-
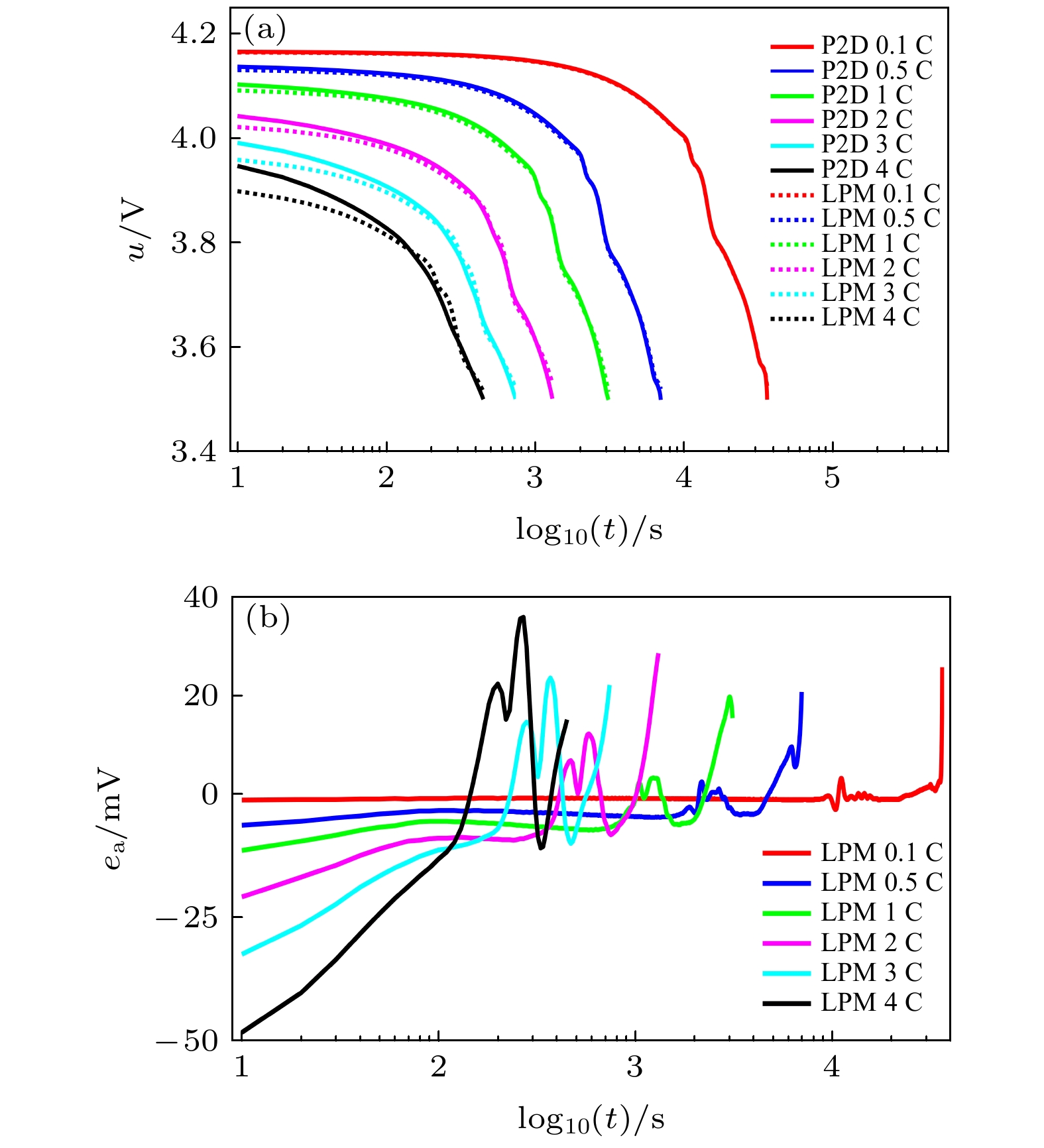
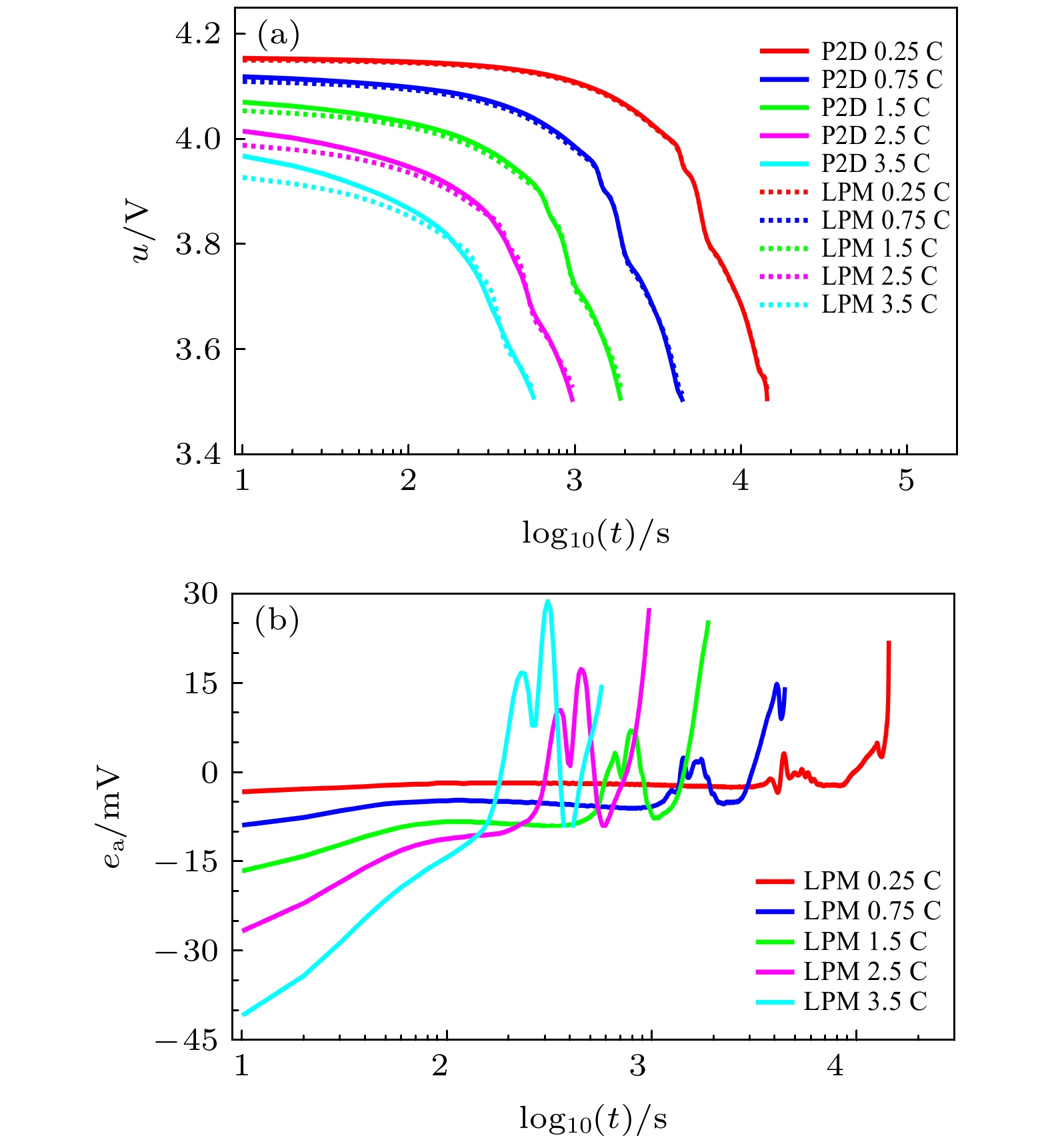
可靠而高效的锂离子电池模型是电池管理系统状态估计与故障诊断的基础. 采用偏微分方程描述的准二维(P2D)机理模型的参数多, 虽然模型准确性高, 但计算费时, 需降阶处理才能更好地应用在车载电池管理系统. 为此, 基于相同模型参数, 建立锂离子电池的P2D模型及其降阶模型—单粒子模型(SPM)和集总粒子模型(LPM), 对三种电化学机理模型电池端电压的计算精度和时间进行对比研究. 结合多孔电极模型和浓溶液理论, 基于电池均匀电流密度的假设条件, 按照电极固相和液相体积比重新分配电流密度, 推导了由液相锂离子浓度分布差异所导致的液相浓差过电压和欧姆内阻, 补偿LPM的电压误差. 采用恒流放电、脉冲放电和动态电应力测试(DST)工况对比分析优化的LPM, P2D模型和SPM的电学性能. 结果表明: 优化的LPM不仅能够以更少的参数大幅降低模型计算时间, 而且能够保证模型电压的计算精度.A reliable and efficient lithium-ion battery model is the basis of state estimation and fault diagnosis in the battery management system (BMS). The pseudo-two-dimensional (P2D) mechanism model represented by using partial differential equations has many parameters and high accuracy, but its calculation is time-consuming. The model order should be reduced for the on-board BMS. Here in this work, by using the same model parameters, the P2D model and the order-reduced models including the single particle model (SPM) and the lumped particle model (LPM) are built and their voltage accuracies and operation time are compared with each other. Based on the porous electrode model, the concentrated solution theory, and the assumption of uniform current of the battery, the current density is redistributed by the volume ratio of the solid phase to the liquid phase of the electrode. Furthermore, the overpotential and ohmic resistance caused by the lithium-ion concentration distribution in the liquid phase is deduced to offset the battery voltage bias under a large discharge rate. An unconstrained convex optimization method is established to optimize the concentration difference overpotential of the liquid phase of the P2D model battery, the optimized results of which are used to compensate for the LPM voltage.Under the conditions of the constant current discharge (CCD) of 0.1C — 4C at the ambient temperature, pulse discharge and dynamic stress test (DST), the electrical performance and the operation time of both the LPM and the SPM are compared to those of the P2D model. Some results are validated and given by the model simulation. Firstly, the accuracy of LPM is over 30% higher than those of the SPM at the CCD rates of 0.1 C, 0.5 C and 1 C, and about 30% worse than those of the SPM at the CCD rates of 2 C, 3 C, and 4 C. Secondly, the optimized LPM can produce a voltage in good approximation to the voltage of the P2D model with an absolute relative error of the model voltage below 1.5% whether it is caused by the CCD or the DST. Thirdly, the optimized LPM can run efficiently and the needed calculation time cuts down by 85% and 65% for the P2D and the SPM, respectively. For the real-time applications of lithium-ion batteries, the proposed compensation method by adding the overpotential in the liquid phase can make the LPM produce reliable voltage with shorter operation time than either the SPM or the P2D model.
-
Keywords:
- lithium-ion battery /
- electrochemical model /
- order reduction /
- model optimization
[1] Hu X S, Li S B, Peng H 2012 J. Power Sources 198 359
 Google Scholar
Google Scholar
[2] Wang Y J, Tian J Q, Sun Z D, Wang L, Chen Z h 2020 Renewable Sustainable Energy Rev. 131 110015-1
[3] Lai X, Wang S, Ma S, Xie J Y, Zheng Y J 2020 Electrochim. Acta 330 135239
 Google Scholar
Google Scholar
[4] Tang K, Yu X Q, Sun J P, Li H, Huang X J 2011 Electrochim. Acta 56 4869
 Google Scholar
Google Scholar
[5] Shen Z, Cao L, Rahn C, Wang C Y 2013 J. Electrochem. Soc. 160 A1842
 Google Scholar
Google Scholar
[6] 凌仕刚, 吴妖杨, 张舒, 高健, 王少飞, 李泓 2015 储能科学与技术 4 83
 Google Scholar
Google Scholar
Ling S G, Wu J Y, Zhang S, Gao J, Wang S F, Li H 2015 Energy Storage Science and Technology 4 83
 Google Scholar
Google Scholar
[7] Subramanian V R, Diwakar V D, Tapriyal D 2005 J. Electrochem. Soc. 152 A2002
 Google Scholar
Google Scholar
[8] Dao T S, Vyasarayani C P, McPhee J 2012 J. Power Sources 198 329
 Google Scholar
Google Scholar
[9] Luo W L, Lyu C, Wang L X, Zhang L Q 2013 Microelectron. Reliab. 53 797
 Google Scholar
Google Scholar
[10] Kemper P, Li S E, Kum D 2015 J. Power Sources 286 510
 Google Scholar
Google Scholar
[11] Deng Z, Yang L, Deng H, Cai Y, Li D 2018 Energy 142 838
 Google Scholar
Google Scholar
[12] Cai L, White R E 2009 J. Electrochem. Soc. 156 A154
 Google Scholar
Google Scholar
[13] Forman J, Bashash S, Stein J, Fathy H 2011 J. Electrochem. Soc. 158 A93
 Google Scholar
Google Scholar
[14] Subramanian V, Boovaragavan V, Ramadesigan V, Arabandi M 2009 J. Electrochem. Soc. 156 A260
 Google Scholar
Google Scholar
[15] Luo W L, Lyu C, Wang L X, Zhang L Q 2013 J. Power Sources 241 295
 Google Scholar
Google Scholar
[16] Romero-Becerril A, Alvarez-Icaza L 2011 J. Power Sources 196 10267
 Google Scholar
Google Scholar
[17] Santhanagopalan S, Guo Q, Ramadass P, White R E 2006 J. Power Sources 156 620
 Google Scholar
Google Scholar
[18] Di Domenico D, Stefanopoulou A, Fiengo G 2010 J. Dyn. Syst. Meas. Contr. 132 061302-1
 Google Scholar
Google Scholar
[19] Prada E, Di Domenico D, Creff Y, Bernard J, Sauvant-Moynot V, Huet F 2012 J. Electrochem. Soc. 159 A1508
 Google Scholar
Google Scholar
[20] Ekström H, Fridholm B, Lindbergh G 2018 J. Power Sources 402 296
 Google Scholar
Google Scholar
[21] Doyle M, Fuller T F, Newman J 1993 J. Power Sources 140 1526
 Google Scholar
Google Scholar
[22] Fuller T F, Doyle M, Newman J 1994 J. Electrochem. Soc. 141 1
 Google Scholar
Google Scholar
[23] Han X B, Ouyang M G, Lu L G, Li J Q 2015 J. Power Sources 278 814
 Google Scholar
Google Scholar
-
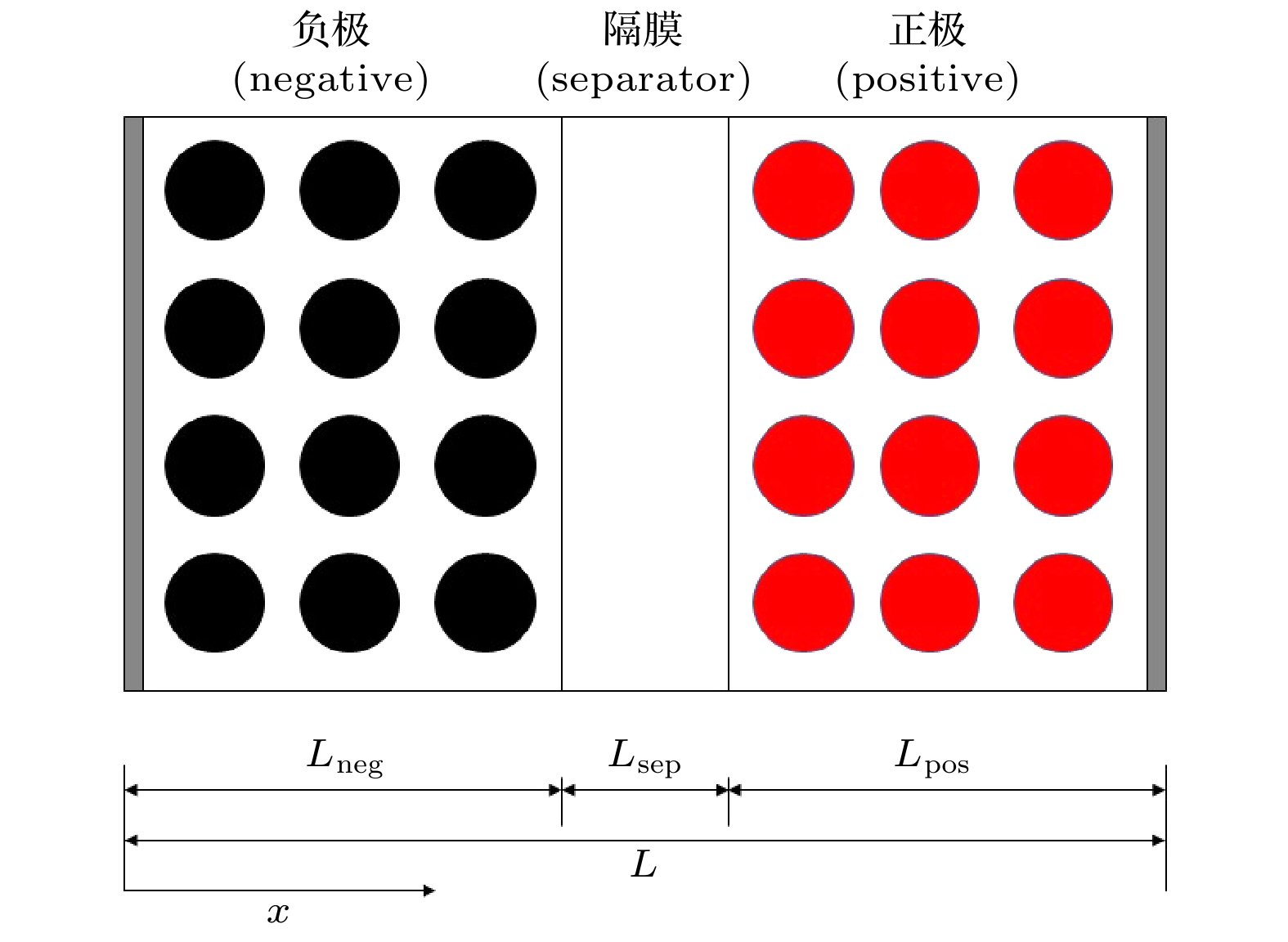
表 1 P2D模型参数
Table 1. Parameter symbols of P2D.
符号 物理量 单位 $c_{\rm{s}}^i$ 多孔电极固相锂离子浓度 ${\rm{mol}} \cdot {{\rm{m}}^{ - 3}}$ $c_{\rm{e}}^i$ 多孔电极液相锂离子浓度 ${\rm{mol}} \cdot {{\rm{m}}^{ - 3}}$ $c_{\rm{e}}^{{\rm{sep}}}$ 隔膜液相锂离子浓度 ${\rm{mol}} \cdot {{\rm{m}}^{ - 3}}$ $c_{{\rm{s, 0}}}^i$ 多孔电极固相锂离子初始浓度 ${\rm{mol}} \cdot {{\rm{m}}^{ - 3}}$ ${c_{{\rm{e, 0}}}}$ 液相锂离子初始浓度 ${\rm{mol}} \cdot {{\rm{m}}^{ - 3}}$ $c_{{\rm{s, max}}}^i$ 多孔电极固相粒子最大
锂离子浓度${\rm{mol}} \cdot {{\rm{m}}^{ - 3}}$ $c_{{\rm{s, surf}}}^i$ 多孔电极固相粒子表面
锂离子浓度${\rm{mol}} \cdot {{\rm{m}}^{ - 3}}$ $D_{\rm{s}}^i$ 多孔电极固相锂离子扩散系数 $ { {\rm{m} }^{2} } \cdot { {\rm{s} }^{ - 1} } $ $D_{\rm{e}}^{}$ 液相锂离子扩散系数 $ { {\rm{m} }^{2} } \cdot { {\rm{s} }^{ - 1} } $ $D_{{\rm{e, eff}}}^i$ 多孔电极液相锂离子
有效扩散系数$ { {\rm{m} }^{2} } \cdot { {\rm{s} }^{ - 1} } $ $D_{{\rm{e, eff}}}^{{\rm{sep}}}$ 隔膜液相锂离子有效扩散系数 $ { {\rm{m} }^{2} } \cdot { {\rm{s} }^{ - 1} } $ $j_{\rm{n}}^i$ 多孔电极锂离子孔壁通量 ${\rm{mol}} \cdot {{\rm{s}}^{ - 1}} \cdot {{\rm{m}}^{ - 2}}$ $j_{}^i$ 多孔电极电流密度 ${\rm{A}} \cdot {{\rm{m}}^{ - 3}}$ ${R^i}$ 多孔电极球形粒子半径 m $\varepsilon _{\rm{e}}^i$ 多孔电极液相体积分数 — $\varepsilon _s^i$ 多孔电极固相体积分数 — $\varepsilon _{\rm{e}}^{{\rm{sep}}}$ 隔膜液相体积分数 — ${t_{ + }}$ 锂离子迁移系数 — ${f_ \pm }$ 液相平均摩尔活度系数 — $a_{\rm{s}}^i$ 多孔电极球形粒子的比表面积 ${{\rm{m}}^{ - 1}}$ $\sigma _{\rm{s}}^i$ 多孔电极固相电导率 ${\rm{S}} \cdot {{\rm{m}}^{ - 1}}$ $\sigma _{{\rm{s, eff}}}^i$ 多孔电极固相有效电导率 ${\rm{S}} \cdot {{\rm{m}}^{ - 1}}$ $\kappa $ 液相离子电导率 ${\rm{S}} \cdot {{\rm{m}}^{ - 1}}$ $\kappa _{{\rm{eff}}}^i$ 液相有效离子电导率 ${\rm{S}} \cdot {{\rm{m}}^{ - 1}}$ ${\kappa _{{\rm{D}}, i}}$ 有效液相离子扩散导电系数 ${\rm{A}} \cdot {{\rm{m}}^2} \cdot {\rm{mo}}{{\rm{l}}^{ - 1}}$ $\phi _{\rm{s}}^i$ 多孔电极固相电势 V $\phi _{\rm{e}}^{}$ 液相电势 V ${I_{\rm{b}}}$ 电池电流 A $ j_0^i $ 多孔电极参考交换电流密度 ${\rm{A}} \cdot {{\rm{m}}^{ - 2}}$ ${\eta ^i}$ 电极过电势 V ${\alpha _{{\rm{a, i}}}}$/${\alpha _{{\rm{c, i}}}}$ 电极氧化还原反应电荷传递系数, 为0.5 — ${k^i}$ 电极反应常数 ${{\rm{m}}^{2.5}} \cdot {\rm{mo}}{{\rm{l}}^{ - 0.5}} \cdot {{\rm{s}}^{ - 1}}$ $U_{}^i$ 电极平衡电势 V $R_{{\rm{SEI}}}^i$ SEI膜电阻 $\Omega \cdot {{\rm{m}}^{ - 2}}$ ${V_{{\rm{P2 D}}}}$ P2D模型端电压 V A 电池横截面积 ${{\rm{m}}^2}$ Brug Bruggman系数, 取值1.5 — F 法拉第常数 ${\rm{C}} \cdot {\rm{mo}}{{\rm{l}}^{ - 1}}$ R 普适气体常数 ${\rm{J}} \cdot {\rm{mo}}{{\rm{l}}^{ - 1}} \cdot {{\rm{K}}^{ - 1}}$ T 温度 K 表 2 SPM参数
Table 2. Parameter symbols of SPM.
符号 物理量 单位 $c_{{\rm{s, avg}}}^i$ 多孔电极粒子平均锂离子浓度 ${\rm{mol}} \cdot {{\rm{m}}^{ - 3}}$ $c_{{\rm{s, max}}}^i$ 多孔电极最大粒子锂离子浓度 ${\rm{mol}} \cdot {{\rm{m}}^{ - 3}}$ ${c_{\rm{e}}}$ 液相锂离子浓度 ${\rm{mol}} \cdot {{\rm{m}}^{ - 3}}$ ${\overline j ^i}$ 平均交换电流密度 ${\rm{A}} \cdot {{\rm{m}}^{ - 2}}$ $\phi _{{\rm{SP}}}^i$ 电池电极电势 V ${V_{{\rm{SP}}}}$ 电池端电压 V 表 3 LPM参数
Table 3. Parameter symbols of LPM.
符号 物理量 单位 c LPM粒子内锂离子浓度 ${\rm{mol}} \cdot {{\rm{m}}^{ - 3}}$ ${c_{\max }}$ LPM最大锂离子浓度 ${\rm{mol}} \cdot {{\rm{m}}^{ - 3}}$ ${\eta _{{\rm{ohm}}}}$ 欧姆过电压 V ${\eta _{{\rm{act}}}}$ 活化过电压 V ${\eta _{{\rm{conc}}}}$ 浓度过电压 V ${c_{{\rm{surf}}}}$ 粒子表面锂离子浓度 ${\rm{mol}} \cdot {{\rm{m}}^{ - 3}}$ ${c_{{\rm{avg}}}}$ 粒子平均锂离子浓度 ${\rm{mol}} \cdot {{\rm{m}}^{ - 3}}$ ${\eta _{{\rm{ohm, 1 C}}}}$ 1 C欧姆过电压 V ${I_{{\rm{1 C}}}}$ 1 C电流 A ${J_0}$ 无量纲交换电流密度 1 $\tau $ 等效扩散系数 s ${Q_{{\rm{cell}}}}$ 电池容量 ${\rm{Ah} }$ X 归一化粒子半径 — 表 4 电化学模型参数值
Table 4. Parameter values of Electrochemical model.
中文参数名 负极 隔膜 正极 单位 厚度 10 25 100 μm 粒子半径 10 — 10 μm 锂离子扩散系数 $3.9 \times 1{0^{ - 14}}$ — $1 \times 1{0^{ - 13}}$ $ { {\rm{m} }^{2} } \cdot { {\rm{s} }^{ - 1} } $ 初始固相浓度 19624 — 20046 ${\rm{mol}} \cdot {{\rm{m}}^{ - 3}}$ 最大锂离子浓度 24983 — 51228 ${\rm{mol}} \cdot {{\rm{m}}^{ - 3}}$ 电导率 100 $\kappa $ 10 S·m–1 初始电解液浓度 1000 1000 1000 $ { {\rm{m} }^{2} } \cdot { {\rm{s} }^{ - 1} } $ 电解液扩散系数 $2.7877 \times 1{0^{ - 10}}$ $2.7877 \times 1{0^{ - 10}}$ $2.7877 \times 1{0^{ - 10}}$ $ { {\rm{m} }^{2} } \cdot { {\rm{s} }^{ - 1} } $ 传递数 0.4 0.4 0.4 — 电解液体积分数 0.3 1 0.3 — SEI膜电阻 $0$ — $0$ ${\rm{m}}\Omega $ 反应速率常数 $9.6487 \times 1{0^{ - 6}}$ — $2.89461 \times 1{0^{ - 6}}$ ${\rm{mo}}{{\rm{l}}^{2.5}} \cdot {{\rm{m}}^{ - 0.5}} \cdot {{\rm{s}}^{ - 1}}$ 活性材料体积分数 0.6 — 0.5 — 填料体积分数 0.1 — 0.2 — 电极横截面积 0.1 — — ${{\rm{m}}^2}$ 1 C电流 30 — — A 放电截止电压 3.4 — — V 充电截止电压 4.2 — V 容量 30 — — Ah 温度 298 — — K 普适气体常数 8.314 — — ${\rm{J}} \cdot {\rm{mo}}{{\rm{l}}^{ - 1}} \cdot {{\rm{K}}^{ - 1}}$ 法拉第常数 96487 — — ${\rm{C}} \cdot {\rm{mo}}{{\rm{l}}^{ - 1}}$ 表 5 测试集电压误差对比
Table 5. Comparison of test set voltage errors.
倍率/C SPM绝对
误差/mVSPM相对
误差/%LPM绝对
误差/mVLPM相对
误差/%0.1 –44.50 –1.28 30.03 0.87 0.5 74.20 2.12 42.30 1.21 1 83.70 2.39 60.70 1.72 2 88.00 2.51 103.40 2.95 3 98.50 2.81 129.30 3.66 4 125.50 3.58 173.20 4.75 表 6 不同倍率下参数a取值
Table 6. Parameter values of a in the different discharge rate.
倍率 参数a 0.1 –0.148 0.5 –0.703 1 –1.331 2 –2.431 3 –3.438 4 –4.476 表 7 优化后LPM电压误差
Table 7. Optimized LPM voltage errors.
倍率/C 绝对误
差/mV相对误
差/%倍率/C 绝对误
差/mV相对误
差/%0.1 25.7 0.73 0.25 22.1 0.63 0.5 20.7 0.59 0.75 14.8 0.42 1 19.7 0.56 1.5 25.5 0.73 2 28.5 0.81 2.5 –32.7 –0.81 3 –39.9 –0.99 3.5 –49.2 –1.23 4 –57.6 –1.45 DST –55.4 –1.40 脉冲
放电41.5 1.05 表 8 计算用时对比
Table 8. Comparison of the calculation time.
测试集
用时/s验证集
用时/s脉冲放
电用时/sDST工况
用时/sLPM 24 14 8 11 P2D 169 92 85 108 占比 14.2% 15.22% 9.41% 10.19% -
[1] Hu X S, Li S B, Peng H 2012 J. Power Sources 198 359
 Google Scholar
Google Scholar
[2] Wang Y J, Tian J Q, Sun Z D, Wang L, Chen Z h 2020 Renewable Sustainable Energy Rev. 131 110015-1
[3] Lai X, Wang S, Ma S, Xie J Y, Zheng Y J 2020 Electrochim. Acta 330 135239
 Google Scholar
Google Scholar
[4] Tang K, Yu X Q, Sun J P, Li H, Huang X J 2011 Electrochim. Acta 56 4869
 Google Scholar
Google Scholar
[5] Shen Z, Cao L, Rahn C, Wang C Y 2013 J. Electrochem. Soc. 160 A1842
 Google Scholar
Google Scholar
[6] 凌仕刚, 吴妖杨, 张舒, 高健, 王少飞, 李泓 2015 储能科学与技术 4 83
 Google Scholar
Google Scholar
Ling S G, Wu J Y, Zhang S, Gao J, Wang S F, Li H 2015 Energy Storage Science and Technology 4 83
 Google Scholar
Google Scholar
[7] Subramanian V R, Diwakar V D, Tapriyal D 2005 J. Electrochem. Soc. 152 A2002
 Google Scholar
Google Scholar
[8] Dao T S, Vyasarayani C P, McPhee J 2012 J. Power Sources 198 329
 Google Scholar
Google Scholar
[9] Luo W L, Lyu C, Wang L X, Zhang L Q 2013 Microelectron. Reliab. 53 797
 Google Scholar
Google Scholar
[10] Kemper P, Li S E, Kum D 2015 J. Power Sources 286 510
 Google Scholar
Google Scholar
[11] Deng Z, Yang L, Deng H, Cai Y, Li D 2018 Energy 142 838
 Google Scholar
Google Scholar
[12] Cai L, White R E 2009 J. Electrochem. Soc. 156 A154
 Google Scholar
Google Scholar
[13] Forman J, Bashash S, Stein J, Fathy H 2011 J. Electrochem. Soc. 158 A93
 Google Scholar
Google Scholar
[14] Subramanian V, Boovaragavan V, Ramadesigan V, Arabandi M 2009 J. Electrochem. Soc. 156 A260
 Google Scholar
Google Scholar
[15] Luo W L, Lyu C, Wang L X, Zhang L Q 2013 J. Power Sources 241 295
 Google Scholar
Google Scholar
[16] Romero-Becerril A, Alvarez-Icaza L 2011 J. Power Sources 196 10267
 Google Scholar
Google Scholar
[17] Santhanagopalan S, Guo Q, Ramadass P, White R E 2006 J. Power Sources 156 620
 Google Scholar
Google Scholar
[18] Di Domenico D, Stefanopoulou A, Fiengo G 2010 J. Dyn. Syst. Meas. Contr. 132 061302-1
 Google Scholar
Google Scholar
[19] Prada E, Di Domenico D, Creff Y, Bernard J, Sauvant-Moynot V, Huet F 2012 J. Electrochem. Soc. 159 A1508
 Google Scholar
Google Scholar
[20] Ekström H, Fridholm B, Lindbergh G 2018 J. Power Sources 402 296
 Google Scholar
Google Scholar
[21] Doyle M, Fuller T F, Newman J 1993 J. Power Sources 140 1526
 Google Scholar
Google Scholar
[22] Fuller T F, Doyle M, Newman J 1994 J. Electrochem. Soc. 141 1
 Google Scholar
Google Scholar
[23] Han X B, Ouyang M G, Lu L G, Li J Q 2015 J. Power Sources 278 814
 Google Scholar
Google Scholar
计量
- 文章访问数: 15220
- PDF下载量: 401
- 被引次数: 0













 下载:
下载: