci. 609 155331
 Google Scholar
Google Scholar
[32] Cai F, Lei X L, Ke Q, Ouyang C Y 2023 J. Phys. Chem. C 127 14232
 Google Scholar
Google Scholar
[33] Sun B, Huang X D, Chen S Q, Munroe P, Wang G X 2014 Nano Lett. 14 3145
 Google Scholar
Google Scholar
[34] Yoo E, Zhou H S 2011 ACS Nano 5 3020
 Google Scholar
Google Scholar
[35] Zhang Y, Ge J, Wang L, Wang D, Ding F, Tao X, Chen W 2013 Sci. Rep. 3 2771
 Google Scholar
Google Scholar
[36] 侯滨朋, 淦作亮, 雷雪玲, 钟淑英, 徐波, 欧阳楚英 2019 物理学报 68 128801
 Google Scholar
Google Scholar
Hou B P, Gan Z L, Lei X L, Zhong S Y, Xu B, Ouyang C Y 2019 Acta Phys. Sin. 68 128801
 Google Scholar
Google Scholar
[37] Tang Y B, Yin L C, Yang Y, Bo X H, Cao Y L, Wang H E, Zhang W J, Bello I, Lee S T, Cheng H M, Lee C S 2012 ACS Nano 6 1970
 Google Scholar
Google Scholar
[38] Wang H, Zhou Y, Wu D, Liao L, Zhao S L, Peng H L, Liu Z F 2013 Small 9 1316
 Google Scholar
Google Scholar
[39] Ren X D, Zhu J Z, Du F, Liu J J, Zhang W Q 2014 J. Phys. Chem. C 118 22412
 Google Scholar
Google Scholar
[40] Ren X D, Wang B Z, Zhu J Z, Liu J J, Zhang W Q, Wen Z Y 2015 Phys. Chem. Chem. Phys. 17 14605
 Google Scholar
Google Scholar
[41] Hou B P, Lei X L, Zhong S Y, Sun B Z, Ouyang C Y 2020 Phys. Chem. Chem. Phys. 22 14216
 Google Scholar
Google Scholar
[42] Salehi M, Shariatinia Z, Sadeghi A 2019 J. Electroanal. Chem. 832 165
 Google Scholar
Google Scholar
[43] Kresse G, Furthmüller J 1996 Phys. Rev. B 54 11169
 Google Scholar
Google Scholar
[44] Blöchl P E 1994 Phys. Rev. B 50 17953
 Google Scholar
Google Scholar
[45] Perdew J P, Burke K, Ernzerhof M 1996 Phys. Rev. Lett. 77 3865
 Google Scholar
Google Scholar
[46] Monkhorst H J, Pack J D 1976 Phys. Rev. B 13 5188
 Google Scholar
Google Scholar
[47] Grimme S, Antony J, Ehrlich S, Krieg H 2010 J. Chem. Phys. 132 154104
 Google Scholar
Google Scholar
[48] Topsakal M, Ciraci S 2012 Phys. Rev. B 86 205402
 Google Scholar
Google Scholar
-
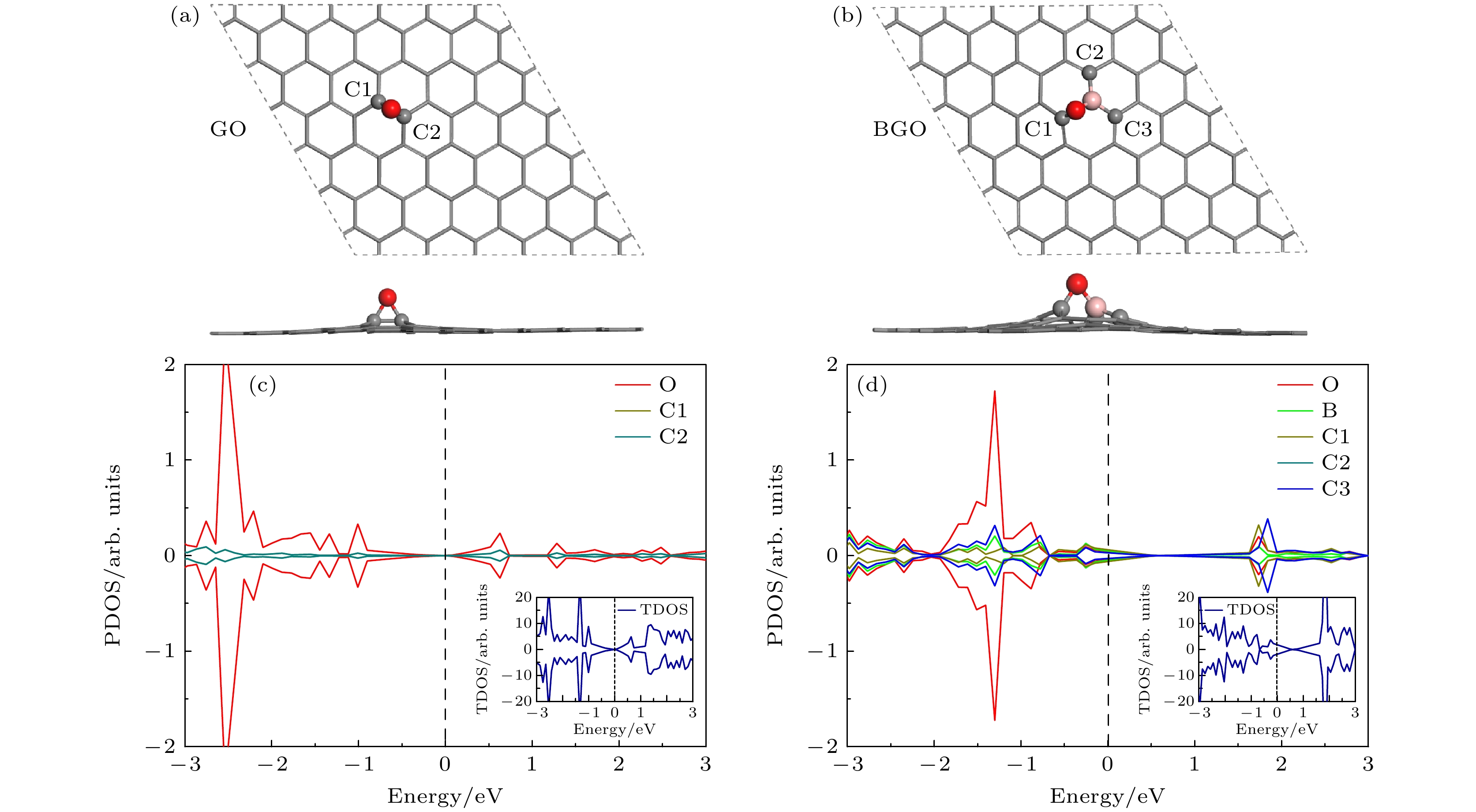
图 1 氧化石墨烯和硼掺杂氧化石墨烯的几何结构和电子性质 (a), (b)几何结构, 灰色、红色和粉色小球分别代表C, O和B原子; (c), (d)电子性质, C1, C2和C3与结构图中相一致, 图中插入的是总态密度
Fig. 1. Geometric structure and electronic properties of oxide graphene (GO) and B doped oxide graphene (BGO): (a), (b) Geometric structures, grey, red and pink balls represent the C, O and B atoms, respectively; (c), (d) electronic properties, C1, C2 and C3 are consistent with those in the geometric structures, the insert is total density of states (TDOS).
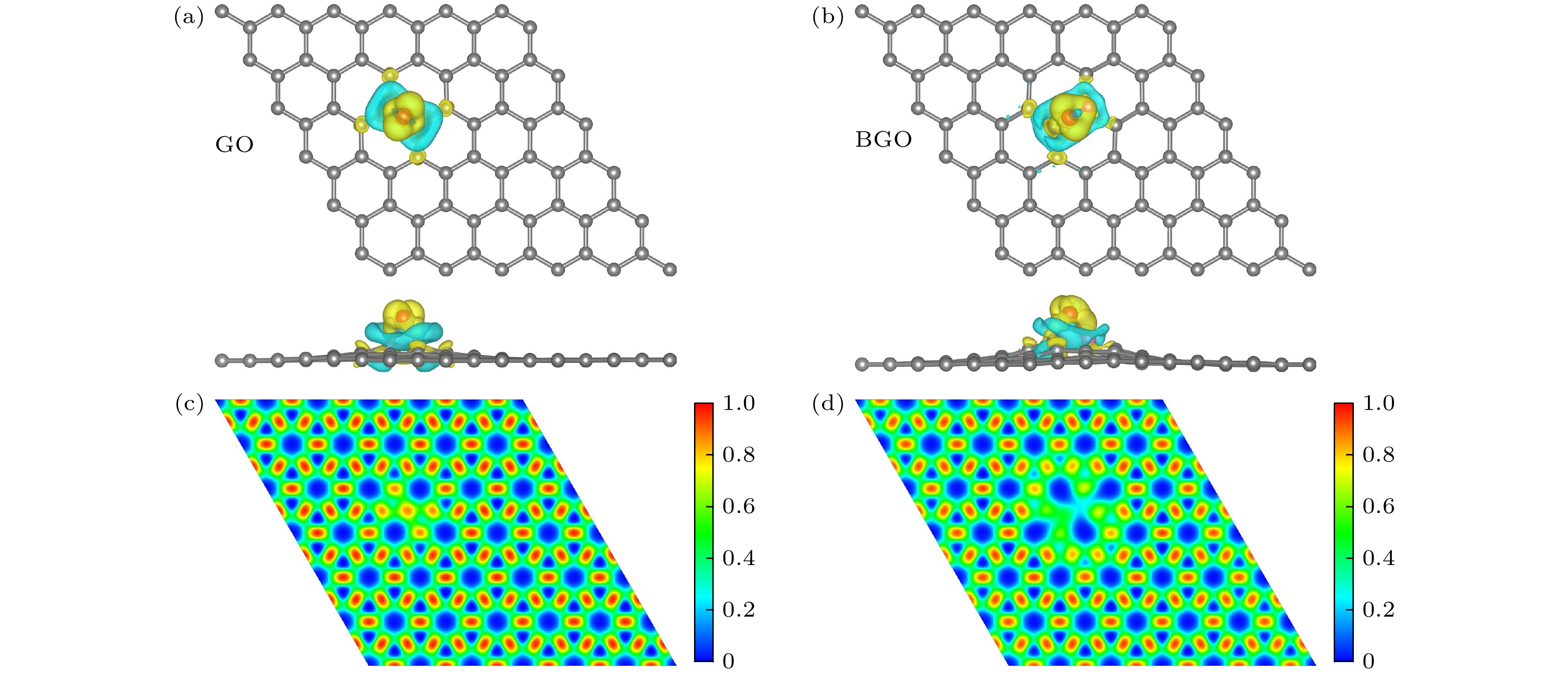
图 2 氧化石墨烯和硼掺杂氧化石墨烯的电荷密度差分和电子局域密度 (a), (b)电荷密度差分, 黄色和蓝色区域分别表示电荷聚积和电荷消失, 电荷等值面为0.004 e/Å3; (c), (d) 电子局域密度, 从红色到蓝色表示电子由多到少
Fig. 2. Charge density difference and electron localization function (ELF) of GO and BGO: (a), (b) Charge density difference, yellow and blue indicate the gain and the loss of electrons, and the isosurface value is 0.004 e/Å3; (c), (d) electron localization function, red to blue indicates more to less electrons.
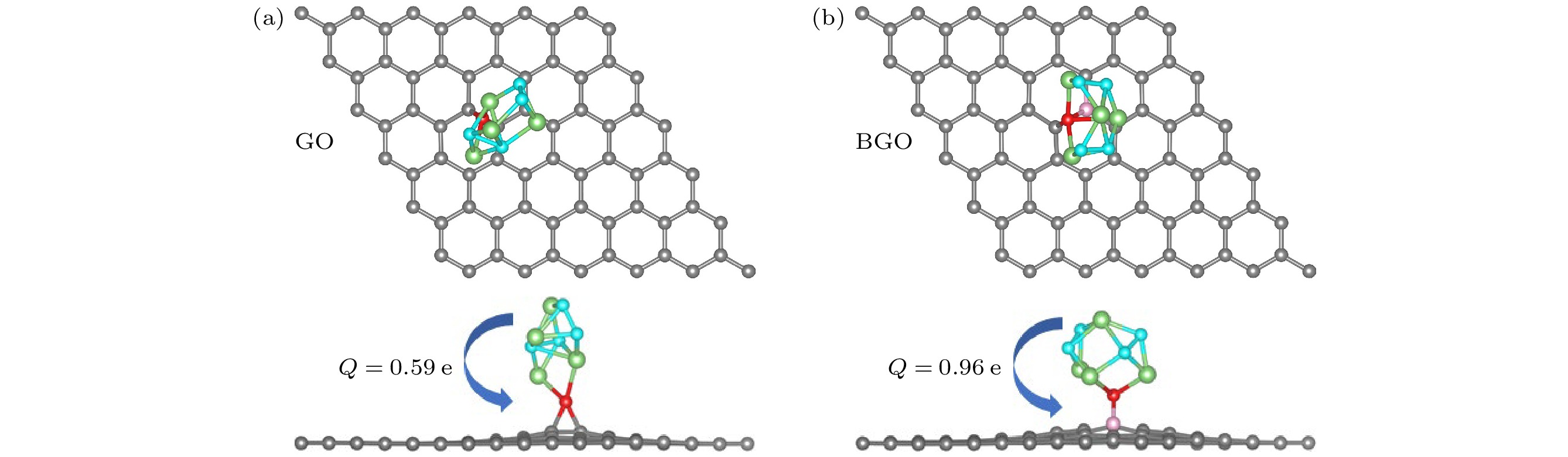
图 3 (Li2O2)2团簇在氧化石墨烯(a)和硼掺杂氧化石墨烯(b)上吸附的俯视图和侧视图以及电荷转移, 箭头表示电荷转移方向, 青色和绿色分别代表(Li2O2)2团簇的O原子和Li原子
Fig. 3. Top view and side view of (Li2O2)2 cluster adsorbed on the GO (a) and BGO catalysts (b) along with the charge transfer, arrows indicate the direction of charge transfer. The cyan and green represent the O and Li atoms in the (Li2O2)2 cluster, respectively.
-
[1] Liu T, Zhao S Y, Xiong Q, Yu J, Wang J, Huang G, Ni M, Zhang X B 2023 Adv. Mater. 35 2208925
 Google Scholar
Google Scholar
[2] Hou X Y, Rong X H, Lu Y X, Hu Y S 2022 Chin. Phys. B 31 098801
 Google Scholar
Google Scholar
[3] Guo Q, Han S, Lu Y X, Chen L, Hu Y S 2023 Chin. Phys. Lett. 40 028801
 Google Scholar
Google Scholar
[4] 丁飞翔, 容晓晖, 王海波, 杨佯, 胡紫霖, 党荣彬, 陆雅翔, 胡勇胜 2022 物理学报 71 108801
 Google Scholar
Google Scholar
Ding F X, Rong X H, Wang H B, Yang Y, Hu Z L, Dang R B, Lu Y X, Hu Y S 2022 Acta Phys. Sin. 71 108801
 Google Scholar
Google Scholar
[5] Zhao W Y, Xu L C, Guo Y H, Yang Z, Liu R P, Li X Y 2022 Chin. Phys. B 31 047101
 Google Scholar
Google Scholar
[6] Aurbach D, Mccloskey B D, Nazar L F, Bruce P G 2016 Nat. Energy 1 16128
 Google Scholar
Google Scholar
[7] Zhang X P, Mu X W, Yang S X, Wang P F, Guo S H, Han M, He P, Zhou H S 2018 Energy Environ. Mater. 1 61
 Google Scholar
Google Scholar
[8] Zhang T, Wu N, Zhao Y, Zhang X, Wu J, Weng J, Li S, Huo F W, Huang W 2022 Adv. Sci. 9 e2103954
 Google Scholar
Google Scholar
[9] Wu Z Z, Tian Y H, Chen H, Wang L G, Qian S S, Wu T P, Zhang S Q, Lu J 2022 Chem. Soc. Rev. 51 8045
 Google Scholar
Google Scholar
[10] Kwak W J, Rosy, Sharon D, Xia C, Kim H, Johnson L R, Bruce P G, Nazar L F, Sun Y K, Frimer A A, Noked M, Freunberger S A, Aurbach D 2020 Chem. Rev. 120 6626
 Google Scholar
Google Scholar
[11] Chen K, Yang D Y, Huang G, Zhang X B 2021 Acc. Chem. Res. 54 632
 Google Scholar
Google Scholar
[12] Peng Z, Freunberger S A, Chen Y, Bruce P G 2012 Science 337 563
 Google Scholar
Google Scholar
[13] Lyu Z Y, Zhou Y, Dai W R, Cui X H, Lai M, Wang L, Huo F W, Huang W, Hu Z, Chen W 2017 Chem. Soc. Rev. 46 6046
 Google Scholar
Google Scholar
[14] Kang J H, Lee J, Jung J W, Park J, Jang T, Kim H S, Nam J S, Lim H, Yoon K R, Ryu W H, Kim I D, Byon H R 2020 ACS Nano 14 14549
 Google Scholar
Google Scholar
[15] Hummelshoj J S, Blomqvist J, Datta S, Vegge T, Rossmeisl J, Thygesen K S, Luntz A C, Jacobsen K W, Norskov J K 2010 J. Chem. Phys. 132 071101
 Google Scholar
Google Scholar
[16] Kang J, Jung Y S, Wei S H, Dillon A C 2012 Phys. Rev. B 85 035210
 Google Scholar
Google Scholar
[17] Assary R S, Lau K C, Amine K, Sun Y K, Curtiss L A 2013 J. Phys. Chem. C 117 8041
 Google Scholar
Google Scholar
[18] Mccloskey B D, Scheffler R, Speidel A, Bethune D S, Shelby R M, Luntz A C 2011 J. Am. Chem. Soc. 133 18038
 Google Scholar
Google Scholar
[19] Freunberger S A, Chen Y, Peng Z, Griffin J M, Hardwick L J, Barde F, Novak P, Bruce P G 2011 J. Am. Chem. Soc. 133 8040
 Google Scholar
Google Scholar
[20] Mo Y, Ong S P, Ceder G 2011 Phys. Rev. B 84 205446
 Google Scholar
Google Scholar
[21] Debart A, Paterson A J, Bao J, Bruce P G 2008 Angew. Chem. Int. Ed. Engl. 47 4521
 Google Scholar
Google Scholar
[22] Yu Y, Zhang B, He Y B, Huang Z D, Oh S W, Kim J K 2013 J. Mater. Chem. A 1 1163
 Google Scholar
Google Scholar
[23] Debart A, Bao J, Armstrong G, Bruce P G 2007 J. Power Sources 174 1177
 Google Scholar
Google Scholar
[24] Black R, Lee J H, Adams B, Mims C A, Nazar L F 2013 Angew. Chem. Int. Ed. Engl. 52 392
 Google Scholar
Google Scholar
[25] Cui Y, Wen Z, Liu Y 2011 Energy Environ. Sci. 4 4727
 Google Scholar
Google Scholar
[26] Zhu J, Ren X, Liu J, Zhang W, Wen Z 2015 Acs Catalysis 5 73
 Google Scholar
Google Scholar
[27] Lim H D, Song H, Gwon H, Park K Y, Kim J, Bae Y, Kim H, Jung S K, Kim T, Kim Y H, Lepro X, Ovalle Robles R, Baughman R H, Kang K 2013 Energy Environ. Sci. 6 3570
 Google Scholar
Google Scholar
[28] Lu Y C, Xu Z, Gasteiger H A, Chen S, Hamad Schifferli K, Shao Horn Y 2010 J. Am. Chem. Soc. 132 12170
 Google Scholar
Google Scholar
[29] Li F J, Tang D M, Chen Y, Golberg D, Kitaura H, Zhang T, Yamada A, Zhou H S 2013 Nano Lett. 13 4702
 Google Scholar
Google Scholar
[30] Jung H G, Jeong Y S, Park J B, Sun Y K, Scrosati B, Lee Y J 2013 ACS Nano 7 3532
 Google Scholar
Google Scholar
[31] Cai F, Lei X L 2023 Appl. Surf. Sci. 609 155331
 Google Scholar
Google Scholar
[32] Cai F, Lei X L, Ke Q, Ouyang C Y 2023 J. Phys. Chem. C 127 14232
 Google Scholar
Google Scholar
[33] Sun B, Huang X D, Chen S Q, Munroe P, Wang G X 2014 Nano Lett. 14 3145
 Google Scholar
Google Scholar
[34] Yoo E, Zhou H S 2011 ACS Nano 5 3020
 Google Scholar
Google Scholar
[35] Zhang Y, Ge J, Wang L, Wang D, Ding F, Tao X, Chen W 2013 Sci. Rep. 3 2771
 Google Scholar
Google Scholar
[36] 侯滨朋, 淦作亮, 雷雪玲, 钟淑英, 徐波, 欧阳楚英 2019 物理学报 68 128801
 Google Scholar
Google Scholar
Hou B P, Gan Z L, Lei X L, Zhong S Y, Xu B, Ouyang C Y 2019 Acta Phys. Sin. 68 128801
 Google Scholar
Google Scholar
[37] Tang Y B, Yin L C, Yang Y, Bo X H, Cao Y L, Wang H E, Zhang W J, Bello I, Lee S T, Cheng H M, Lee C S 2012 ACS Nano 6 1970
 Google Scholar
Google Scholar
[38] Wang H, Zhou Y, Wu D, Liao L, Zhao S L, Peng H L, Liu Z F 2013 Small 9 1316
 Google Scholar
Google Scholar
[39] Ren X D, Zhu J Z, Du F, Liu J J, Zhang W Q 2014 J. Phys. Chem. C 118 22412
 Google Scholar
Google Scholar
[40] Ren X D, Wang B Z, Zhu J Z, Liu J J, Zhang W Q, Wen Z Y 2015 Phys. Chem. Chem. Phys. 17 14605
 Google Scholar
Google Scholar
[41] Hou B P, Lei X L, Zhong S Y, Sun B Z, Ouyang C Y 2020 Phys. Chem. Chem. Phys. 22 14216
 Google Scholar
Google Scholar
[42] Salehi M, Shariatinia Z, Sadeghi A 2019 J. Electroanal. Chem. 832 165
 Google Scholar
Google Scholar
[43] Kresse G, Furthmüller J 1996 Phys. Rev. B 54 11169
 Google Scholar
Google Scholar
[44] Blöchl P E 1994 Phys. Rev. B 50 17953
 Google Scholar
Google Scholar
[45] Perdew J P, Burke K, Ernzerhof M 1996 Phys. Rev. Lett. 77 3865
 Google Scholar
Google Scholar
[46] Monkhorst H J, Pack J D 1976 Phys. Rev. B 13 5188
 Google Scholar
Google Scholar
[47] Grimme S, Antony J, Ehrlich S, Krieg H 2010 J. Chem. Phys. 132 154104
 Google Scholar
Google Scholar
[48] Topsakal M, Ciraci S 2012 Phys. Rev. B 86 205402
 Google Scholar
Google Scholar
计量
- 文章访问数: 4982
- PDF下载量: 187
- 被引次数: 0













 下载:
下载: