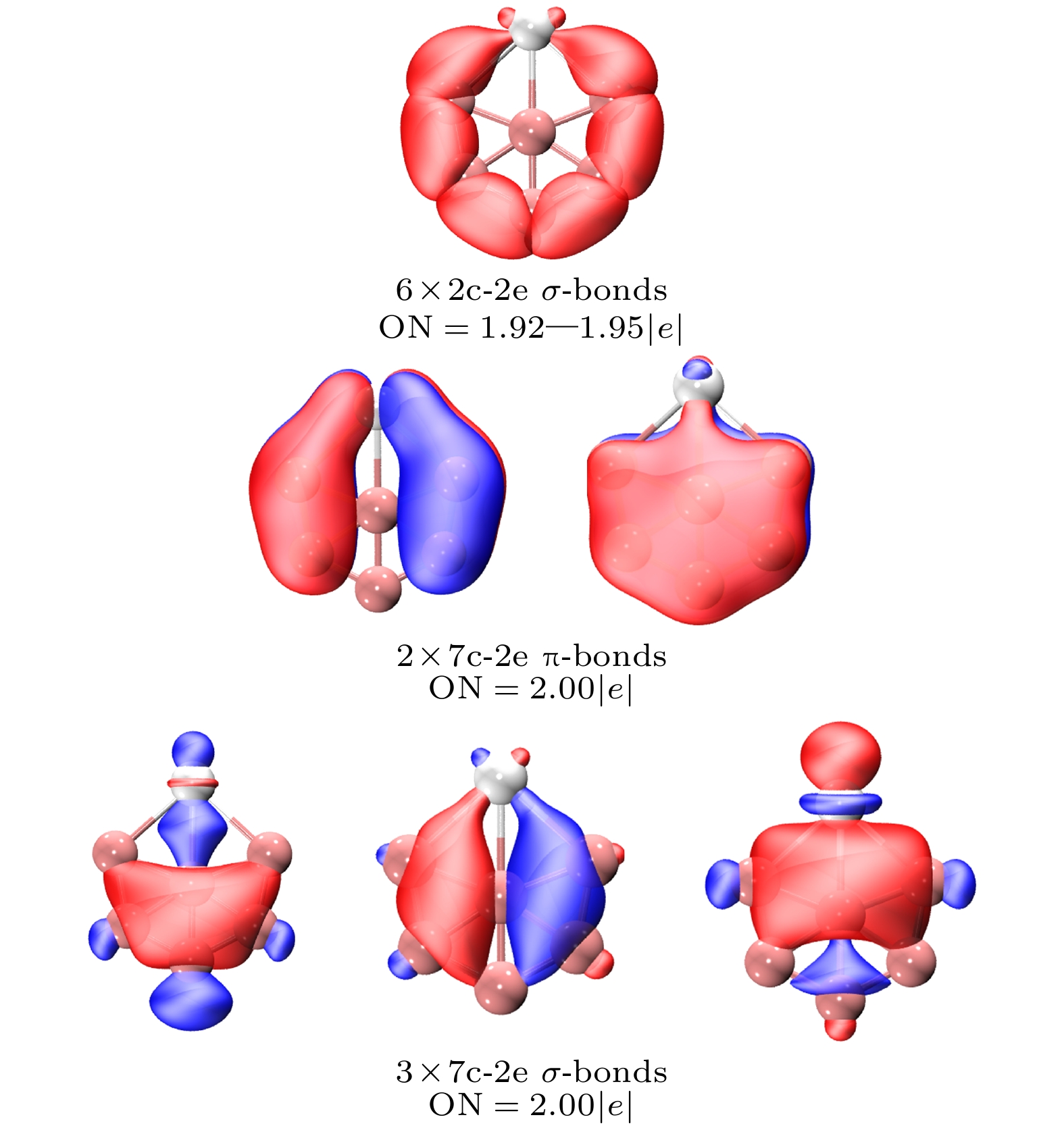
-
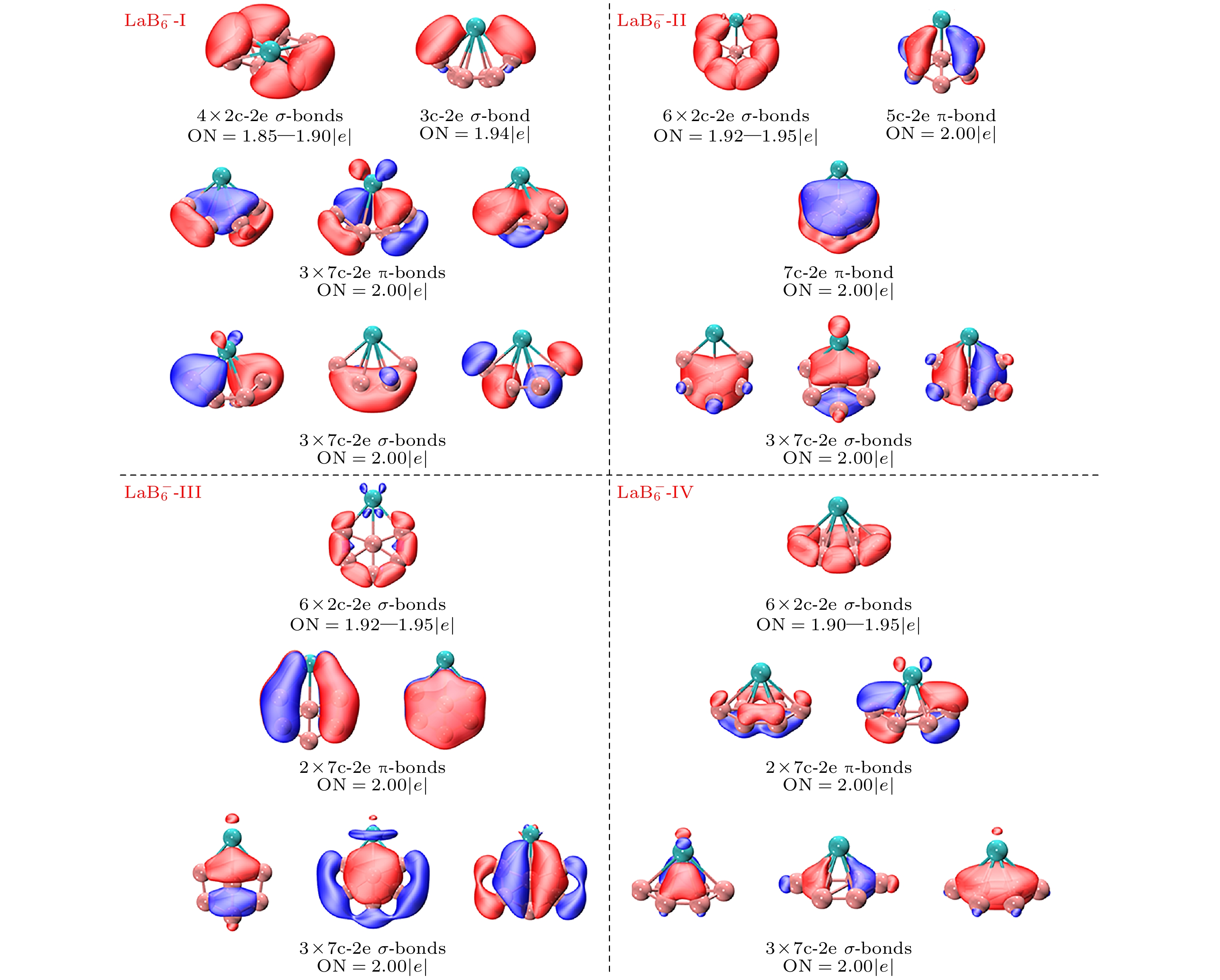
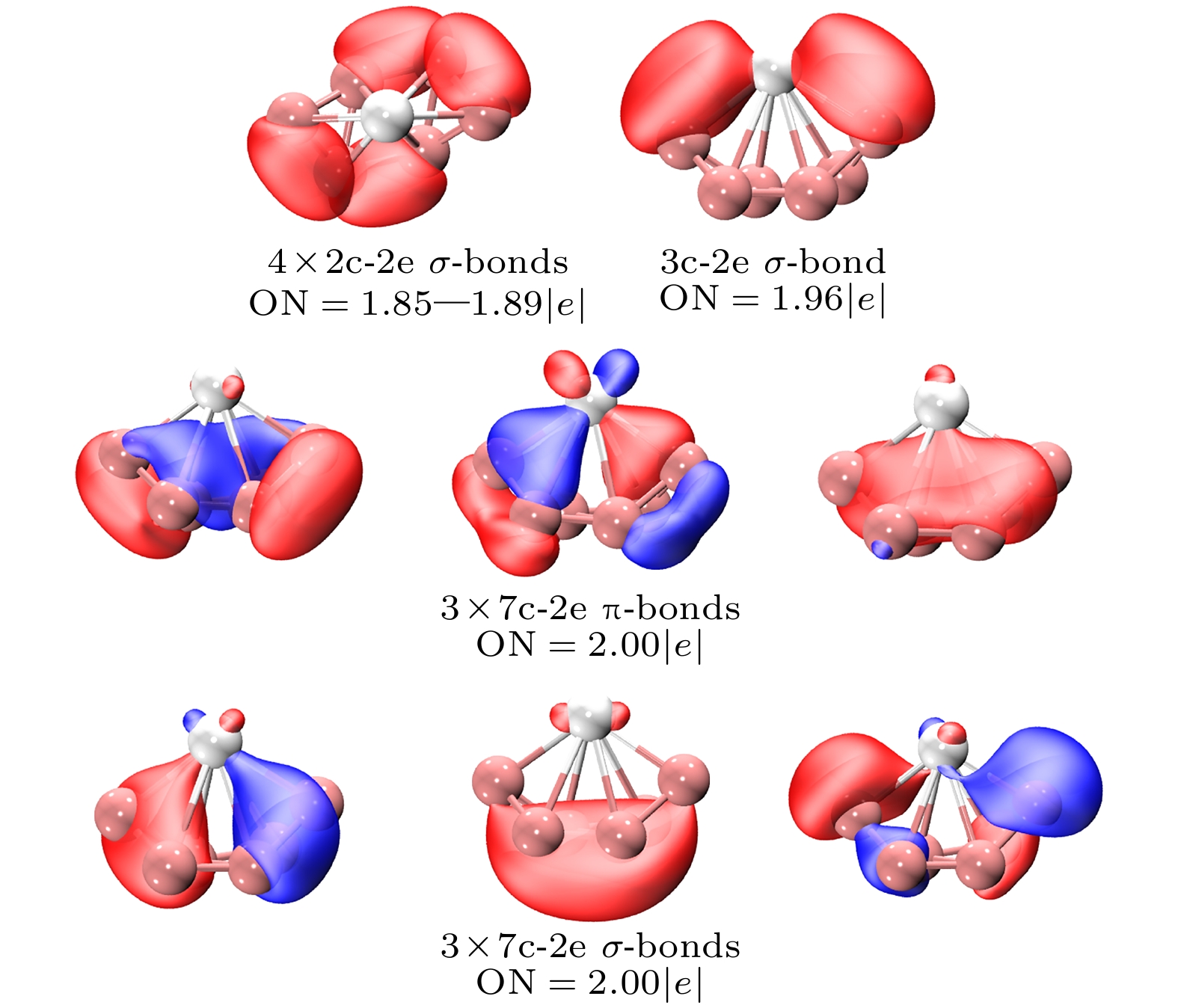
稀土掺杂硼团簇因其特殊的光学, 电学和磁学性质受到广泛关注. 本文采用人工蜂群算法结合密度泛函理论, 在PBE0/RE/SDD//B/6-311+G*水平下研究了稀土掺杂阴离子硼团簇$ {\text{REB}}_n^ - $(RE = La, Sc; n = 6, 8) 的几何结构、电子性质、稳定性和芳香性. 计算结果表明, 阴离子$ {\text{REB}}_n^ - $ (RE = La, Sc; n = 6) 的基态结构具有C2对称性, 掺杂的镧系原子位于顶部中心形成“船形”结构. 通过与实验光电子能谱的比较, 证实$ {\text{LaB}}_{8}^ - $ 的基态结构类似于三维的“筝形”结构, 而$ {\text{ScB}}_{8}^ - $ 的基态结构则是Sc原子位于“伞柄”处形成的具有C7v对称性的“伞状”结构. B—B之间存在通过共享电子对的相互作用, 而RE—B之间的电子定域性不如B—B之间的电子定域性. 模拟得出的光电子能谱峰值位置与实验结果的吻合度较高, 充分证实了研究获取的全局能量最低结构与实验观测结构的一致性. $ {\text{LaB}}_{6}^ - $ 和$ {\text{ScB}}_{6}^ - $ 的最低能量结构均为σ-π双芳香簇, 表现出明显的芳香性. 此外, 分别计算了$ {\text{REB}}_n^ - $(RE = La, Sc; n = 6, 8)的总态密度, 以及RE原子和硼簇的局部态密度, 并对其轨道能级密度进行了评估. 开壳层的$ {\text{ScB}}_{8}^ - $态密度谱呈现出自旋极化现象, 这表明其作为基元可以组装成具有磁性的纳米材料. 这些对稀土掺杂硼团簇的研究有助于深入理解纳米材料的结构和性质演变规律, 为设计具有实际价值的纳米材料提供了重要的理论支持.Rare earth doped boron clusters have attracted much attention due to their special optical, electrical and magnetic properties. The geometric structures, stability, electronic properties and aromaticity of negative rare earth doped boron clusters $ {\text{REB}}_n^ - $ (RE = La, Sc; n = 6, 8) are investigated with the artificial bee colony algorithm combined with density functional theory calculations at the PBE0/RE/SDD//B/6-311+G* level of theory. Calculations show that the ground state structures of $ {\text{REB}}_n^ - $ (RE = La, Sc; n = 6, 8) are all of C2 symmetry, and the doped lanthanide atom is located in a “boat-shaped” structure at the top center. By comparing with the experimental photoelectron spectra, it is confirmed that the ground state structure of $ {\text{LaB}}_{8}^ - $ is a “zither-like” three-dimensional structure, and the ground state structure of $ {\text{ScB}}_{8}^ - $ is an “umbrella” structure with C7v symmetry formed by the scandium atom at the “umbrella handle”. The electron localization between RE—B is not as good as that between B—B. The simulated photoelectron spectra have similar spectral characteristics to the experimental results. The lowest energy structures of $ {\text{LaB}}_{6}^ - $ and $ {\text{ScB}}_{6}^ - $ are σ-π double aromatic clusters, and the structures exhibit aromaticity. The density of states of low-energy isomers shows that the open shell $ {\text{ScB}}_{8}^ - $ density of states spectrum exhibits spin polarization phenomenon, which is expected to assemble magnetic material components. These studies contribute to understanding the evolution of structure and properties of nanomaterials, and provide important theoretical support for designing nanomaterials with practical value.
-
Keywords:
- density functional theory /
- rare earth doped clusters /
- structural optimization /
- aromaticity
[1] 张超江, 许洪光, 徐西玲, 郑卫军 2021 物理学报 70 023601
 Google Scholar
Google Scholar
Zhang C J, Xu H G, Xu X L, Zheng W J 2021 Acta Phys. Sin. 70 023601
 Google Scholar
Google Scholar
[2] Boustani I 1997 Phys. Rev. B 5 16426
[3] Kiran B, Bulusu S, Zhai H J, Yoo S, Zeng X C, Wang L S 2005 Proc. Natl. Acad. Sci. 102 961
 Google Scholar
Google Scholar
[4] 李世雄, 张正平, 隆正文, 秦水介 2017 物理学报 6 103102
 Google Scholar
Google Scholar
Li S X, Zhang Z P, Long Z W, Qin S J 2017 Acta Phys. Sin. 6 103102
 Google Scholar
Google Scholar
[5] Sergeeva A P, Popov I A, Piazza Z A, Li W L, Romanescu C, Wang L S, Boldyrev A I 2014 Acc. Chem. Res. 47 1349
 Google Scholar
Google Scholar
[6] Jian T, Chen X, Li S D, Boldyrev A I, Li J, Wang L S 2019 Chem. Soc. Rev. 48 3550
 Google Scholar
Google Scholar
[7] Wang L S 2016 Int. Rev. Phys. Chem. 35 69
 Google Scholar
Google Scholar
[8] Bai H, Chen T T, Chen Q, Zhao X Y, Zhang Y Y, Chen W J, Li W L, Cheng L F, Bai B, Cavanagh J, Huang W, Li S D, Li J, Wang L S 2019 Nanoscale 11 23286
 Google Scholar
Google Scholar
[9] 刘立仁, 雷雪玲, 陈杭, 祝恒江 2009 物理学报 58 5355
 Google Scholar
Google Scholar
Liu L R, Lei X L, Chen H, Zhu H J 2009 Acta Phys. Sin. 58 5355
 Google Scholar
Google Scholar
[10] Pham H T, Duong L V, Pham B Q, Nguyen M T 2013 Chem. Phys. Lett. 577 32
 Google Scholar
Google Scholar
[11] Zhai H J, Alexandrova A N, Birch K A, Boldyrev A I, Wang L S 2003 Angew. Chem. Int. Ed. 42 6004
 Google Scholar
Google Scholar
[12] Zhai H J, Zhao Y F, Li W L, Chen Q, Bai H, Hu H S, Piazza Z A, Tian W J, Lu H G, Wu Y B, Mu Y W, Wei G F, Liu Z P, Li J, Li S D, Wang L S 2014 Nature Chem. 6 727
 Google Scholar
Google Scholar
[13] Islas R, Heine T, Ito K, Schleyer P V, Merino G 2007 J. Am. Chem. Soc. 129 14767
 Google Scholar
Google Scholar
[14] Romanescu C, Galeev T R, Li W L, Boldyrev A I, Wang L S 2013 Acc. Chem. Res. 46 350
 Google Scholar
Google Scholar
[15] Saha R, Kar S, Pan S, Martínez-Guajardo G, Merino G, Chattaraj P K 2017 J. Phys. Chem. A 121 2971
 Google Scholar
Google Scholar
[16] 李世雄, 陈德良, 张正平, 隆正文 2020 物理学报 69 193101
 Google Scholar
Google Scholar
Li S X, Chen D L, Zhang Z P, Long Z W 2020 Acta Phys. Sin. 69 193101
 Google Scholar
Google Scholar
[17] Li H F, Wang H Q, Zhang J M, Qin L X, Zheng H, Zhang Y H 2024 Molecules 29 1692
 Google Scholar
Google Scholar
[18] 蒋贤明, 王怀谦, 曹宇, 孙之惠, 曹玉芳, 吴伟宾 2018 高等学校化学学报 39 1976
 Google Scholar
Google Scholar
Jiang X M, Wang H Q, Cao Y, Sun Z H, Cao Y F, Wu W B 2018 Chemical Journal of Chinese Universities 39 1976
 Google Scholar
Google Scholar
[19] Zheng H, Wang H Q, Li H F, Zhang J M, Zhang Y H, Qin L X, Mei X J, Jiang K L, Zeng J K, Zhang B, Wu W H 2024 Chem. Phys. 583 112321
 Google Scholar
Google Scholar
[20] Wen S H, Zhou J J, Zheng K Z, Bednarkiewicz A, Liu X G, Jin D Y 2018 Nat. Commun. 9 2415
 Google Scholar
Google Scholar
[21] Jiang L Y, Wang H Q, Li H F, Xie B, Zhang J M, Ji J Y 2023 Chem. Phys. 567 111819
 Google Scholar
Google Scholar
[22] Wang H Q, Li H F 2014 RSC Adv. 4 29782
 Google Scholar
Google Scholar
[23] Yi Z G, Luo Z C, Qin X, Chen Q S, Liu X G 2020 Acc. Chem. Res. 53 2692
 Google Scholar
Google Scholar
[24] Qin L X, Li H F, Xiao B W, Zhang J M, Zeng J K, Mei X J, Zhang Y H, Zheng H, Wang H Q 2023 Chem. Phys. 575 112064
 Google Scholar
Google Scholar
[25] Li W L, Chen T T, Xing D H, Chen X, Li J, Wang L S 2018 Proc. Natl. Acad. Sci. 115 E6972
[26] Robinson P J, Zhang X X, McQueen T, Bowen K H, Alexandrova A N 2017 J. Phys. Chem. A 121 1849
 Google Scholar
Google Scholar
[27] Chen T T, Li W L, Li J, Wang L S 2019 Chem. Sci. 10 2534
 Google Scholar
Google Scholar
[28] Zuo J N, Zhang L L, Chen B L, He K H, Dai W, Ding K W, Lu C 2024 J. Phys. Condens. Matter 36 015302
 Google Scholar
Google Scholar
[29] Xiang Z Y, Luo Z J, Bi J, Jin S Y, Zhang Z Q, Lu C 2022 J. Phys. Condens. Matter 34 445302
 Google Scholar
Google Scholar
[30] Jin S Y, Sun W G, Chen B L, Kuang X Y, Lu H Y, Lu C 2021 J. Phys. Chem. A 125 4126
 Google Scholar
Google Scholar
[31] Lu C, Gong W, Li Q, Chen C 2020 J. Phys. Chem. Lett. 11 9165
 Google Scholar
Google Scholar
[32] Li W L, Chen T T, Chen W J, Li J, Wang L S, 2021 Nat. Commun. 12 6467
 Google Scholar
Google Scholar
[33] Zhang J, Dolg M 2015 Phys. Chem. Chem. Phys. 17 24173
 Google Scholar
Google Scholar
[34] Frisch M J, Trucks G W, Schlegel H B, et al. 2016 Gaussian 09 (Revision Ed. 01) (Wallingford, CT: Gaussian, Inc.
[35] Lu T, Chen F W 2012 J. Comput. Chem. 33 580
 Google Scholar
Google Scholar
[36] Humphrey W, Dalke A, Schulten K 1996 J. Mol. Graph. 14 33
 Google Scholar
Google Scholar
[37] Dolg M, Stoll H, Savin A, Preuss H 1989 Theor. Chim. Acta. 75 173
 Google Scholar
Google Scholar
[38] Peterson A, Kirk F, Detlev G, Erich S H, Michael D 2003 J. Chem. Phys. 119 11113
 Google Scholar
Google Scholar
[39] Binkley, Stephen J, Pople, John A, Hehre, Warren J 1980 J. Am. Chem. Soc. 102 939
 Google Scholar
Google Scholar
[40] Adamo C, Barone V 1999 J. Chem. Phys. 110 6158
 Google Scholar
Google Scholar
[41] Wadt W R, Hay P J 1985 J. Chem. Phys. 82 284
 Google Scholar
Google Scholar
[42] Krishnan R, Binkley J S, Seeger R, Pople J A 1980 J. Chem. Phys. 72 650
 Google Scholar
Google Scholar
[43] Tozer D J, Handy N C 1998 J. Chem. Phys. 109 10180
 Google Scholar
Google Scholar
[44] Alexandrova A N, Boldyrev A I, Zhai H J, Wang L S, Steiner E, Fowler P W 2003 J. Phys. Chem. A 107 1362
-
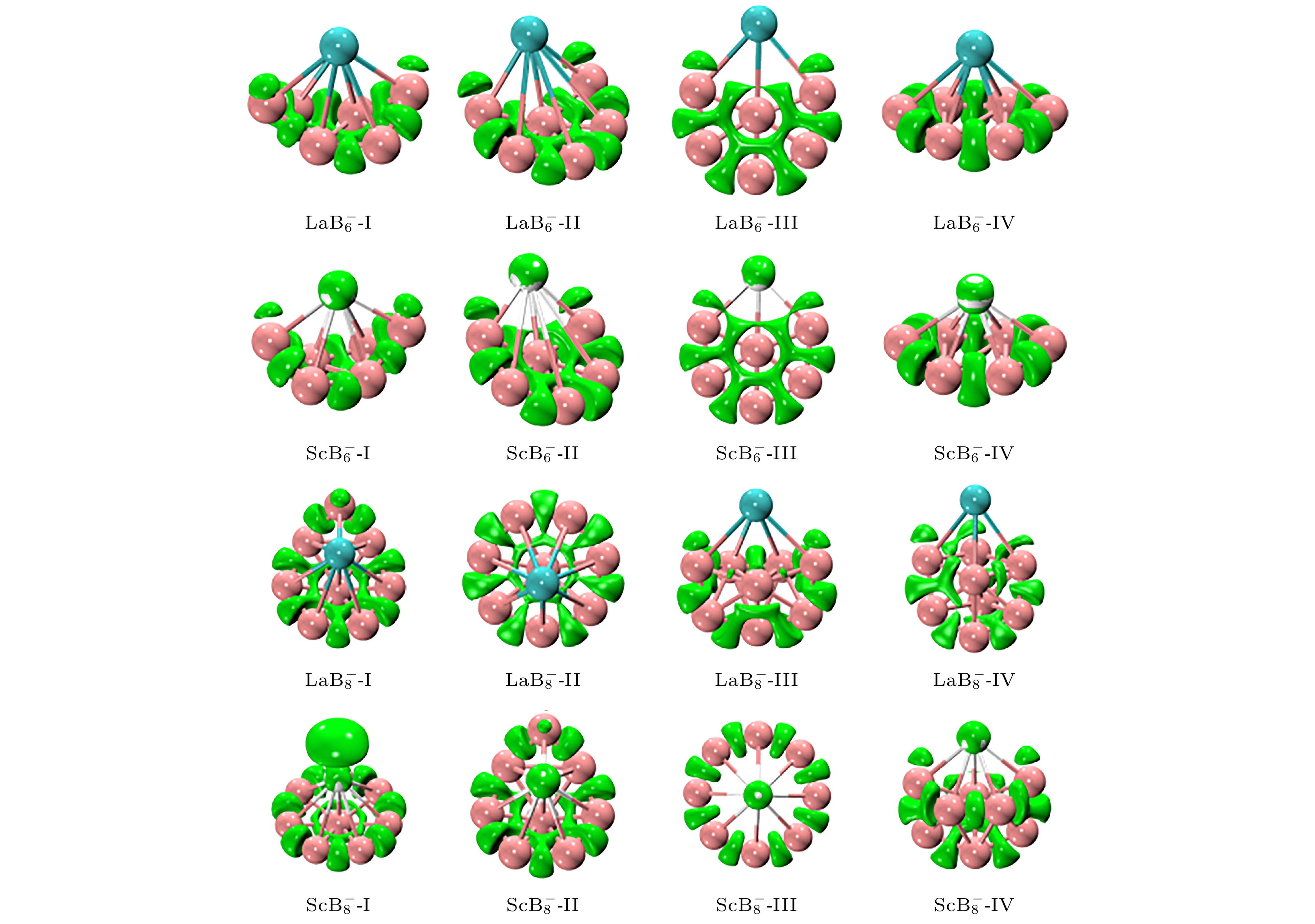
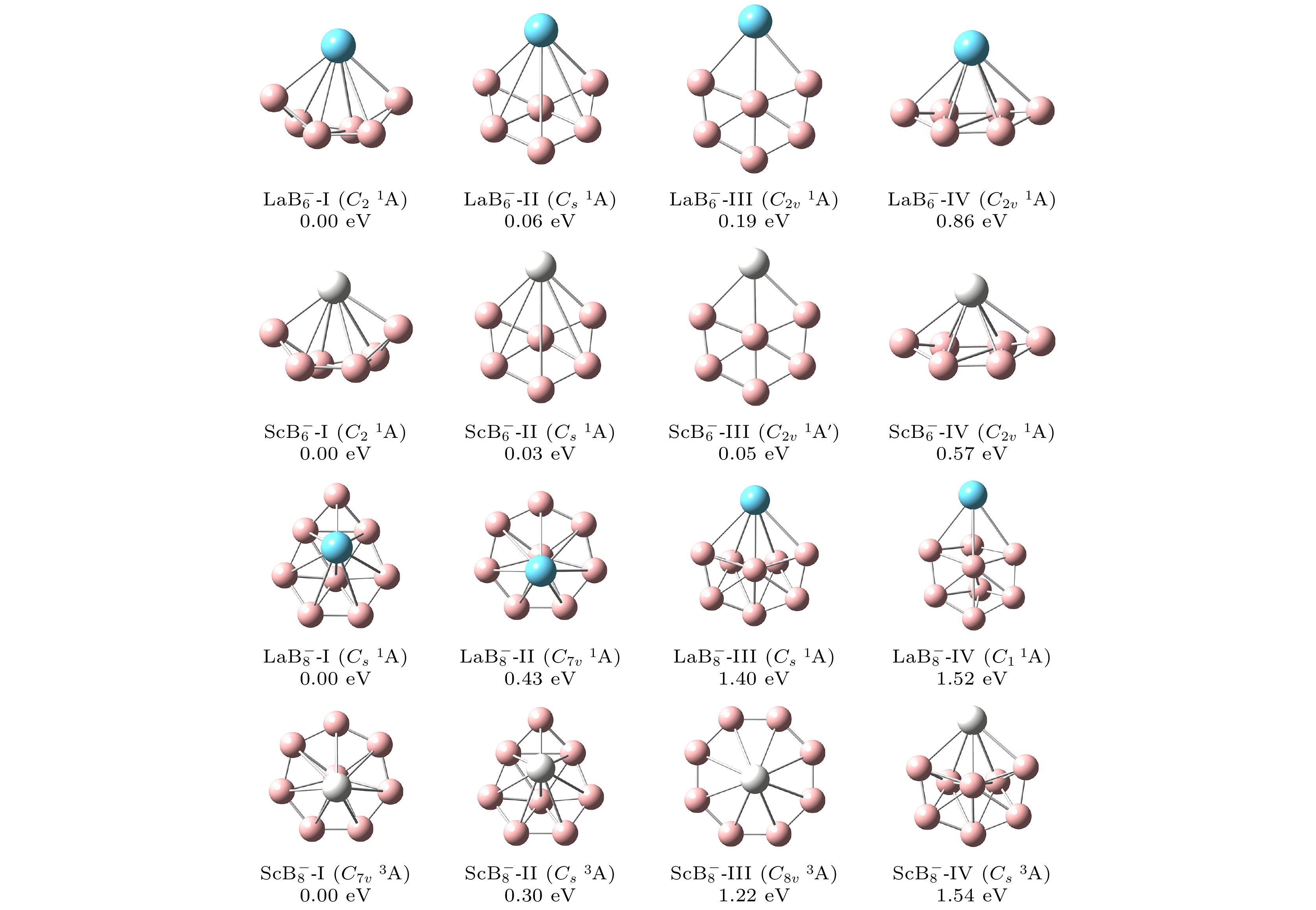
图 1 在PBE0/RE/SDD//B/6-311+G*理论水平下, $ {\text{REB}}_n^ - $ (RE = La, Sc; n = 6, 8) 团簇低能异构体的结构、点群对称性、相对能
Fig. 1. Structures, symmetry point group and relative energy (eV) of the lower-lying isomers for $ {\text{REB}}_n^ - $ (RE = La, Sc; n = 6, 8) clusters at the PBE0/RE/SDD//B/6-311+G* level of theory.
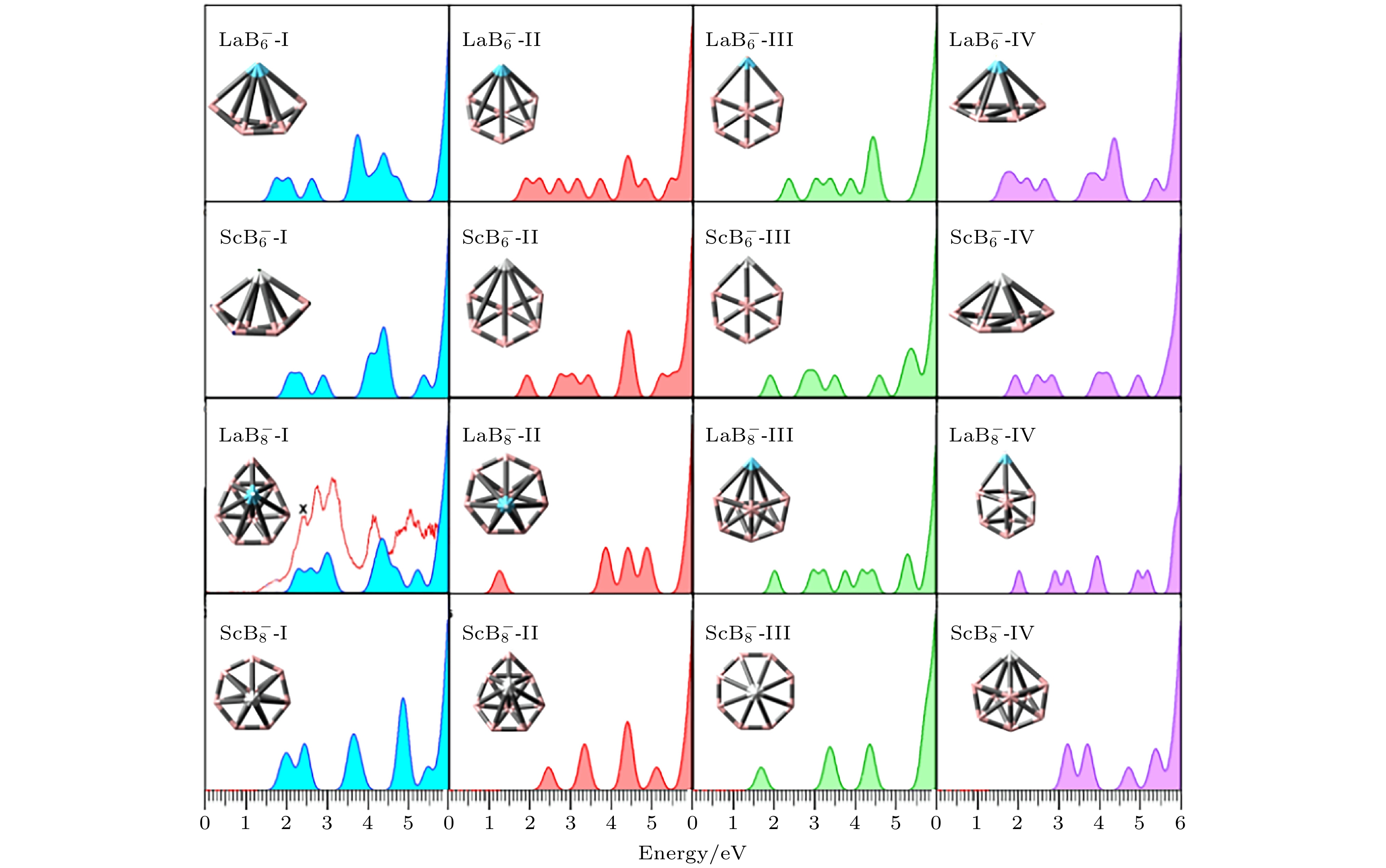
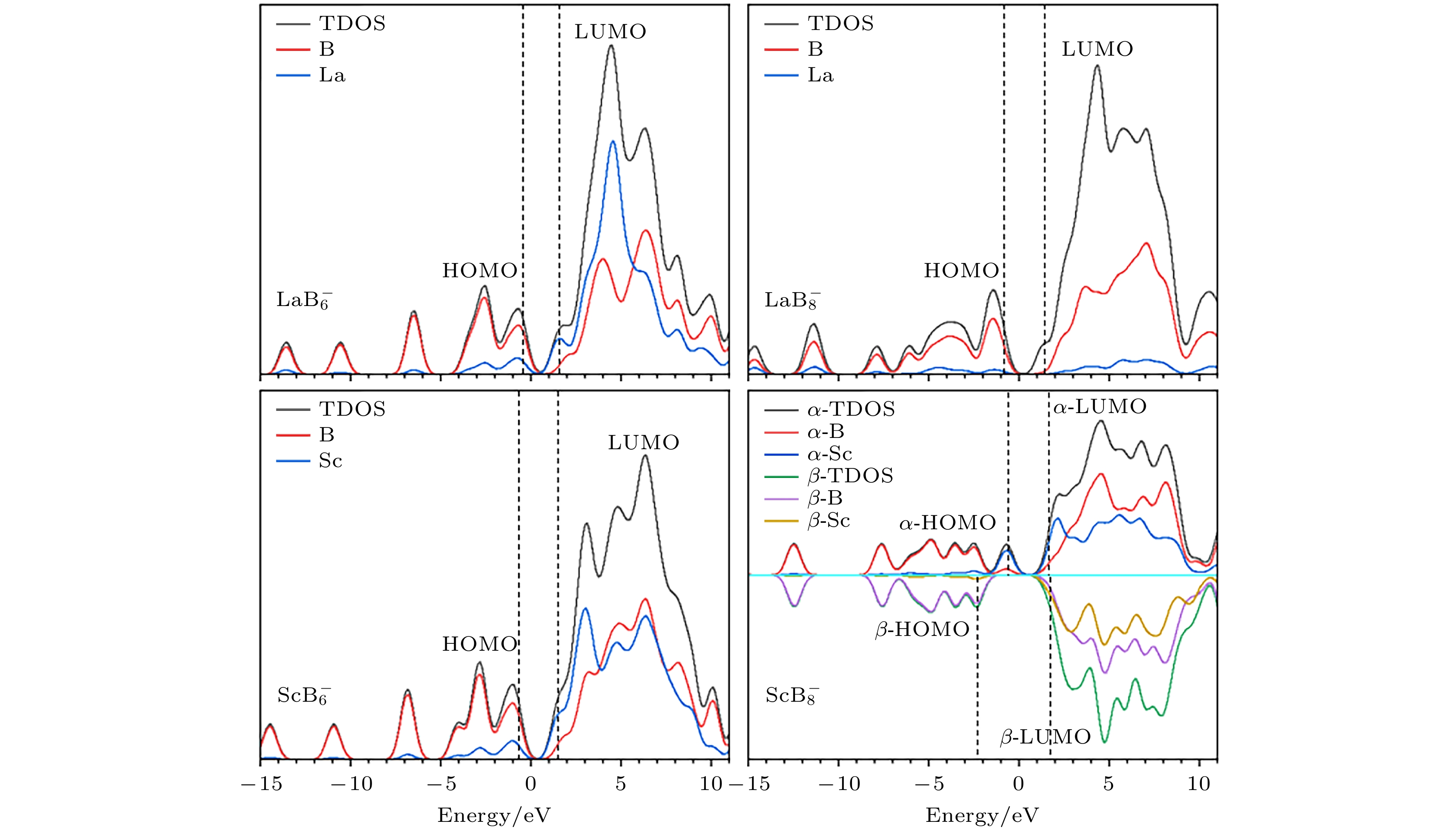
图 3 $ {\text{REB}}_n^ - $ (RE = La, Sc; n = 6, 8)团簇低能异构体的模拟光电子能谱图, $ {\text{LaB}}_{8}^ - $的实验光电子能谱来自于文献[32], 共同绘制在$ {\text{LaB}}_{8}^ - $-Ⅰ处用红色曲线表示
Fig. 3. Simulated PES spectra for low-lying isomers of $ {\text{REB}}_n^ - $ (RE = La, Sc; n = 6, 8) clusters, the experimental PES spectra of the anionic ground state structure of $ {\text{LaB}}_{8}^ - $ was obtained from Ref. [32].
-
[1] 张超江, 许洪光, 徐西玲, 郑卫军 2021 物理学报 70 023601
 Google Scholar
Google Scholar
Zhang C J, Xu H G, Xu X L, Zheng W J 2021 Acta Phys. Sin. 70 023601
 Google Scholar
Google Scholar
[2] Boustani I 1997 Phys. Rev. B 5 16426
[3] Kiran B, Bulusu S, Zhai H J, Yoo S, Zeng X C, Wang L S 2005 Proc. Natl. Acad. Sci. 102 961
 Google Scholar
Google Scholar
[4] 李世雄, 张正平, 隆正文, 秦水介 2017 物理学报 6 103102
 Google Scholar
Google Scholar
Li S X, Zhang Z P, Long Z W, Qin S J 2017 Acta Phys. Sin. 6 103102
 Google Scholar
Google Scholar
[5] Sergeeva A P, Popov I A, Piazza Z A, Li W L, Romanescu C, Wang L S, Boldyrev A I 2014 Acc. Chem. Res. 47 1349
 Google Scholar
Google Scholar
[6] Jian T, Chen X, Li S D, Boldyrev A I, Li J, Wang L S 2019 Chem. Soc. Rev. 48 3550
 Google Scholar
Google Scholar
[7] Wang L S 2016 Int. Rev. Phys. Chem. 35 69
 Google Scholar
Google Scholar
[8] Bai H, Chen T T, Chen Q, Zhao X Y, Zhang Y Y, Chen W J, Li W L, Cheng L F, Bai B, Cavanagh J, Huang W, Li S D, Li J, Wang L S 2019 Nanoscale 11 23286
 Google Scholar
Google Scholar
[9] 刘立仁, 雷雪玲, 陈杭, 祝恒江 2009 物理学报 58 5355
 Google Scholar
Google Scholar
Liu L R, Lei X L, Chen H, Zhu H J 2009 Acta Phys. Sin. 58 5355
 Google Scholar
Google Scholar
[10] Pham H T, Duong L V, Pham B Q, Nguyen M T 2013 Chem. Phys. Lett. 577 32
 Google Scholar
Google Scholar
[11] Zhai H J, Alexandrova A N, Birch K A, Boldyrev A I, Wang L S 2003 Angew. Chem. Int. Ed. 42 6004
 Google Scholar
Google Scholar
[12] Zhai H J, Zhao Y F, Li W L, Chen Q, Bai H, Hu H S, Piazza Z A, Tian W J, Lu H G, Wu Y B, Mu Y W, Wei G F, Liu Z P, Li J, Li S D, Wang L S 2014 Nature Chem. 6 727
 Google Scholar
Google Scholar
[13] Islas R, Heine T, Ito K, Schleyer P V, Merino G 2007 J. Am. Chem. Soc. 129 14767
 Google Scholar
Google Scholar
[14] Romanescu C, Galeev T R, Li W L, Boldyrev A I, Wang L S 2013 Acc. Chem. Res. 46 350
 Google Scholar
Google Scholar
[15] Saha R, Kar S, Pan S, Martínez-Guajardo G, Merino G, Chattaraj P K 2017 J. Phys. Chem. A 121 2971
 Google Scholar
Google Scholar
[16] 李世雄, 陈德良, 张正平, 隆正文 2020 物理学报 69 193101
 Google Scholar
Google Scholar
Li S X, Chen D L, Zhang Z P, Long Z W 2020 Acta Phys. Sin. 69 193101
 Google Scholar
Google Scholar
[17] Li H F, Wang H Q, Zhang J M, Qin L X, Zheng H, Zhang Y H 2024 Molecules 29 1692
 Google Scholar
Google Scholar
[18] 蒋贤明, 王怀谦, 曹宇, 孙之惠, 曹玉芳, 吴伟宾 2018 高等学校化学学报 39 1976
 Google Scholar
Google Scholar
Jiang X M, Wang H Q, Cao Y, Sun Z H, Cao Y F, Wu W B 2018 Chemical Journal of Chinese Universities 39 1976
 Google Scholar
Google Scholar
[19] Zheng H, Wang H Q, Li H F, Zhang J M, Zhang Y H, Qin L X, Mei X J, Jiang K L, Zeng J K, Zhang B, Wu W H 2024 Chem. Phys. 583 112321
 Google Scholar
Google Scholar
[20] Wen S H, Zhou J J, Zheng K Z, Bednarkiewicz A, Liu X G, Jin D Y 2018 Nat. Commun. 9 2415
 Google Scholar
Google Scholar
[21] Jiang L Y, Wang H Q, Li H F, Xie B, Zhang J M, Ji J Y 2023 Chem. Phys. 567 111819
 Google Scholar
Google Scholar
[22] Wang H Q, Li H F 2014 RSC Adv. 4 29782
 Google Scholar
Google Scholar
[23] Yi Z G, Luo Z C, Qin X, Chen Q S, Liu X G 2020 Acc. Chem. Res. 53 2692
 Google Scholar
Google Scholar
[24] Qin L X, Li H F, Xiao B W, Zhang J M, Zeng J K, Mei X J, Zhang Y H, Zheng H, Wang H Q 2023 Chem. Phys. 575 112064
 Google Scholar
Google Scholar
[25] Li W L, Chen T T, Xing D H, Chen X, Li J, Wang L S 2018 Proc. Natl. Acad. Sci. 115 E6972
[26] Robinson P J, Zhang X X, McQueen T, Bowen K H, Alexandrova A N 2017 J. Phys. Chem. A 121 1849
 Google Scholar
Google Scholar
[27] Chen T T, Li W L, Li J, Wang L S 2019 Chem. Sci. 10 2534
 Google Scholar
Google Scholar
[28] Zuo J N, Zhang L L, Chen B L, He K H, Dai W, Ding K W, Lu C 2024 J. Phys. Condens. Matter 36 015302
 Google Scholar
Google Scholar
[29] Xiang Z Y, Luo Z J, Bi J, Jin S Y, Zhang Z Q, Lu C 2022 J. Phys. Condens. Matter 34 445302
 Google Scholar
Google Scholar
[30] Jin S Y, Sun W G, Chen B L, Kuang X Y, Lu H Y, Lu C 2021 J. Phys. Chem. A 125 4126
 Google Scholar
Google Scholar
[31] Lu C, Gong W, Li Q, Chen C 2020 J. Phys. Chem. Lett. 11 9165
 Google Scholar
Google Scholar
[32] Li W L, Chen T T, Chen W J, Li J, Wang L S, 2021 Nat. Commun. 12 6467
 Google Scholar
Google Scholar
[33] Zhang J, Dolg M 2015 Phys. Chem. Chem. Phys. 17 24173
 Google Scholar
Google Scholar
[34] Frisch M J, Trucks G W, Schlegel H B, et al. 2016 Gaussian 09 (Revision Ed. 01) (Wallingford, CT: Gaussian, Inc.
[35] Lu T, Chen F W 2012 J. Comput. Chem. 33 580
 Google Scholar
Google Scholar
[36] Humphrey W, Dalke A, Schulten K 1996 J. Mol. Graph. 14 33
 Google Scholar
Google Scholar
[37] Dolg M, Stoll H, Savin A, Preuss H 1989 Theor. Chim. Acta. 75 173
 Google Scholar
Google Scholar
[38] Peterson A, Kirk F, Detlev G, Erich S H, Michael D 2003 J. Chem. Phys. 119 11113
 Google Scholar
Google Scholar
[39] Binkley, Stephen J, Pople, John A, Hehre, Warren J 1980 J. Am. Chem. Soc. 102 939
 Google Scholar
Google Scholar
[40] Adamo C, Barone V 1999 J. Chem. Phys. 110 6158
 Google Scholar
Google Scholar
[41] Wadt W R, Hay P J 1985 J. Chem. Phys. 82 284
 Google Scholar
Google Scholar
[42] Krishnan R, Binkley J S, Seeger R, Pople J A 1980 J. Chem. Phys. 72 650
 Google Scholar
Google Scholar
[43] Tozer D J, Handy N C 1998 J. Chem. Phys. 109 10180
 Google Scholar
Google Scholar
[44] Alexandrova A N, Boldyrev A I, Zhai H J, Wang L S, Steiner E, Fowler P W 2003 J. Phys. Chem. A 107 1362
计量
- 文章访问数: 3960
- PDF下载量: 77
- 被引次数: 0















 下载:
下载: