-
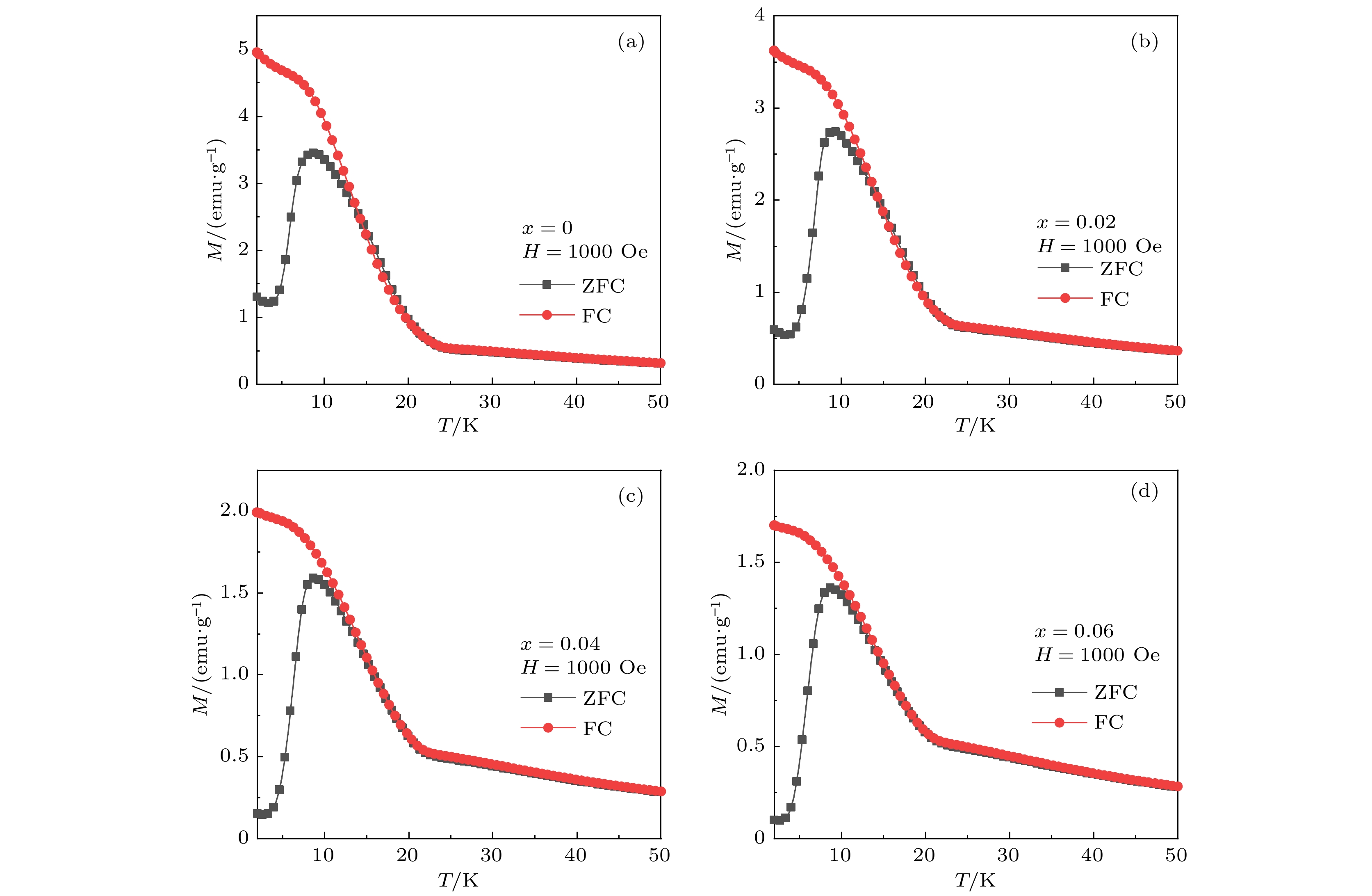
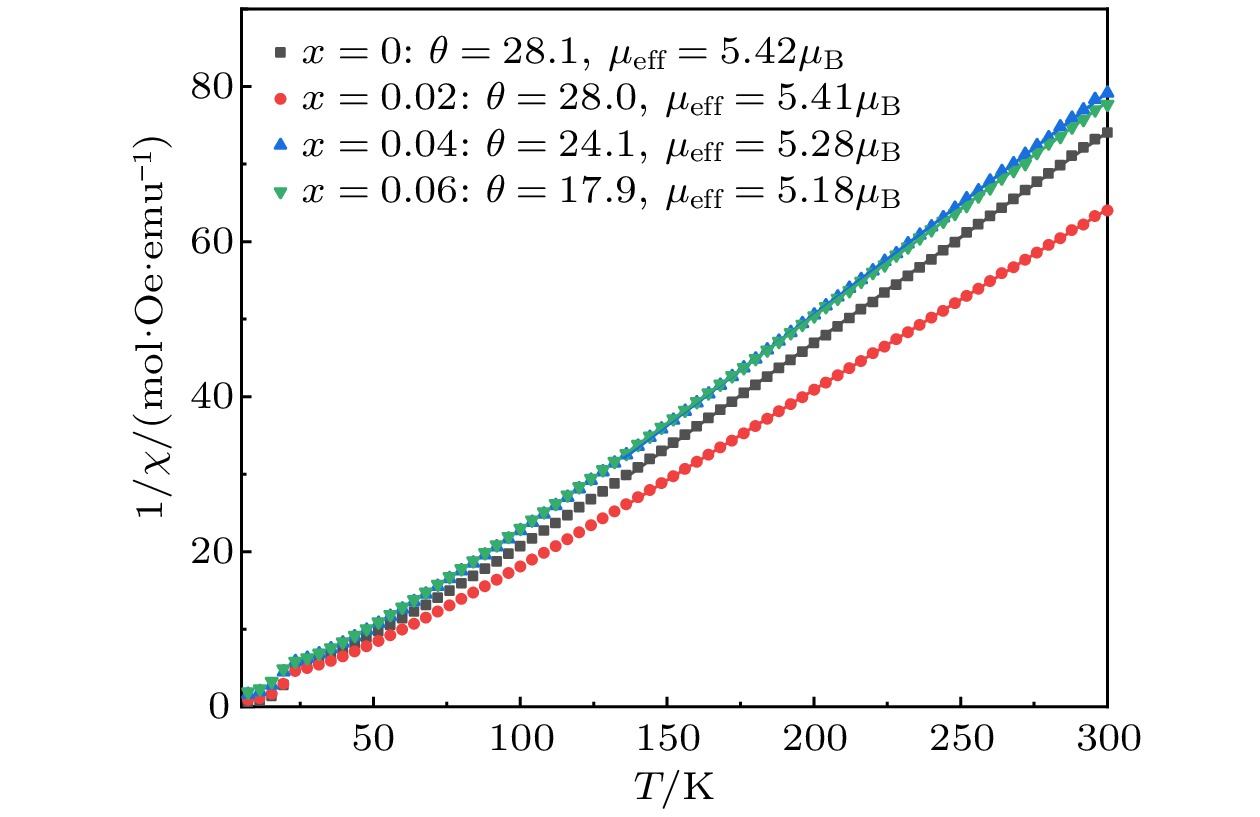
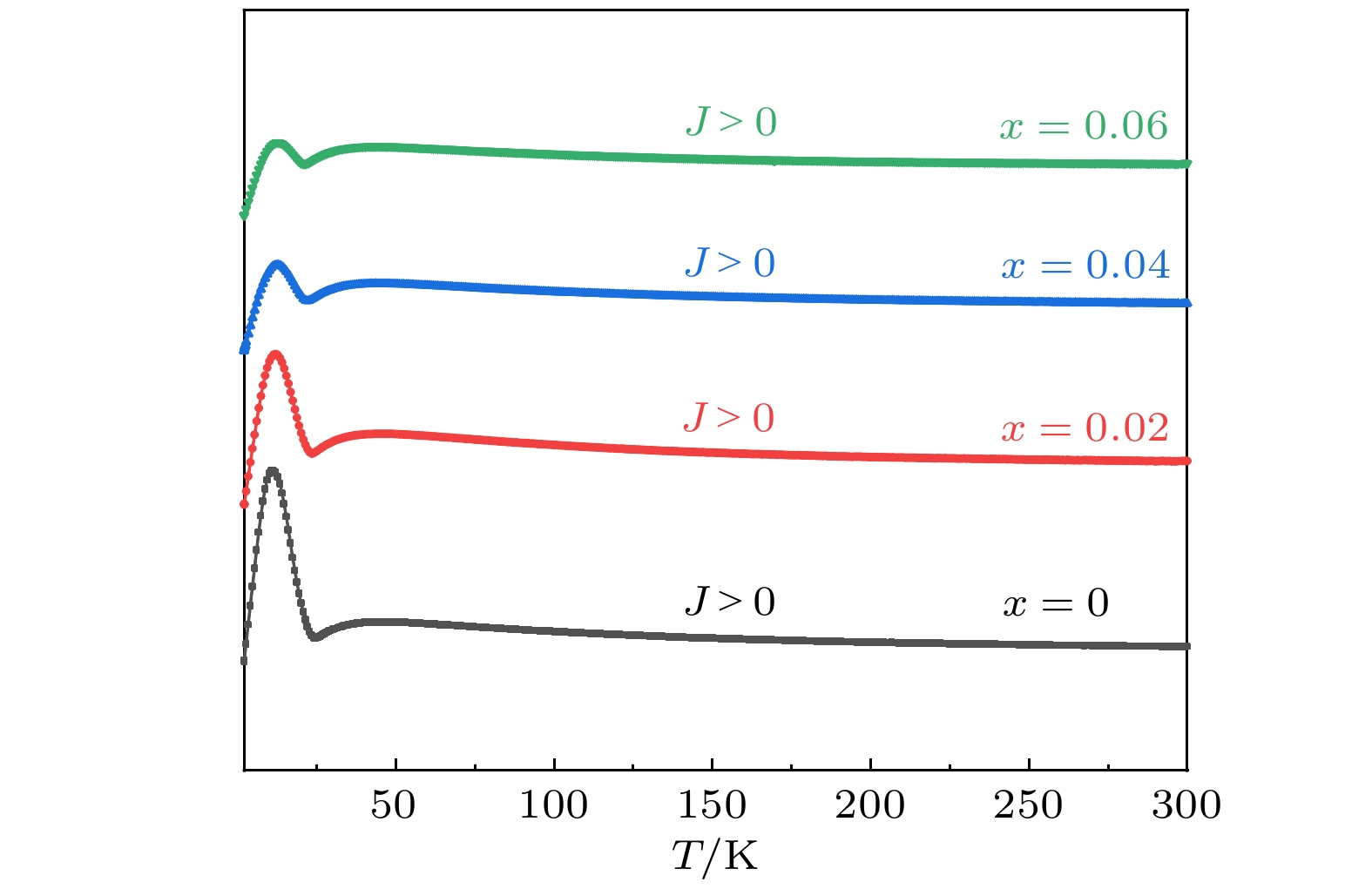
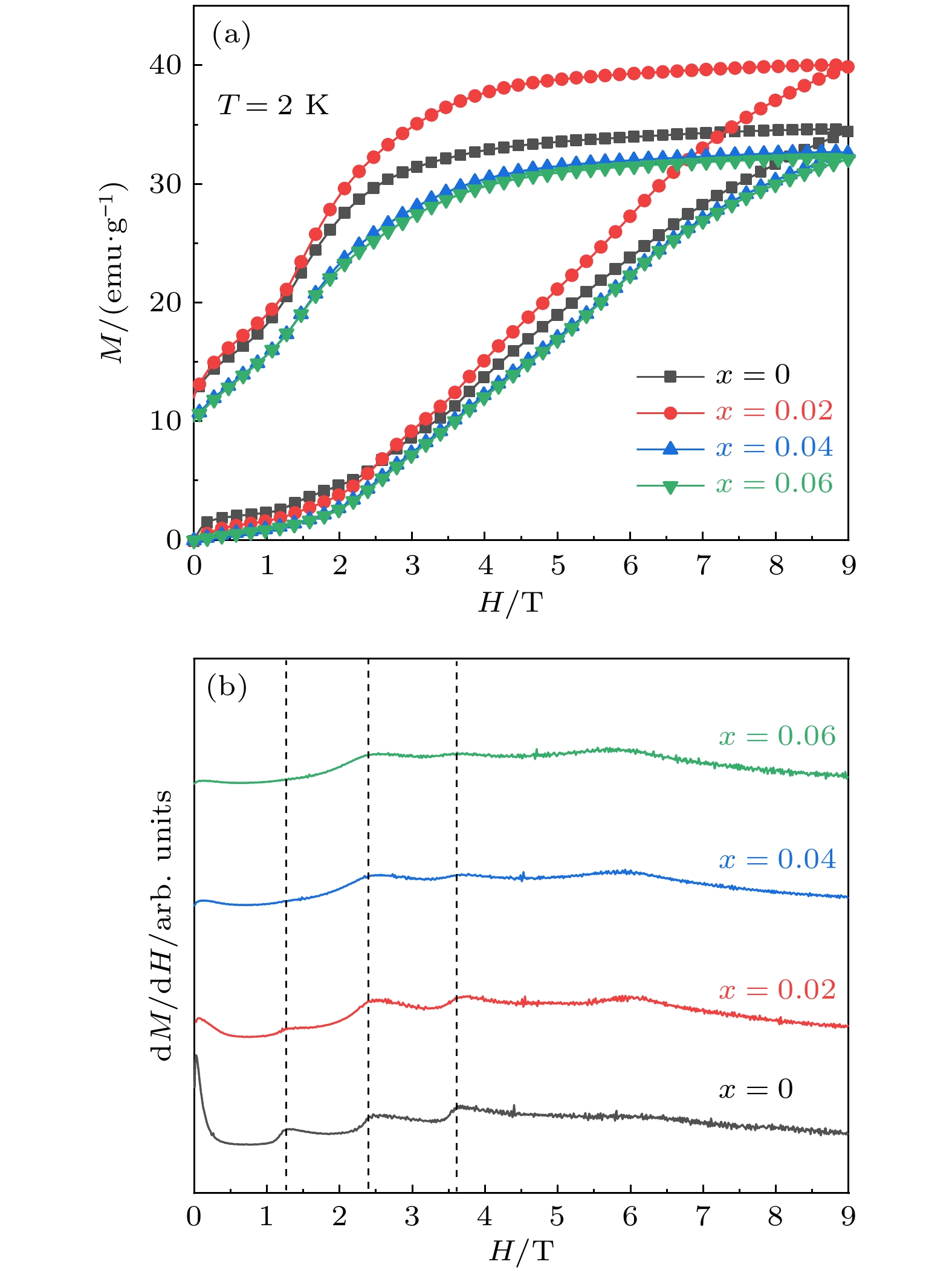
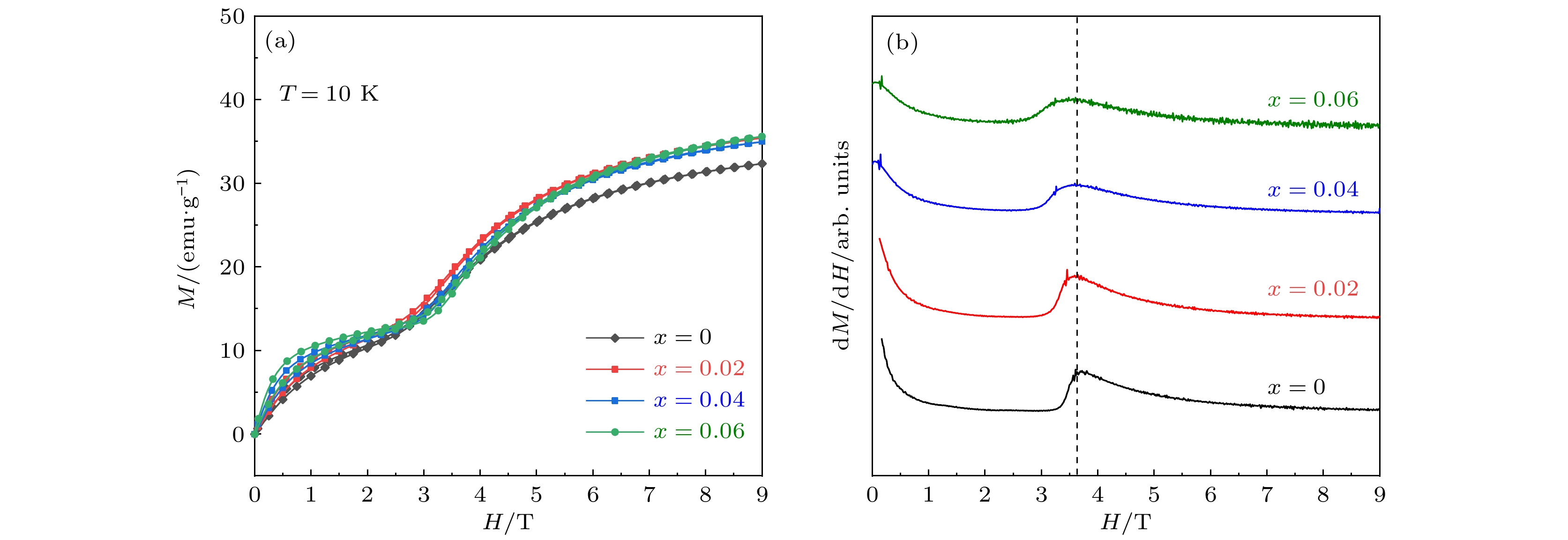
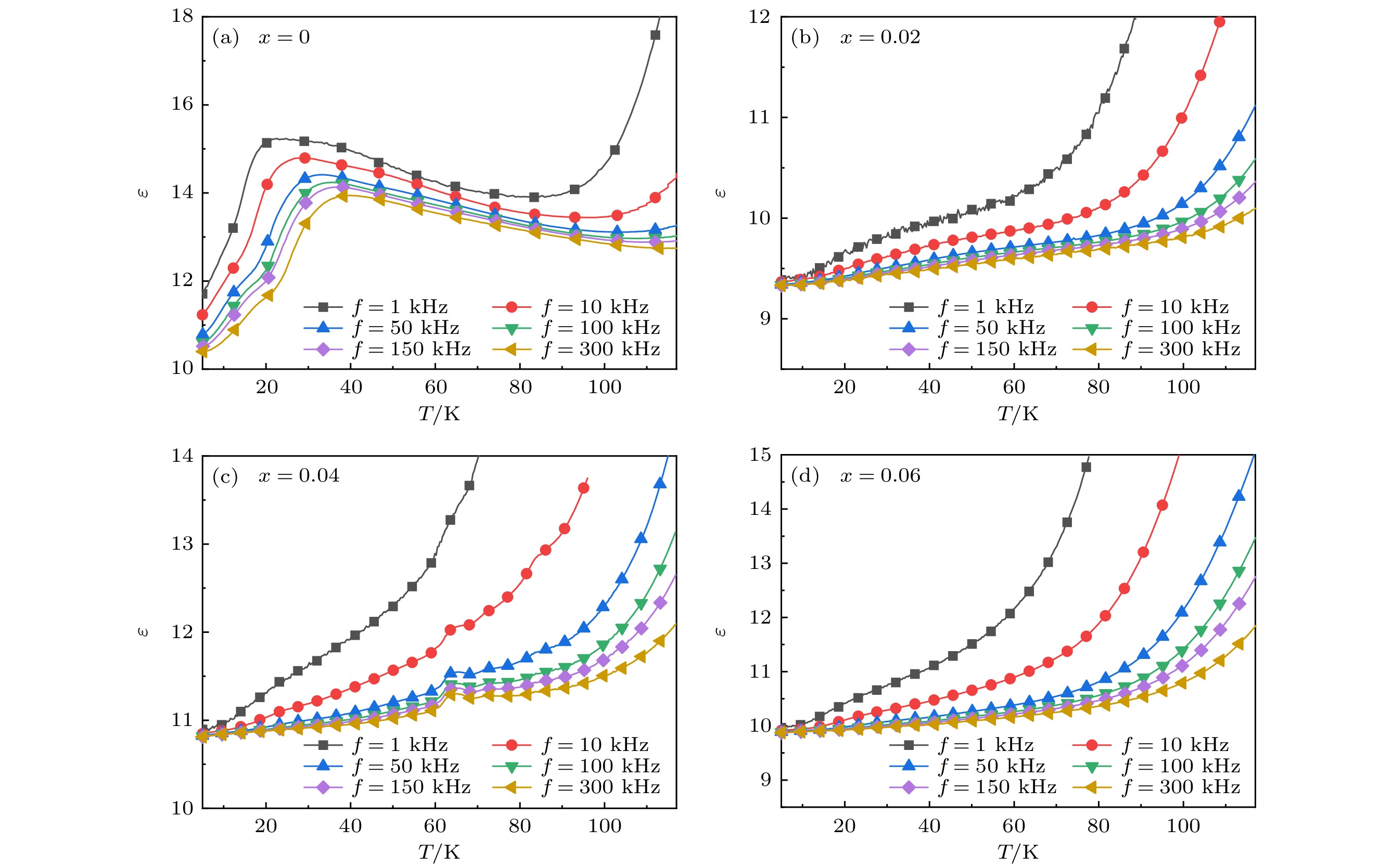
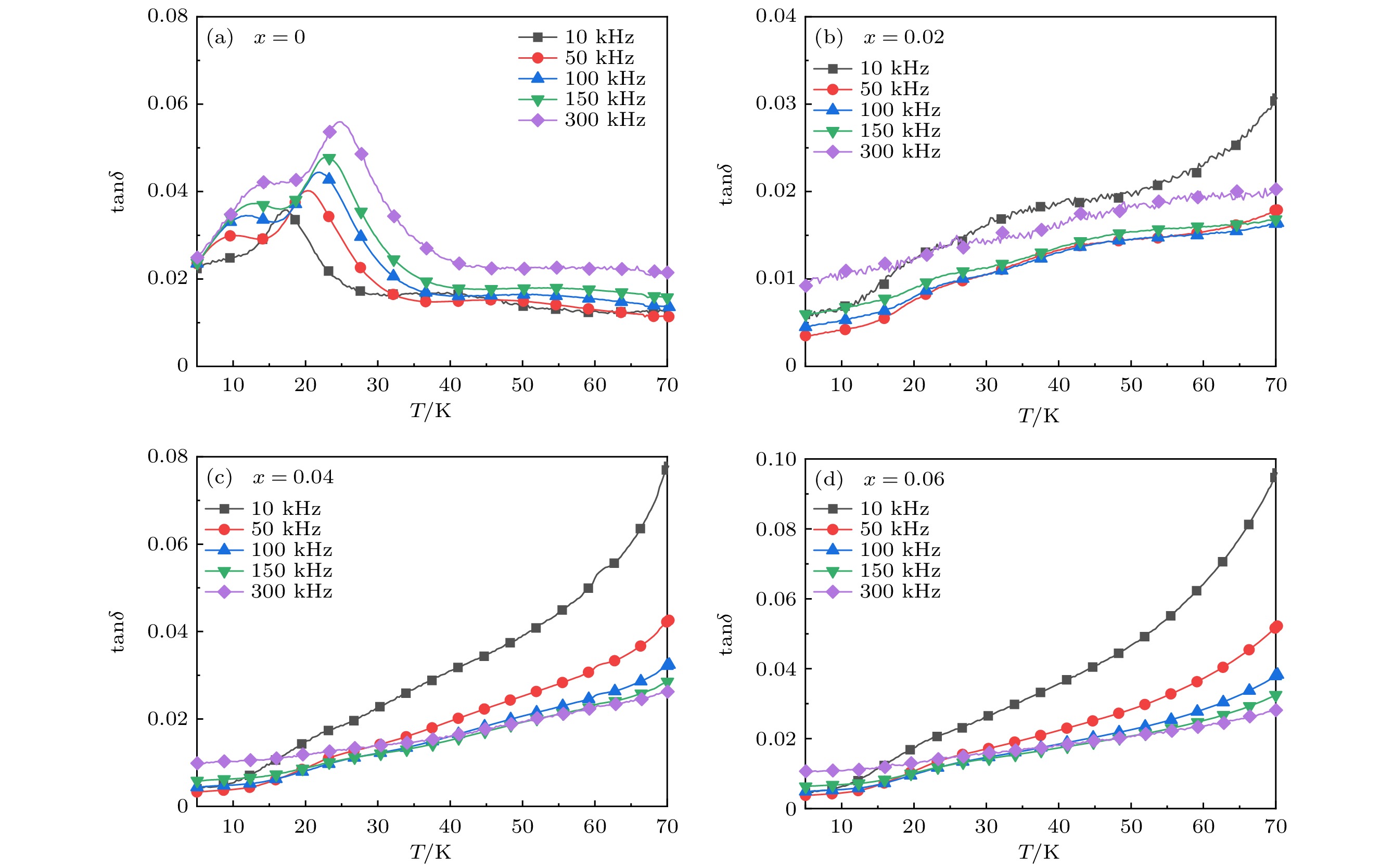
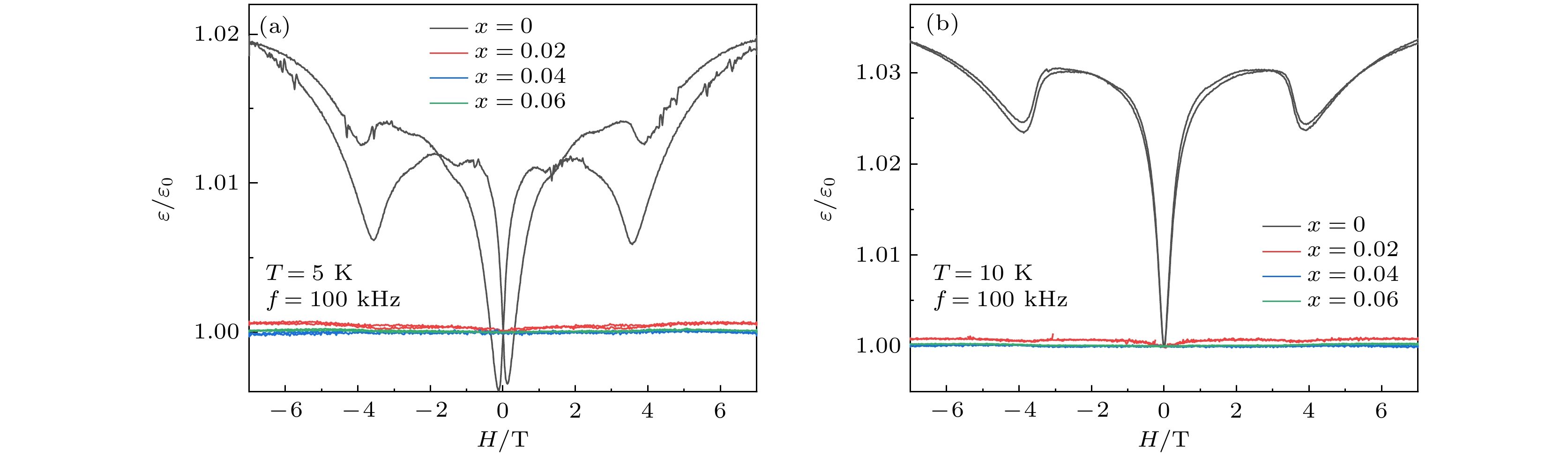
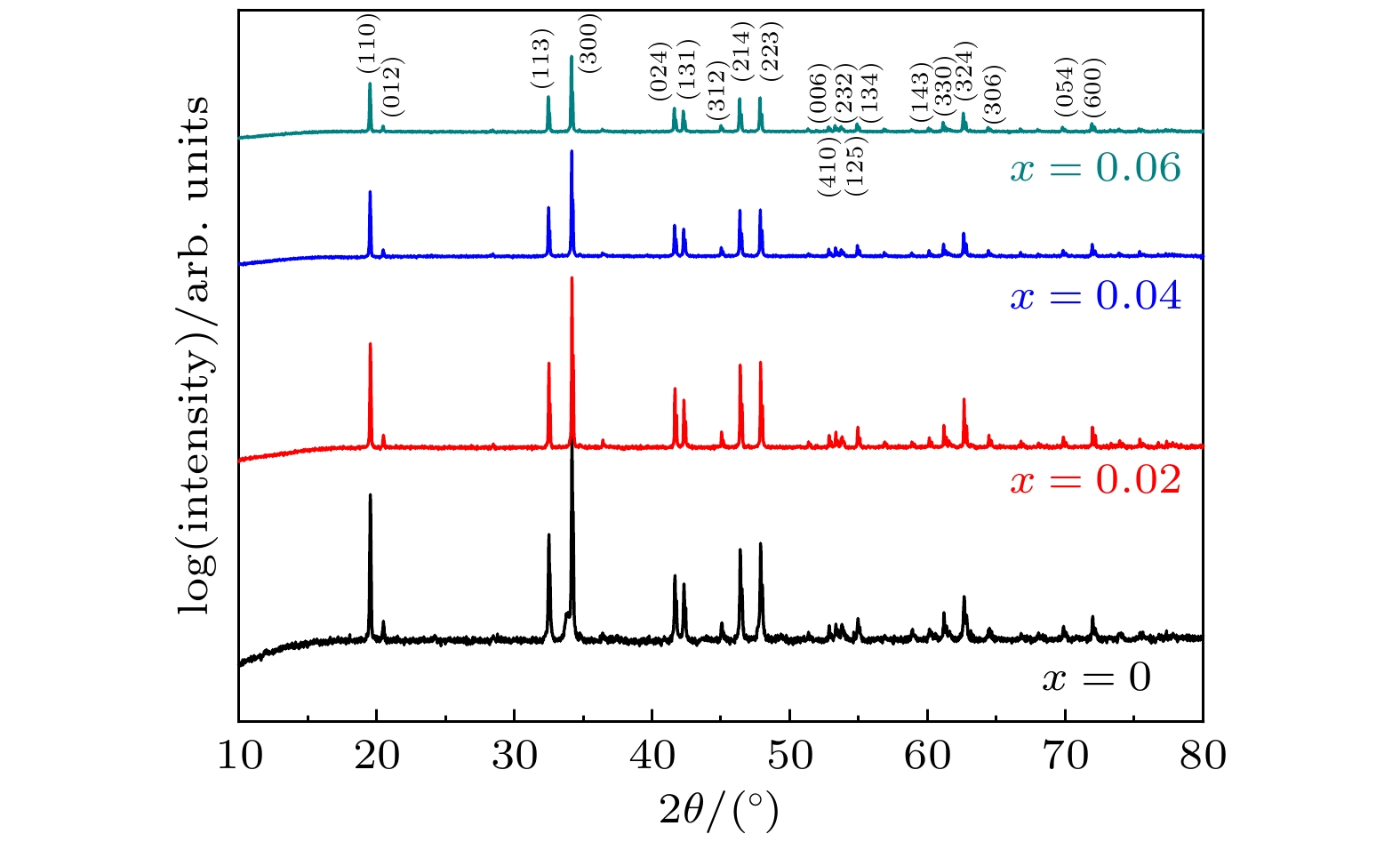
As a quasi-one-dimensional spin frustrated material, Ca3Co2O6 has a series of interesting physical properties such as low-temperature spin freezing and multiple magnetized steps due to its unique structure. The magnetic properties of Ca3Co2O6 mainly come from Co ions, and the doping of different elements at the Co site has a great effect on the magnetic structure of Ca3Co2O6. At present, the magnetic research of Ca3Co2O6 and its related compounds mainly focuses on exploring the influence of other elements replacing Co sites. For example, non-magnetic Sc3+ can dilute the intrachain ferromagnetic exchange, while the doping of magnetic ions Mn4+, Fe3+ or Cr3+ can inhibit the intrachain ferromagnetic interaction and enhance the antiferromagnetic interchain interaction. Doping Ti4+ ions, which are high-valence non-magnetic ions, not only dilutes the magnetic interaction of Ca3Co2O6, but also changes the valence state of cobalt ions. i.e. it can convert part of Co3+ ions into Co2+ ions. Therefore, comparing with other doped ions, their introduction may have a more significant effect on the magnetoelectric properties of Ca3Co2O6. In this study, a series of Ca3Co2–xTixO6 (x = 0, 0.02, 0.04, 0.06) polycrystalline samples is prepared by sol-gel method. Their magnetic, dielectric and magnetodielectric properties are measured. The XRD patterns show that a small number of Ti4+ ions do not change the crystal structure of Ca3Co2O6. Due to the destruction of the long-range ferromagnetic correlation of Ca3Co2O6 by non-magnetic Ti4+ ions, the ferromagnetic interaction is inhibited to some extent. Because Ti4+ ions are non-magnetic ions, they cannot form antiferromagnetic coupling with Co ions, resulting in the decrease of the Curie-Weiss temperature(θ). The positive θ value and exchange constant still indicate that the ferromagnetic interaction is dominant in Ti4+ doped Ca3Co2–xTixO6 (x = 0, 0.02, 0.04, 0.06) samples. The substitution of non-magnetic ions Ti4+ for Co3+ ions also makes the effective magnetic moment of Ca3Co2–xTixO6 (x = 0, 0.02, 0.04, 0.06) monotonically decrease from μeff = 5.42μB for x = 0 to μeff = 5.18μB for x = 0.06. Accompanying the introduction of Ti4+ ions, the spin frustration of Ca3Co2O6 is released partly, thus gradually fading the magnetization steps of Ca3Co2O6. As the Ca3Co2O6 is a typical magnetodielectric material, the released spin frustration in Ti4+ doped samples and the variation of the subtle magnetic structure exert a large influence on the magnetodielectric coupling effect of Ca3Co2–xTixO6 (x = 0, 0.02, 0.04, 0.06) compounds.
-
Keywords:
- Ca3Co2O6 /
- magnetization step /
- magneto dielectric effect
[1] Gong G S, Guo J J, Ma Y M, Zhang YP, Wang YQ, Su Y L 2019 J. Magn. Magn. Mater. 482 323
 Google Scholar
Google Scholar
[2] Kudasov Y B, Korshunov A S, Pavlov V N, Maslov D A 2010 J. Low. Temp. Phys. 159 76
 Google Scholar
Google Scholar
[3] Gong G S, Wang M H, Li Z, Duan Y R, Zuo Y Y, Zhou J, Wang Y Q, Su Y L 2024 J. Magn. Magn. Mater. 590 171653
 Google Scholar
Google Scholar
[4] Gong G S, Xu L M, Bai Y M, Wang Y Q, Yuan S L, Liu Y, Tian Z M 2021 Phys. Rev. Mater. 5 034405
 Google Scholar
Google Scholar
[5] Zhou J, Gong G S, Duan Y R, Wang L C, Zuo Y Y, Wang Y Q, Su Y L 2023 J. Solid State Chem. 323 124021
 Google Scholar
Google Scholar
[6] Xu L M, Gong G S, Zhao C W, Song X X, Yuan S L, Tian Z M 2020 J. Phys. Chem. C 124 22656
 Google Scholar
Google Scholar
[7] Ashtar M, Guo J J, Wan Z T, Wang Y Q, Gong G S, Liu Y, Su Y L, Tian Z M 2020 Inorg. Chem. 59 5368
 Google Scholar
Google Scholar
[8] Maignan A, Michel C, Masset A C, Martin C, Raveau B 2000 Eur. Phys. J. B 15 657
 Google Scholar
Google Scholar
[9] Fjellvåg H, Gulbrandsen E, Aasland S, Olsen A, Hauback B C 1996 J. Solid. State. Chem. 124 190
 Google Scholar
Google Scholar
[10] Bellido N, Simon C, Maignan A 2008 Phys. Rev. B 77 054430
 Google Scholar
Google Scholar
[11] Kudasov Y B, Korshunov A S, Pavlov V N, Maslov D A 2011 Phys. Rev. B 83 092404
 Google Scholar
Google Scholar
[12] Takubo K, Mizokawa T, Hirata S, Son J, Fujimori A, Topwal D, Sarma D D, Rayaprol S, Sampathkumaran E 2005 Phys. Rev. B 71 073406
 Google Scholar
Google Scholar
[13] Shimizu Y, Horibe M, Nanba H, Takami T, Itoh M 2010 Phys. Rev. B 82 094430
 Google Scholar
Google Scholar
[14] Allodi G, Santini P, Carretta S, Agrestini S, Mazzoli C, Bombardi A, Lees M R, Renzi R D 2014 Phys. Rev. B 89 104401
 Google Scholar
Google Scholar
[15] Allodi G, Renzi R D, Agrestini S, Mazzoli C, Lees M R 2011 Phys. Rev. B 83 104408
 Google Scholar
Google Scholar
[16] Hardy V, Lambert S, Lees M R, Paul D M 2003 Phys. Rev. B 68 014424
 Google Scholar
Google Scholar
[17] Hardy V, Flahaut D, Lees M, Petrenko O 2004 Phys. Rev. B 70 214439
 Google Scholar
Google Scholar
[18] Burnus T, Hu Z, Haverkort M W, Cezar J C, Flahaut D, Hardy V, Maignan A, Brookes N B, Tanaka A, Hsieh H H, Lin H, Chen C T, Tjeng L H 2006 Phys. Rev. B 74 245111
 Google Scholar
Google Scholar
[19] Agrestini S, Mazzoli C, Bombardi A, Lees M R 2008 Phys. Rev. B 77 140403
 Google Scholar
Google Scholar
[20] Bisht G S, Pal D 2022 J. Phys-Condens. Mat. 34 285803
 Google Scholar
Google Scholar
[21] Agrestini S, Chapon L C, Daoud-Aladine A, Schefer J, Gukasov A, Mazzoli C, Lees M R, Petrenko O A 2008 Phys. Rev. L 101 097207
 Google Scholar
Google Scholar
[22] Kamiya Y, Batista C D 2012 Phys. Rev. L 109 067204
 Google Scholar
Google Scholar
[23] Flahaut D, Maignan A, Hébert S, Martin C, Retoux R, Hardy V 2004 Phys. Rev. B 70 094418
 Google Scholar
Google Scholar
[24] Hervoches C H, Fredenborg V M, Kjekshus A, Fjellvåg H, Hauback B C 2007 J. Solid. State. Chem. 180 834
 Google Scholar
Google Scholar
[25] Das R, Dang N T, Kalappattil V, Madhogaria R P, Kozlenko D P, Kichanov S E, Lukin E V, A Rutkaukas V, Nguyen T P T, Thao L T P, Bingham N S, Srikanth H, Phan M H 2021 J. Alloys. Compd. 851 156897
 Google Scholar
Google Scholar
[26] Gong G S, Shi C F, Zerihun G, Guo J J, Wang Y Q, Qiu Y, Su Y L 2020 Mater. Res. Bull. 130 110934
 Google Scholar
Google Scholar
[27] Kim J W, Mun E D, Ding X, Hansen A, Jaime M, Harrison N, Yi H T, Chai Y, Sun Y, Cheong S W, Zapf V S 2018 Phys. Rev. B 98 024407
 Google Scholar
Google Scholar
[28] Duan Y R, Gong G S, Wang M H, Zhou J, Li Z, Zuo Y Y, Wang L C, Wang Y Q, Su Y L, Zhang H J 2023 Physics B 671 415429
 Google Scholar
Google Scholar
[29] Gong G S, Wang Y Q, Su Y L, Liu D W, Zerihun G, Qiu Y 2018 Mater. Res. Bull. 99 419
 Google Scholar
Google Scholar
-
-
[1] Gong G S, Guo J J, Ma Y M, Zhang YP, Wang YQ, Su Y L 2019 J. Magn. Magn. Mater. 482 323
 Google Scholar
Google Scholar
[2] Kudasov Y B, Korshunov A S, Pavlov V N, Maslov D A 2010 J. Low. Temp. Phys. 159 76
 Google Scholar
Google Scholar
[3] Gong G S, Wang M H, Li Z, Duan Y R, Zuo Y Y, Zhou J, Wang Y Q, Su Y L 2024 J. Magn. Magn. Mater. 590 171653
 Google Scholar
Google Scholar
[4] Gong G S, Xu L M, Bai Y M, Wang Y Q, Yuan S L, Liu Y, Tian Z M 2021 Phys. Rev. Mater. 5 034405
 Google Scholar
Google Scholar
[5] Zhou J, Gong G S, Duan Y R, Wang L C, Zuo Y Y, Wang Y Q, Su Y L 2023 J. Solid State Chem. 323 124021
 Google Scholar
Google Scholar
[6] Xu L M, Gong G S, Zhao C W, Song X X, Yuan S L, Tian Z M 2020 J. Phys. Chem. C 124 22656
 Google Scholar
Google Scholar
[7] Ashtar M, Guo J J, Wan Z T, Wang Y Q, Gong G S, Liu Y, Su Y L, Tian Z M 2020 Inorg. Chem. 59 5368
 Google Scholar
Google Scholar
[8] Maignan A, Michel C, Masset A C, Martin C, Raveau B 2000 Eur. Phys. J. B 15 657
 Google Scholar
Google Scholar
[9] Fjellvåg H, Gulbrandsen E, Aasland S, Olsen A, Hauback B C 1996 J. Solid. State. Chem. 124 190
 Google Scholar
Google Scholar
[10] Bellido N, Simon C, Maignan A 2008 Phys. Rev. B 77 054430
 Google Scholar
Google Scholar
[11] Kudasov Y B, Korshunov A S, Pavlov V N, Maslov D A 2011 Phys. Rev. B 83 092404
 Google Scholar
Google Scholar
[12] Takubo K, Mizokawa T, Hirata S, Son J, Fujimori A, Topwal D, Sarma D D, Rayaprol S, Sampathkumaran E 2005 Phys. Rev. B 71 073406
 Google Scholar
Google Scholar
[13] Shimizu Y, Horibe M, Nanba H, Takami T, Itoh M 2010 Phys. Rev. B 82 094430
 Google Scholar
Google Scholar
[14] Allodi G, Santini P, Carretta S, Agrestini S, Mazzoli C, Bombardi A, Lees M R, Renzi R D 2014 Phys. Rev. B 89 104401
 Google Scholar
Google Scholar
[15] Allodi G, Renzi R D, Agrestini S, Mazzoli C, Lees M R 2011 Phys. Rev. B 83 104408
 Google Scholar
Google Scholar
[16] Hardy V, Lambert S, Lees M R, Paul D M 2003 Phys. Rev. B 68 014424
 Google Scholar
Google Scholar
[17] Hardy V, Flahaut D, Lees M, Petrenko O 2004 Phys. Rev. B 70 214439
 Google Scholar
Google Scholar
[18] Burnus T, Hu Z, Haverkort M W, Cezar J C, Flahaut D, Hardy V, Maignan A, Brookes N B, Tanaka A, Hsieh H H, Lin H, Chen C T, Tjeng L H 2006 Phys. Rev. B 74 245111
 Google Scholar
Google Scholar
[19] Agrestini S, Mazzoli C, Bombardi A, Lees M R 2008 Phys. Rev. B 77 140403
 Google Scholar
Google Scholar
[20] Bisht G S, Pal D 2022 J. Phys-Condens. Mat. 34 285803
 Google Scholar
Google Scholar
[21] Agrestini S, Chapon L C, Daoud-Aladine A, Schefer J, Gukasov A, Mazzoli C, Lees M R, Petrenko O A 2008 Phys. Rev. L 101 097207
 Google Scholar
Google Scholar
[22] Kamiya Y, Batista C D 2012 Phys. Rev. L 109 067204
 Google Scholar
Google Scholar
[23] Flahaut D, Maignan A, Hébert S, Martin C, Retoux R, Hardy V 2004 Phys. Rev. B 70 094418
 Google Scholar
Google Scholar
[24] Hervoches C H, Fredenborg V M, Kjekshus A, Fjellvåg H, Hauback B C 2007 J. Solid. State. Chem. 180 834
 Google Scholar
Google Scholar
[25] Das R, Dang N T, Kalappattil V, Madhogaria R P, Kozlenko D P, Kichanov S E, Lukin E V, A Rutkaukas V, Nguyen T P T, Thao L T P, Bingham N S, Srikanth H, Phan M H 2021 J. Alloys. Compd. 851 156897
 Google Scholar
Google Scholar
[26] Gong G S, Shi C F, Zerihun G, Guo J J, Wang Y Q, Qiu Y, Su Y L 2020 Mater. Res. Bull. 130 110934
 Google Scholar
Google Scholar
[27] Kim J W, Mun E D, Ding X, Hansen A, Jaime M, Harrison N, Yi H T, Chai Y, Sun Y, Cheong S W, Zapf V S 2018 Phys. Rev. B 98 024407
 Google Scholar
Google Scholar
[28] Duan Y R, Gong G S, Wang M H, Zhou J, Li Z, Zuo Y Y, Wang L C, Wang Y Q, Su Y L, Zhang H J 2023 Physics B 671 415429
 Google Scholar
Google Scholar
[29] Gong G S, Wang Y Q, Su Y L, Liu D W, Zerihun G, Qiu Y 2018 Mater. Res. Bull. 99 419
 Google Scholar
Google Scholar
Catalog
Metrics
- Abstract views: 3428
- PDF Downloads: 180
- Cited By: 0














 DownLoad:
DownLoad: