-
单片层氧化石墨烯由于其优异的物理化学性能, 在离子和分子筛选、水脱盐和净化、气体分离、生物传感、质子导体、锂电池和超级电容等领域有巨大的潜在应用. 然而普遍采用的Hummers法等化学、物理方法制备的氧化石墨烯是一种亚稳态材料. 其最终形态的物理化学性能的转变与调控至关重要, 亟需系统研究. 本文采用恒温处理方法对氧化石墨烯亚稳态的转变进行调控, 利用X射线光电子吸收谱、傅里叶红外吸收谱、扫描电子显微镜等方法检测氧化石墨烯含氧基团的含量、类型和形貌与温度的变化关系, 并利用Zeta电位、紫外吸收谱、拉力测试分析温度对氧化石墨烯在转变过程中的溶液悬浮稳定性、光子能带、拉伸强度等性能的影响. 所得定量测试结果发现, 氧化石墨烯亚稳态的转变过程中存在环氧减少、羟基增加, 以及整体含氧量下降的现象, 而在此过程中氧化石墨烯的单片层形貌并无明显变化. 但是这种结构的转变使得悬浮液黏稠度和亲水性大幅度增强, 能带减小和拉伸强度增强效应明显. 而当转变过程足够长时, 氧化石墨烯的亲水性转而下降, 并出现沉淀现象, 表明羟基之间进一步发生了脱水转变. 另外, 本文还分析了恒温处理的时间、悬浮液的浓度对这种转变过程的影响. 相关研究结果有利于理解亚稳态氧化石墨烯悬浮液随温度变化的性能转变, 对氧化石墨烯具体应用有一定参考价值.Monolayer of graphene oxide has great potential applications in ion and molecular screening, desalination and purification of water, gas separation, biosensing, proton conductors, lithium batteries, super capacitors and other fields, due to its excellent physical and chemical properties. However, the graphene oxide prepared by chemical and physical methods, such as the commonly used Hummers method is a metastable material. The transformation and regulation of the physical and chemical properties of the final morphology are essential, and systematic research is urgently needed. In this paper, the thermostat treatment method is used to control the metastable transformation of graphene oxide. The relationship of content, type, and morphology with temperature of oxygen-containing graphene oxide are detected by X-ray photoelectron absorption spectroscopy, Fourier infrared absorption spectroscopy, scanning electron microscope, etc.; and the effects of temperature on the stability of solution suspension stability, photon energy band and tensile strength of graphene oxide in the transformation process are analyzed by using Zeta potential, ultraviolet absorption spectrum, tensile force. The quantitative test results show that there exists a phenomenon in which the epoxy phase decreases, the hydroxyl group increases and the overall oxygen content decreases in the metastable transition process of graphene oxide, and the monolayer morphology of graphene oxide does not change significantly in this process. This structural transformation, however, greatly enhances the viscosity and hydrophilicity of the suspension, and remarkably reduces the energy band and considerably raises the tensile strength enhancement effect. When the transformation process is long enough, the hydrophilicity of the graphene oxide will decrease and precipitate. It is indicated that a further dehydration transition occurs between the hydroxyl groups. In addition, in the paper we also analyze the effect of constant temperature treatment time and concentration of suspension on this transformation process. The relevant research results are helpful in understanding the performance change of metastable graphene oxide suspension with temperature, and have certain reference value for the specific application of graphene oxide.
-
Keywords:
- graphene oxide /
- metastable /
- group transformation
[1] McAllister M J, Li J L, Adamson D H, Schniepp H C, Abdala A A, Liu J, Herrera-Alonso M, Milius D L, Car R, Prud'homme R K, Aksay I A 2007 Chem. Mater. 19 4396
 Google Scholar
Google Scholar
[2] 刘学文, 朱重阳, 董辉, 徐峰, 孙立涛 2016 物理学报 65 118802
 Google Scholar
Google Scholar
Liu X W, Zhu C Y, Dong H, Xu F, Sun L T 2016 Acta Phys. Sin. 65 118802
 Google Scholar
Google Scholar
[3] 林文强, 徐斌, 陈亮, 周峰, 陈均朗 2016 物理学报 65 133102
 Google Scholar
Google Scholar
Lin W Q, Xu B, Chen L, Zhou F, Chen J L 2016 Acta Phys. Sin. 65 133102
 Google Scholar
Google Scholar
[4] Li J, Lu C H, Yao Q H, Zhang X L, Liu J J, Yang H H, Chen G N 2011 Biosens. Bioelectron. 26 3894
 Google Scholar
Google Scholar
[5] Mattevi C, Eda G, Agnoli S, Miller S, Mkhoyan K A, Celik O, Mastrogiovanni D, Granozzi G, Garfunkel E, Chhowalla M 2009 Adv. Funct. Mater. 19 2577
 Google Scholar
Google Scholar
[6] Peper S, Telting-Diaz M, Almond P, Albrecht-Schmitt T, Bakker E 2002 Anal. Chem. 74 1327
 Google Scholar
Google Scholar
[7] Liu G, Jin W, Xu N 2016 Angew. Chem. Int. Ed. 55 13384
 Google Scholar
Google Scholar
[8] Joshi R K, Carbone P, Wang F C, Kravets V G, Su Y, Grigorieva I V, Wu H A, Geim A K, Nair R R 2014 Science 343 752
 Google Scholar
Google Scholar
[9] Chen L, Shi G, Shen J, Peng B, Zhang B, Wang Y, Bian F, Wang J, Li D, Qian Z, Xu G, Liu G, Zeng J, Zhang L, Yang Y, Zhou G, Wu M, Jin W, Li J, Fang H 2017 Nature 550 380
 Google Scholar
Google Scholar
[10] Pei S, Cheng H M 2012 Carbon 50 3210
 Google Scholar
Google Scholar
[11] Yang J, Shi G, Tu Y, Fang H 2014 Angew. Chem. Int. Ed. 53 10190
 Google Scholar
Google Scholar
[12] Kim S, Zhou S, Hu Y, Acik M, Chabal Y J, Berger C, de Heer W, Bongiorno A, Riedo E 2012 Nat. Mater. 11 544
 Google Scholar
Google Scholar
[13] Qian Z, Chen L, Li D Y, Peng B Q, Shi G S, Xu G, Fang H P, Wu M H 2017 Chin. Phys. B 2 6
[14] Eigler S, Hirsch A 2014 Angew. Chem. Int. Ed. 53 7720
 Google Scholar
Google Scholar
[15] 陈浩, 彭同江, 刘波, 孙红娟, 雷德会 2017 物理学报 66 080701
 Google Scholar
Google Scholar
Chen H, Peng T J, Liu B, Sun H J, Lei D H 2017 Acta Phys. Sin. 66 080701
 Google Scholar
Google Scholar
[16] Chen D, Feng H, Li J 2012 Chem. Rev. 112 6027
 Google Scholar
Google Scholar
[17] Stankovich S, Dikin D A, Piner R D, Kohlhaas K A, Kleinhammes A, Jia Y, Wu Y, Nguyen S T, Ruoff R S 2007 Carbon 45 1558
 Google Scholar
Google Scholar
[18] Neklyudov V V, Khafizov N R, Sedov I A, Dimiev A M 2017 Phys. Chem. Chem. Phys. 19 17000
 Google Scholar
Google Scholar
[19] Shulga Y M, Martynenko V M, Muradyan V E, Baskakov S A, Smirnov V A, Gutsev G L 2010 Chem. Phys. Lett. 498 287
 Google Scholar
Google Scholar
[20] Li J, Xiao G, Chen C, Li R, Yan D 2013 J. Mater. Chem. A 1 1481
 Google Scholar
Google Scholar
[21] Serpone N, Lawless D, Khairutdinov R 1995 J. Phys. Chem. 99 16646
 Google Scholar
Google Scholar
-
图 1 氧化石墨烯悬浮液在不同温度热处理后的状态和基团变化 (a)不同温度处理后的溶液状态; (b) XPS检测的C, O元素的原子数含量百分比; (c) C 1s精细谱及分峰拟合; (d)各基团的原子数含量百分比
Fig. 1. State and changing of group of the graphene oxide suspension after heat treatment at different temperatures: (a) Solution state after different temperature treatment; (b) percentage of atomic content of C and O elements detected by XPS; (c) C 1s XPS spectra and peak fitting; (d) percentage of atomic content of each group.
图 2 不同温度热处理后的形貌、红外光谱以及悬浮稳定性表征 (a) SEM图像, 三种温度处理后的氧化石墨烯单片层形貌; (b)多个平行样的红外吸收光谱; (c)三种温度处理后的氧化石墨烯悬浮液的Zeta电位
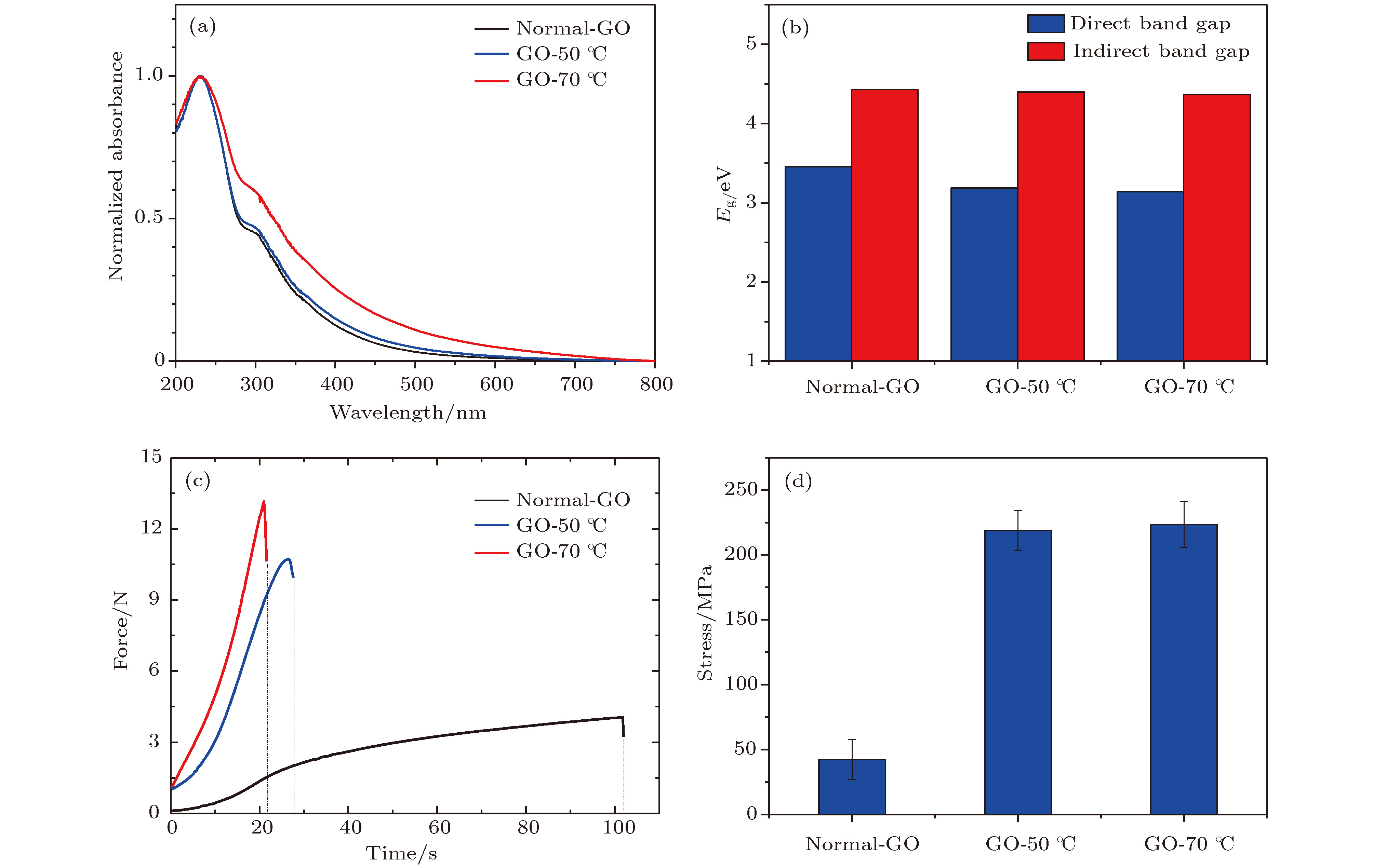
Fig. 2. Morphology, FT-IR spectra and suspension stability characterization after heat treatment at different temperatures: (a) SEM images, namely, the morphology of monolayer of graphene oxide after three temperature treatments; (b) FT-IR spectra of multiple parallel samples; (c) Zeta potential basic of graphene oxide after three temperature treatments.
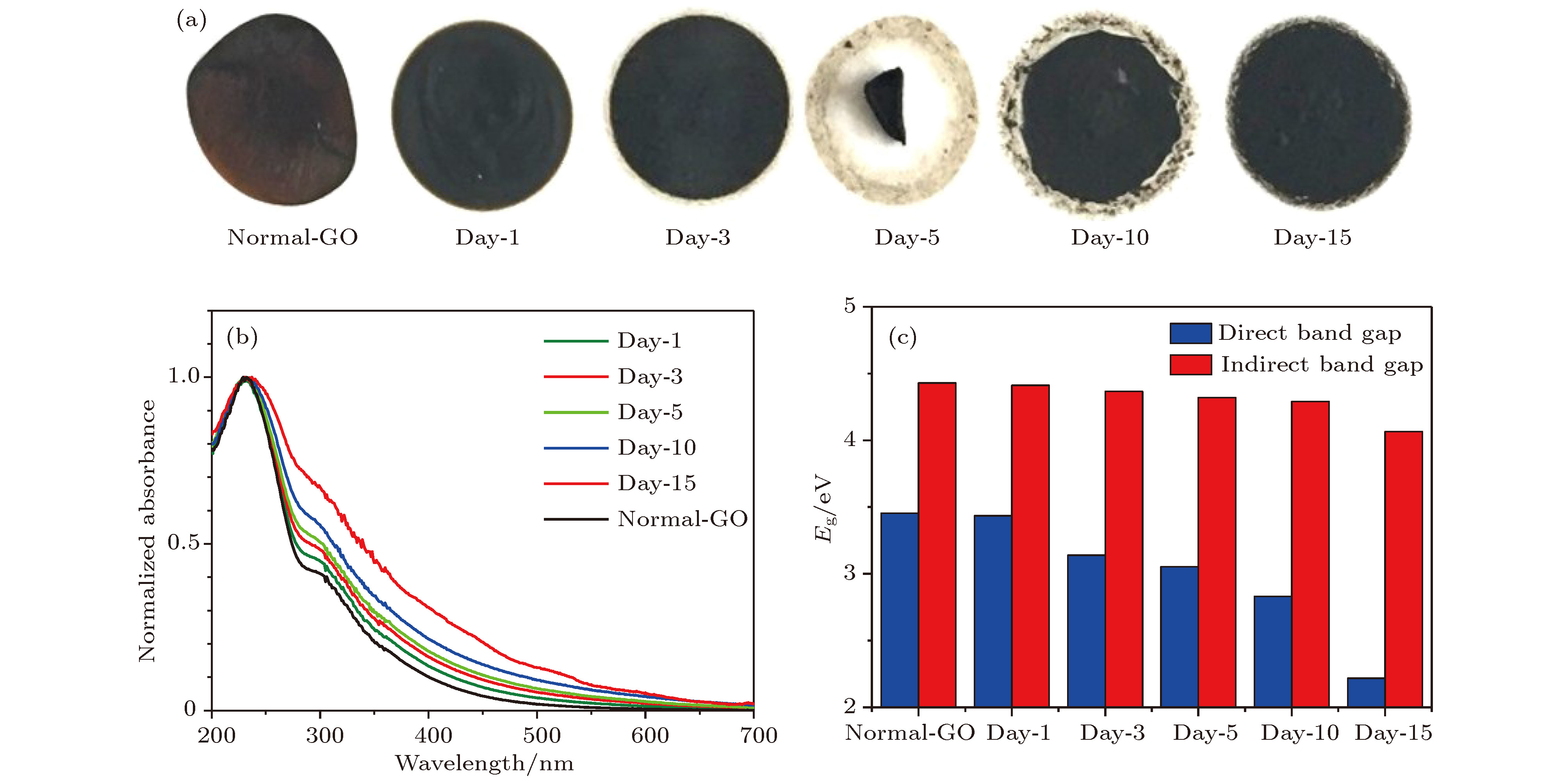
图 4 氧化石墨烯悬浮液在70 ℃热处理不同时间的性能变化 (a)不同时间(1, 3, 5, 10, 15 d)热处理的氧化石墨烯悬浮液, 分别滴涂制备的充分干燥的氧化石墨烯膜; (b)紫外吸收光谱; (c)光子带隙
Fig. 4. Performance change of graphene oxide at 70 ℃ heat treatment for different time: (a) Dried graphene oxide film separately prepared by drop coating after graphene oxide heat treatment for different time (1, 3, 5, 10, 15 d); (b) UV-vis spectra; (c) photonic band.
-
[1] McAllister M J, Li J L, Adamson D H, Schniepp H C, Abdala A A, Liu J, Herrera-Alonso M, Milius D L, Car R, Prud'homme R K, Aksay I A 2007 Chem. Mater. 19 4396
 Google Scholar
Google Scholar
[2] 刘学文, 朱重阳, 董辉, 徐峰, 孙立涛 2016 物理学报 65 118802
 Google Scholar
Google Scholar
Liu X W, Zhu C Y, Dong H, Xu F, Sun L T 2016 Acta Phys. Sin. 65 118802
 Google Scholar
Google Scholar
[3] 林文强, 徐斌, 陈亮, 周峰, 陈均朗 2016 物理学报 65 133102
 Google Scholar
Google Scholar
Lin W Q, Xu B, Chen L, Zhou F, Chen J L 2016 Acta Phys. Sin. 65 133102
 Google Scholar
Google Scholar
[4] Li J, Lu C H, Yao Q H, Zhang X L, Liu J J, Yang H H, Chen G N 2011 Biosens. Bioelectron. 26 3894
 Google Scholar
Google Scholar
[5] Mattevi C, Eda G, Agnoli S, Miller S, Mkhoyan K A, Celik O, Mastrogiovanni D, Granozzi G, Garfunkel E, Chhowalla M 2009 Adv. Funct. Mater. 19 2577
 Google Scholar
Google Scholar
[6] Peper S, Telting-Diaz M, Almond P, Albrecht-Schmitt T, Bakker E 2002 Anal. Chem. 74 1327
 Google Scholar
Google Scholar
[7] Liu G, Jin W, Xu N 2016 Angew. Chem. Int. Ed. 55 13384
 Google Scholar
Google Scholar
[8] Joshi R K, Carbone P, Wang F C, Kravets V G, Su Y, Grigorieva I V, Wu H A, Geim A K, Nair R R 2014 Science 343 752
 Google Scholar
Google Scholar
[9] Chen L, Shi G, Shen J, Peng B, Zhang B, Wang Y, Bian F, Wang J, Li D, Qian Z, Xu G, Liu G, Zeng J, Zhang L, Yang Y, Zhou G, Wu M, Jin W, Li J, Fang H 2017 Nature 550 380
 Google Scholar
Google Scholar
[10] Pei S, Cheng H M 2012 Carbon 50 3210
 Google Scholar
Google Scholar
[11] Yang J, Shi G, Tu Y, Fang H 2014 Angew. Chem. Int. Ed. 53 10190
 Google Scholar
Google Scholar
[12] Kim S, Zhou S, Hu Y, Acik M, Chabal Y J, Berger C, de Heer W, Bongiorno A, Riedo E 2012 Nat. Mater. 11 544
 Google Scholar
Google Scholar
[13] Qian Z, Chen L, Li D Y, Peng B Q, Shi G S, Xu G, Fang H P, Wu M H 2017 Chin. Phys. B 2 6
[14] Eigler S, Hirsch A 2014 Angew. Chem. Int. Ed. 53 7720
 Google Scholar
Google Scholar
[15] 陈浩, 彭同江, 刘波, 孙红娟, 雷德会 2017 物理学报 66 080701
 Google Scholar
Google Scholar
Chen H, Peng T J, Liu B, Sun H J, Lei D H 2017 Acta Phys. Sin. 66 080701
 Google Scholar
Google Scholar
[16] Chen D, Feng H, Li J 2012 Chem. Rev. 112 6027
 Google Scholar
Google Scholar
[17] Stankovich S, Dikin D A, Piner R D, Kohlhaas K A, Kleinhammes A, Jia Y, Wu Y, Nguyen S T, Ruoff R S 2007 Carbon 45 1558
 Google Scholar
Google Scholar
[18] Neklyudov V V, Khafizov N R, Sedov I A, Dimiev A M 2017 Phys. Chem. Chem. Phys. 19 17000
 Google Scholar
Google Scholar
[19] Shulga Y M, Martynenko V M, Muradyan V E, Baskakov S A, Smirnov V A, Gutsev G L 2010 Chem. Phys. Lett. 498 287
 Google Scholar
Google Scholar
[20] Li J, Xiao G, Chen C, Li R, Yan D 2013 J. Mater. Chem. A 1 1481
 Google Scholar
Google Scholar
[21] Serpone N, Lawless D, Khairutdinov R 1995 J. Phys. Chem. 99 16646
 Google Scholar
Google Scholar
计量
- 文章访问数: 15271
- PDF下载量: 108
- 被引次数: 0














 下载:
下载: