-
Organic-inorganic metal halide perovskites are a new type of photovoltaic material, they have attracted wide attention and made excellent progress in recent years. The power conversion efficiency of a single-junction perovskite solar cell has been increased to 25.2% just within a decade. Meanwhile, crystalline silicon solar cells account for nearly 90% of industrialized solar cells and have a maximum efficiency of 26.7%, approaching to their theoretical limit. It is more difficult to further improve the efficiency of single junction solar cells. It has been shown that multi-junction tandem solar cells prepared by stacking absorption layers with different bandgaps can better use sunlight, which is one of the most promising strategies to break the efficiency limitation of single-junction solar cells. Due to the bandgap tunability and low-temperature solution processability, perovskites stand out among many other materials for manufacturing multi-junction tandem solar cells. Wide bandgap perovskites with a bandgap of 1.63 eV or above have been combined with narrow band gap inorganic absorption layers such as silicon, copper indium gallium selenide, cadmium telluride or narrow bandgap perovskite to produce high efficiency tandem solar cells. In addition to the promoting of the efficiency improvement of solar cells, the wide bandgap perovskites have broad applications in photovoltaic building integration and photocatalytic fields. Therefore, it is very important to explore and develop high quality wide bandgap perovskite materials and solar cells. Unfortunately, the wide bandgap perovskites have several intrinsic weaknesses, including being more vulnerable to the migration of halogen ions under being illuminated, more defects, and greater possibility of energy level mismatching with the charge transport layers than the narrow bandgap counterparts, which limits the further development of the wide bandgap perovskite solar cells. In this review, the development status of wide bandgap perovskite solar cells is summarized and corresponding strategies for improving their performance are put forward. Furthermore, some personal views on the future development of wide bandgap perovskite solar cells are also presented here in this paper.
[1] Xing G C, Mathews N, Lim S S, Yantara N, Liu X F, Sabba D, Grätzel M, Mhaisalkar S, Sum T C 2014 Nat. Mater. 13 476
 Google Scholar
Google Scholar
[2] Tan Z K, Moghaddam R S, Lai M L, Docampo P, Higler R, Deschler F, Price M, Sadhanala A, Pazos L M, Credgington D 2014 Nat. Nanotechnol. 9 687
 Google Scholar
Google Scholar
[3] Dou L, Yang Y, You J B, Hong Z, Chang W H, Li G, Yang Y 2014 Nat. Commun. 5 5404
 Google Scholar
Google Scholar
[4] Kojima A, Teshima K, Shirai Y, Miyasaka T 2009 J. Am. Chem. Soc. 131 6050
 Google Scholar
Google Scholar
[5] Kim M, Kim G H, Lee T K, Choi I W, Choi H W, Jo Y, Yoon Y J, Kim J W, Lee J Y, Huh D, Lee H, Kwak S K, Kim J Y, Kim D S 2019 Joule 3 2179
 Google Scholar
Google Scholar
[6] Jiang Q, Zhao Y, Zhang X W, Yang X L, Chen Y, Chu Z, Ye Q, Li X X, Yin Z G, You J B 2019 Nat. Photonics 13 460
 Google Scholar
Google Scholar
[7] Wang P Y, Li R J, Chen B B, Hou F H, Zhang J, Zhao Y, Zhang X D 2020 Adv. Mater. 32 1905766
 Google Scholar
Google Scholar
[8] Min H, Kim M, Lee S U, Kim H, Kim G, Choi K, Lee J Hee, Seok S I 2019 Science 366 749
 Google Scholar
Google Scholar
[9] Yoo J J, Wieghold S, Sponseller M C, Chua M R, Bertram S N, Hartono N T P, Tresback J S, Hansen E C, Correa-Baena J P, Bulovic V 2019 Energy Environ. Sci. 12 2192
 Google Scholar
Google Scholar
[10] Liu Y H, Akin S, Pan L F, Uchida R, Grätzel M 2019 Sci. Adv. 5 eaaw2543
 Google Scholar
Google Scholar
[11] Zhu P C, Gu S, Luo X, Gao Y, Li S L, Zhu J, Tan H R 2019 Adv. Energy Mater. 10 1903083
 Google Scholar
Google Scholar
[12] Zheng X P, Hou Y, Bao C X, Yin J, Yuan F L, Huang Z R, Song K P, Liu J K, Troughton J, Gasparini N, Zhou C, Lin Y B, Xue D J, Chen B, Johnston A K, Wei N N, Hedhili M N, Wei M, Alsalloum A Y, Maity P, Turedi B, Yang C, Baran D, Anthopoulos T D, Han Y, Lu Z H, Mohammed O F, Gao F, Sargent E H, Bakr O M 2020 Nat. Energy 5 131
 Google Scholar
Google Scholar
[13] Shockley W, Queisser H J 1961 J. Appl. Phys. 32 510
 Google Scholar
Google Scholar
[14] Beiley Z M, McGehee M D 2012 Energy Environ. Sci. 5 9173
 Google Scholar
Google Scholar
[15] Mailoa J P, Bailie C D, Johlin E C, Hoke E T, Akey A J, Nguyen W H, McGehee M D, Buonassisi T 2015 Appl. Phys. Lett. 106 121105
 Google Scholar
Google Scholar
[16] Albrecht S, Saliba M, Correa Baena J P, Lang F, Kegelmann L, Mews M, Steier L, Abate A, Rappich J, Korte L, Schlatmann R, Nazeeruddin M K, Hagfeldt A, Grätzel M, Rech B 2016 Energy Environ. Sci. 9 81
 Google Scholar
Google Scholar
[17] Altazin S, Stepanova L, Werner J, Niesen B, Ballif C, Ruhstaller B 2018 Opt. Express 26 A579
 Google Scholar
Google Scholar
[18] Fang Z M, Wang S Z, Yang S F, Ding L M 2018 Inorg. Chem. Front. 5 1690
 Google Scholar
Google Scholar
[19] Hu W P, He X, Fang Z M, Lian W T, Shang Y B, Li X C, Zhou W R, Zhang M M, Chen T, Lu Y L, Zhang L J, Ding L M, Yang S F 2020 Nano Energy 68 104362
 Google Scholar
Google Scholar
[20] Jia X, Ding L M 2018 Sci. China Mater. 62 54
 Google Scholar
Google Scholar
[21] Zuo C T, Ding L M 2017 Angew. Chem. Int. Ed. 56 6528
 Google Scholar
Google Scholar
[22] Chen B, Zheng X P, Bai Y, Padture N P, Huang J S 2017 Adv. Energy Mater. 7 1602400
 Google Scholar
Google Scholar
[23] Hu J N, Cheng Q, Fan R D, Zhou H P 2017 Sol. RRL 1 1700045
 Google Scholar
Google Scholar
[24] Lal N N, Dkhissi Y, Li W, Hou Q C, Cheng Y B, Bach U 2017 Adv. Energy Mater. 7 1602761
 Google Scholar
Google Scholar
[25] Jošt M, Kegelmann L, Korte L, Albrecht S 2020 Adv. Energy Mater. 10 1904102
 Google Scholar
Google Scholar
[26] Al-Ashouri A, Magomedov A, Roß M, et al. 2019 Energy Environ. Sci. 12 3356
 Google Scholar
Google Scholar
[27] Kim D H, Muzzillo C P, Tong J, et al. 2019 Joule 3 1734
 Google Scholar
Google Scholar
[28] Lin R, Xiao K, Qin Z Y, Han Q L, Zhang C F, Wei M Y, Saidaminov M I, Gao Y, Xu J, Xiao M, Li A D, Zhu J, Sargent E H, Tan H R 2019 Nat. Energy 4 864
 Google Scholar
Google Scholar
[29] McMeekin D P, Mahesh S, Noel N K, Klug M T, Lim J, Warby J H, Ball J M, Herz L M, Johnston M B, Snaith H J 2019 Joule 3 387
 Google Scholar
Google Scholar
[30] Xue Q F, Xia R X, Brabec C J, Yip H L 2018 Energy Environ. Sci. 11 1688
 Google Scholar
Google Scholar
[31] Henemann A 2008 Renew. Energy Focus 9 14
 Google Scholar
Google Scholar
[32] Shi B, Duan L R, Zhao Y, Luo J S, Zhang X D 2020 Adv. Mater. 32 1806474
 Google Scholar
Google Scholar
[33] Park S, Chang W J, Lee C W, Park S, Ahn H Y, Nam K T 2016 Nat. Energy 2 16185
 Google Scholar
Google Scholar
[34] 陈为, 魏伟, 孙予罕 2017 中国科学: 化学 47 1251
 Google Scholar
Google Scholar
Chen W, Wei W, Sun Y H 2017 Sci. China: Chem. 47 1251
 Google Scholar
Google Scholar
[35] Hu M, Bi C, Yuan Y B, Bai Y, Huang J S 2016 Adv. Sci. 3 1500301
 Google Scholar
Google Scholar
[36] Lin Y Z, Chen B, Zhao F W, Zheng X P, Deng Y H, Shao Y C, Fang Y J, Bai Y, Wang C R, Huang J S 2017 Adv. Mater. 29 1700607
 Google Scholar
Google Scholar
[37] Bush K A, Frohna K, Prasanna R, Beal R E, Leijtens T, Swifter S A, McGehee M D 2018 ACS Energy Lett. 3 428
 Google Scholar
Google Scholar
[38] Wang Z P, Lin Q Q, Chmiel F P, Sakai N, Herz L M, Snaith H J 2017 Nat. Energy 2 17135
 Google Scholar
Google Scholar
[39] Chen C, Song Z N, Xiao C X, Zhao D W, Shrestha N, Li C W, Yang G, Yao F, Zheng X L, Ellingson R J, Jiang C S, Al-Jassim M, Zhu K, Fang G J, Yan Y F 2019 Nano Energy 61 141
 Google Scholar
Google Scholar
[40] Chen B, Yu Z S, Liu K, Zheng X P, Liu Y, Shi J W, Spronk D, Rudd P N, Holman Z, Huang J S 2019 Joule 3 177
 Google Scholar
Google Scholar
[41] Wang J, Zhang J, Zhou Y Z, Liu H B, Xue Q F, Li X S, Chueh C C, Yip L P, Zhu Z L, Jen A K Y 2020 Nat. Commun. 11 177
 Google Scholar
Google Scholar
[42] Liu C, Yang Y Z, Zhang C L, Wu S H, Wei L Y, Guo F, Arumugam G M, Hu J L, Liu X Y, Lin J, Schropp R E L, Mai Y H 2020 Adv. Mater. 32 1907361
 Google Scholar
Google Scholar
[43] Palmstrom A F, Eperon G E, Leijtens T, et al. 2019 Joule 3 2193
 Google Scholar
Google Scholar
[44] Xu J X, Boyd C C, Yu Z J, et al. 2020 Science 367 1097
 Google Scholar
Google Scholar
[45] Kim D, Jung H J, Park I J, Larson B W, Dunfield S P, Xiao C X, Kim J, Tong J H, Boonmongkolras P, Ji S G, Zhang F, Pae S R, Kim M, Kang S B, Dravid V, Berry J J, Kim J Y, Zhu K, Kim D H, Shin B 2020 Science 368 155
 Google Scholar
Google Scholar
[46] Ye J Y, Tong J H, Hu J, et al. 2020 Sol. RRL 4 2000082
 Google Scholar
Google Scholar
[47] Duong T, Wu Y L, Shen H P, et al. 2017 Adv. Energy Mater. 7 1700228
 Google Scholar
Google Scholar
[48] Yang M J, Kim D H, Yu Y, Li Z, Reid O G, Song Z N, Zhao D W, Wang C L, Li L W, Meng Y, Guo T, Yan Y F, Zhu K 2018 Mater. Today Energy 7 232
 Google Scholar
Google Scholar
[49] Zhou Y, Wang F, Cao Y, Wang J P, Fang H H, Loi M A, Zhao N, Wong C P 2017 Adv. Energy Mater. 7 1701048
 Google Scholar
Google Scholar
[50] Yu Y, Wang C L, Grice C R, Shrestha N, Zhao D W, Liao W Q, Guan L, Awni R A, Meng W W, Cimaroli A J, Zhu K, Ellingson R J, Yan Y F 2017 ACS Energy Lett. 2 1177
 Google Scholar
Google Scholar
[51] Zhou Y, Jia Y H, Fang H H, Loi M A, Xie F Y, Gong L, Qin M C, Lu X H, Wong C P, Zhao N 2018 Adv. Funct. Mater. 28 1803130
 Google Scholar
Google Scholar
[52] Abdi-Jalebi M, Andaji-Garmaroudi Z, Cacovich S, et al. 2018 Nature 555 497
 Google Scholar
Google Scholar
[53] Kim J, Saidaminov M I, Tan H R, et al. 2018 Adv. Mater. 30 1706275
 Google Scholar
Google Scholar
[54] Duong T, Pham H, Kho T H, et al. 2019 Adv. Energy Mater. 10 1903553
 Google Scholar
Google Scholar
[55] Tan H R, Che F L, Wei M Y, Zhao Y C, Saidaminov M I, Petar T, Danny B, Grant W, Tan F R, Zhuang T T 2018 Nat. Commun. 9 3100
 Google Scholar
Google Scholar
[56] Saliba M, Matsui T, Domanski K, Seo J Y, Ummadisingu A, Zakeeruddin S M, Correa-Baena J P, Tress W R, Abate A, Hagfeldt A, Grätzel M 2016 Science 354 206
 Google Scholar
Google Scholar
[57] McMeekin D P, Sadoughi G, Rehman W, Eperon G E, Saliba M, Hörantner M T, Haghighirad A, Sakai N, Korte L, Rech B, Johnston M B, Herz L M, Snaith H J 2016 Science 351 151
 Google Scholar
Google Scholar
[58] Zhuang J, Mao P, Luan Y G, Yi X H, Tu Z Y, Zhang Y Y, Yi Y P, Wei Y Z, Chen N L, Lin T, Wang F Y, Li C, Wang J Z 2019 ACS Energy Lett. 4 2913
 Google Scholar
Google Scholar
[59] Gharibzadeh S, Abdollahi Nejand B, Jakoby M, et al. 2019 Adv. Energy Mater. 9 1803699
 Google Scholar
Google Scholar
[60] Wang P Y, Zhang X W, Zhou Y Q, Jiang Q, Ye Q F, Chu Z M, Li X X, Yang X L, Yin Z G, You J B 2018 Nat. Commun. 9 2225
 Google Scholar
Google Scholar
[61] Zhang J, Bai D L, Jin Z W, Bian H, Wang K, Sun J, Wang Q, Liu S Z F 2018 Adv. Energy Mater. 8 1703246
 Google Scholar
Google Scholar
[62] Wang Y, Dar M I, Ono L K, Zhang T Y, Kan M, Li Y W, Zhang L J, Wang X T, Yang Y G, Gao X Y, Qi Y B, Grätzel M, Zhao Y X 2019 Science 365 591
 Google Scholar
Google Scholar
[63] Ye Q F, Zhao Y, Mu S Q, Ma F, Gao F, Chu Z M, Yin Z G, Gao P Q, Zhang X W, You J B 2019 Adv. Mater. 1 1905143
 Google Scholar
Google Scholar
[64] Xiao Q, Tian J J, Xue Q F, Wang J, Xiong B J, Han M M, Li Z, Zhu Z L, Yip H L, Li Z 2019 Angew. Chem. Int. Ed. 58 17724
 Google Scholar
Google Scholar
[65] Green M A, Ho-Baillie A, Snaith H J 2014 Nat. Photonics 8 506
 Google Scholar
Google Scholar
[66] Kim H S, Im S H, Park N G 2014 J. Phys.Chem. C 118 5615
 Google Scholar
Google Scholar
[67] Li Z, Yang M J, Park J S, Wei S H, Berry J, Zhu K 2015 Chem. Mater. 28 284
 Google Scholar
Google Scholar
[68] Chang Y H, Park C H 2004 J. Korean Phys. Soc. 44 889
[69] Anaya M, Correabaena J P, Lozano G L, Saliba M, Anguita P, Roose B, Abate A, Steiner U, Grätzel M, Calvo M 2016 J. Mater. Chem. A 4 11214
 Google Scholar
Google Scholar
[70] Noh J H, Im S H, Heo J H, Mandal T N, Seok S I 2013 Nano Lett. 13 1764
 Google Scholar
Google Scholar
[71] Eperon G E, Stranks S D, Menelaou C, Johnston M B, Herz L M, Snaith H J 2014 Energy Environ. Sci. 7 982
 Google Scholar
Google Scholar
[72] Saliba M, Correa-Baena J P, Grätzel M, Hagfeldt A, Abate A 2017 Angew. Chem. Int. Ed. 57 2554
 Google Scholar
Google Scholar
[73] Unger E L, Bowring A R, Tassone C J, Pool V L, Gold-Parker A, Cheacharoen R, Stone K H, Hoke E T, Toney M F, McGehee M D 2014 Chem. Mater. 26 7158
 Google Scholar
Google Scholar
[74] Dong Q, Yuan Y B, Shao Y C, Fang Y J, Wang Q, Huang J S 2015 Energy Environ. Sci. 8 2464
 Google Scholar
Google Scholar
[75] Lee B, Hwang T, Lee S, Shin B, Park B 2019 Sci. Rep. 9 4803
 Google Scholar
Google Scholar
[76] Zhang C P, Li Z P, Liu J, Xin Y C, Shao Z P, Cui G, Pang S P 2018 ACS Energy Lett. 3 1801
 Google Scholar
Google Scholar
[77] Ma T, Wang S W, Zhang Y W, Zhang K X, Yi L X 2020 J. Mater. Sci. 55 464
 Google Scholar
Google Scholar
[78] Tong G, Ono L K, Qi Y B 2019 Energy Technol. 8 1900961
 Google Scholar
Google Scholar
[79] Protesescu L, Yakunin S, Bodnarchuk M I, et al. 2015 Nano Lett. 15 3692
 Google Scholar
Google Scholar
[80] Mehrabian M, Dalir S, Mahmoudi G, Miroslaw B, Safin D A 2019 Eur. J. Inorg. Chem. 2019 3699
 Google Scholar
Google Scholar
[81] Leijtens T, Bush K A, Prasanna R, McGehee M D 2018 Nat. Energy 3 828
 Google Scholar
Google Scholar
[82] Mahesh S, Ball J M, Oliver R D J, McMeekin D P, Nayak P K, Johnston M B, Snaith H J 2020 Energy Environ. Sci. 13 258
 Google Scholar
Google Scholar
[83] Levine I, Vera O G, Kulbak M, Ceratti D-R, Rehermann C, Márquez J A, Levcenko S, Unold T, Hodes G, Balberg I, Cahen D, Dittrich T 2019 ACS Energy Lett. 4 1150
 Google Scholar
Google Scholar
[84] Song Z, Chen C, Li C, Awni R A, Zhao D, Yan Y 2019 Semicond. Sci. Technol. 34 093001
 Google Scholar
Google Scholar
[85] Hoke E T, Slotcavage D J, Dohner E R, Bowring A R, Karunadasa H I, McGehee M D 2015 Chem. Sci. 6 613
 Google Scholar
Google Scholar
[86] Slotcavage D J, Karunadasa H I, McGehee M D 2016 ACS Energy Lett. 1 1199
 Google Scholar
Google Scholar
[87] Yun J S, Seidel J, Kim J, Soufiani A M, Huang S, Lau J, Jeon N J, Seok S I, Green M A, Ho-Baillie A 2016 Adv. Energy Mater. 6 1600330
 Google Scholar
Google Scholar
[88] Beal R E, Hagström N Z, Barrier J, Gold-Parker A, Prasanna R, Bush K A, Passarello D, Schelhas L T, Brüning K, Tassone C J, Steinrück H G, McGehee M D, Toney M F, Nogueira A F 2020 Matter 2 207
 Google Scholar
Google Scholar
[89] Bischak C G, Hetherington C L, Wu H, Aloni S, Ogletree D F, Limmer D T, Ginsberg N S 2017 Nano Lett. 17 1028
 Google Scholar
Google Scholar
[90] Brennan M C, Draguta S, Kamat P V, Kuno M 2017 ACS Energy Lett. 3 204
 Google Scholar
Google Scholar
[91] Liu S, Guan Y J, Sheng Y S, Hu Y, Rong Y G, Mei A Y, Han H W 2020 Adv. Energy Mater. 10 1902492
 Google Scholar
Google Scholar
[92] Gao F, Zhao Y, Zhang X W, You J B 2020 Adv. Energy Mater. 10 1902650
 Google Scholar
Google Scholar
[93] Han J H, Luo S P, Yin X W, Zhou Y, Nan H, Li J B, Li X, Oron D, Shen H P, Lin H 2018 Small 14 1801016
 Google Scholar
Google Scholar
[94] Bai S, Da P M, Li C, Wang Z P, Yuan Z C, Fu F, Kawecki M, Liu X J, Sakai N, Wang J T W, Huettner S, Buecheler S, Fahlman M, Gao F, Snaith H J 2019 Nature 571 245
 Google Scholar
Google Scholar
[95] Bi D Q, Yi C Y, Luo J S, Décoppet J D, Zhang F, Zakeeruddin Shaik M, Li X, Hagfeldt A, Grätzel M 2016 Nat. Energy 1 16142
 Google Scholar
Google Scholar
[96] Noel N K, Abate A, Stranks S D, Parrott E S, Burlakov V M, Goriely A, Snaith H J 2014 ACS Nano 8 9815
 Google Scholar
Google Scholar
[97] Correa-Baena J B, Luo Y Q, Brenner T M, Snaider J, Sun S J, Li X Y, Jensen M A, Hartono N P T, Nienhaus L, Wieghold S, Poindexter J R, Wang S, Meng Y S, Wang T, Lai B, Holt M V, Cai Z H, Bawendi M G, Huang L B, Buonassisi T, Fenning D P 2019 Science 363 627
 Google Scholar
Google Scholar
[98] Kieslich G, Sun S, Cheetham A K 2014 Chem. Sci. 12 4712
 Google Scholar
Google Scholar
[99] Kubicki D, Prochowicz D, Hofstetter A, Saski M, Yadav P, Bi D, Pellet N, Lewiński J, Zakeeruddin S M, Grätzel M 2018 J. Mater. Chem. A 140 3345
 Google Scholar
Google Scholar
[100] Jodlowski A D, Roldán-Carmona C, Grancini G, Salado M, Ralaiarisoa M, Ahmad S, Koch N, Camacho L, de Miguel G, Nazeeruddin M K 2017 Nat. Energy 2 972
 Google Scholar
Google Scholar
[101] Chen H, Wei Q, Saidaminov M I, Wang F, Johnston A, Hou Y, Peng Z J, Xu K M, Zhou W J, Liu Z H, Qiao L, Wang X, Xu S W, Li J Y, Long R, Ke Y Q, Sargent E H, Ning Z J 2019 Adv. Mater. 31 e1903559
 Google Scholar
Google Scholar
[102] Stoddard R J, Rajagopal A, Palmer R L, Braly I L, Jen A K Y, Hillhouse H W 2018 ACS Energy Lett. 3 1261
 Google Scholar
Google Scholar
[103] Yu H, Wang F, Xie F Y, Li W W, Chen J, Zhao N 2014 Adv. Funct. Mater. 24 7102
 Google Scholar
Google Scholar
[104] Chae J S, Dong Q F, Huang J S, Centrone A 2015 Nano Lett. 15 8114
 Google Scholar
Google Scholar
[105] Gao C, Liu J, Liao C, Ye Q Y, Zhang Y Z, He X L, Guo X W, Mei J, Lau W 2015 RSC Adv. 5 26175
 Google Scholar
Google Scholar
[106] Xie Y L, Yu H Y, Duan J S, Xu L, Hu B 2020 ACS Appl. Mater. Interfaces 12 11190
 Google Scholar
Google Scholar
[107] Abate A, Saliba M, Hollman D J, Stranks S D, Wojciechowski K, Avolio R, Grancini G, Petrozza A, Snaith H J 2014 Nano Lett. 14 3247
 Google Scholar
Google Scholar
[108] Heo D Y, Lee T H, Iwan A, Kavan L, Omatova M, Majkova E, Kamarás K, Jang H W, Kim S Y 2020 J. Power Sources 458 228067
 Google Scholar
Google Scholar
[109] Tong J H, Song Z N, Kim D H, et al. 2019 Science 364 475
 Google Scholar
Google Scholar
[110] Tang G, Yang C, Stroppa A, Fang D N, Hong J W 2017 J. Chem. Phys. 146 224702
 Google Scholar
Google Scholar
[111] Ke W J, Xiao C X, Wang C L, et al. 2016 Adv. Mater. 28 5214
 Google Scholar
Google Scholar
[112] Xu J X, Buin A, Ip A H, et al. 2015 Nat. Commun. 6 7081
 Google Scholar
Google Scholar
[113] Liang P W, Chueh C C, Williams S T, Jen A K Y 2015 Adv. Energy Mater. 5 1402321
 Google Scholar
Google Scholar
[114] Gatti T, Menna E, Meneghetti M, Maggini M, Petrozza A, Lamberti F 2017 Nano Energy 41 84
 Google Scholar
Google Scholar
[115] Fang Y J, Bi C, Wang D, Huang J S 2017 ACS Energy Lett. 2 782
 Google Scholar
Google Scholar
[116] Cui C H, Li Y W, Li Y F 2017 Adv. Energy Mater. 7 1601251
 Google Scholar
Google Scholar
[117] Lee J W, Park N G 2019 Adv. Energy Mater. 10 1903249
 Google Scholar
Google Scholar
[118] Lee J W, Kim H S, Park N G 2016 Acc. Chem. Res. 49 311
 Google Scholar
Google Scholar
[119] Xue D J, Hou Y, Liu S C, Wei M, Chen B, Huang Z, Li Z, Sun B, Proppe A H, Dong Y, Saidaminov M I, Kelley S O, Hu J S, Sargent E H 2020 Nat. Commun. 11 1514
 Google Scholar
Google Scholar
[120] Jan S, Robby P, Rolf B 2018 Sol. Energy Mater. Sol. Cells 187 39
 Google Scholar
Google Scholar
[121] Luo D Y, Yang W Q, Wang Z P, et al. 2018 Science 360 1442
 Google Scholar
Google Scholar
[122] Bu X N, Westbrook R J E, Lanzetta L, Ding D, Chotchuangchutchaval T, Aristidou N, Haque S A 2019 Sol. RRL 3 1800282
 Google Scholar
Google Scholar
[123] Wang Q, Zheng X P, Deng Y H, Zhao J J, Chen Z L, Huang J S 2017 Joule 1 371
 Google Scholar
Google Scholar
[124] Zheng X P, Chen B, Dai J, Fang Y J, Bai Y, Lin Y Z, Wei H T, Zeng X C, Huang J S 2017 Nat. Energy 2 17102
 Google Scholar
Google Scholar
[125] Elgamel H E, Barnett A M, Rohatgi A, Chen Z, Vinckier C, Nijs J, Mertens R 1995 J. Appl. Phys. 78 3457
 Google Scholar
Google Scholar
[126] Jaysankar M, Raul B A L, Bastos J, Burgess C, Weijtens C, Creatore M, Aernouts T, Kuang Y, Gehlhaar R, Hadipour A, Poortmans J 2018 ACS Energy Lett. 4 259
 Google Scholar
Google Scholar
[127] Stolterfoht M, Caprioglio P, Wolff C M, et al. 2019 Energy Environ. Sci. 12 2778
 Google Scholar
Google Scholar
[128] Bian H, Bai D L, Jin Z W, Wang K, Liang L, Wang H R, Zhang J R, Wang Q, Liu S Z F 2018 Joule 2 1500
 Google Scholar
Google Scholar
[129] Zeng Q, Liu L, Xiao Z, Liu F Y, Hua Y, Yuan Y B, Ding L M 2019 Sci. Bull. 64 885
 Google Scholar
Google Scholar
[130] Zhao D W, Ding L M 2020 Sci. Bull. 65 1144
 Google Scholar
Google Scholar
[131] Zhao D W, Chen C, Wang C L, Junda M M, Song Z N, Grice C R, Yu Y, Li C W, Subedi B, Podraza N J, Zhao X Z, Fang G J, Xiong R G, Zhu K, Yan Y F 2018 Nat. Energy 3 1093
 Google Scholar
Google Scholar
[132] Shen H P, Omelchenko S T, Jacobs D A, et al. 2018 Sci. Adv. 4 eaau9711
 Google Scholar
Google Scholar
[133] Hou F H, Yan L L, Shi B, et al. 2019 ACS Appl. Energy Mater. 2 243
 Google Scholar
Google Scholar
[134] Werner J, Weng C H, Walter A, Fesquet L, Seif J P, De Wolf S, Niesen B, Ballif C 2016 J. Phys. Chem. Lett. 7 161
 Google Scholar
Google Scholar
[135] Sahli F, Kamino B A, Werner J, et al. 2018 Adv. Energy Mater. 8 1701609
 Google Scholar
Google Scholar
[136] Fan R D, Z N, Zhang L, Yang R, Meng Y, Li L W, Guo T, Chen Y H, Xu Z Q, Zheng G H J, Huang Y, Li L, Qin L, Qiu X H, Chen Q, Zhou H P 2017 Sol. RRL 1 1700149
 Google Scholar
Google Scholar
[137] Qiu Z W, Xu Z Q, Li N X, Zhou N, Chen Y H, Wan X X, Liu J L, Li N, Hao X T, Bi P Q, Chen Q, Cao B Q, Zhou H P 2018 Nano Energy 53 798
 Google Scholar
Google Scholar
[138] Zhu S J, Y X, Ren Q S, et al. 2018 Nano Energy 45 280
 Google Scholar
Google Scholar
[139] Zhu S J, Hou F H, Huang W, et al. 2018 Sol. RRL 2 1800176
 Google Scholar
Google Scholar
[140] Hou F H, Han C, Isabella O, et al. 2019 Nano Energy 56 234
 Google Scholar
Google Scholar
[141] Bush K A, Palmstrom A F, Yu Z J, et al. 2017 Nat. Energy 2 17009
 Google Scholar
Google Scholar
[142] Bush K A, Manzoor S, Frohna K, Yu Z J, Raiford J A, Palmstrom A F, Wang H-P, Prasanna R, Bent S F, Holman Z C, McGehee M D 2018 ACS Energy Lett. 3 2173
 Google Scholar
Google Scholar
[143] Marko Jošt, Köhnen E, Morales-Vilches A B, Lipovšek B, Jäger K, Macco B, Al-Ashouri A, Krč J, Korte L, Rech B, Schlatmann R, Topič M, Stannowski B, Albrecht S 2018 Energy Environ. Sci. 11 3511
 Google Scholar
Google Scholar
[144] Sahli F, Werner J, Kamino B A, et al. 2018 Nat. Mater. 17 820
 Google Scholar
Google Scholar
[145] Nogay G, Sahli F, Werner J, Monnard R, Boccard M, Despeisse M, Haug F J, Jeangros Q, Ingenito A, Ballif C 2019 ACS Energy Lett. 4 844
 Google Scholar
Google Scholar
[146] Mazzarella L, Lin Y H, Kirner S, Morales-Vilches A B, Korte L, Albrecht S, Crossland E, Stannowski B, Case C, Snaith H J, Schlatmann R 2019 Adv. Energy Mater. 9 1803241
 Google Scholar
Google Scholar
[147] Chen B, Yu Z J, Manzoor S, Wang S, Weigand W, Yu Z H, Yang G, Ni Z Y, Dai X Z, Holman Z C, Huang J S 2020 Joule 4 850
 Google Scholar
Google Scholar
[148] Berhe T A, Su W N, Chen C H, Pan C J, Cheng J H, Chen H M, Tsai M C, Chen L Y, Dubale A A, Hwang B J 2016 Energy Environ. Sci. 9 323
 Google Scholar
Google Scholar
[149] Li Z, Xiao C X, Yang Y, et al. 2017 Energy Environ. Sci. 10 1234
 Google Scholar
Google Scholar
-
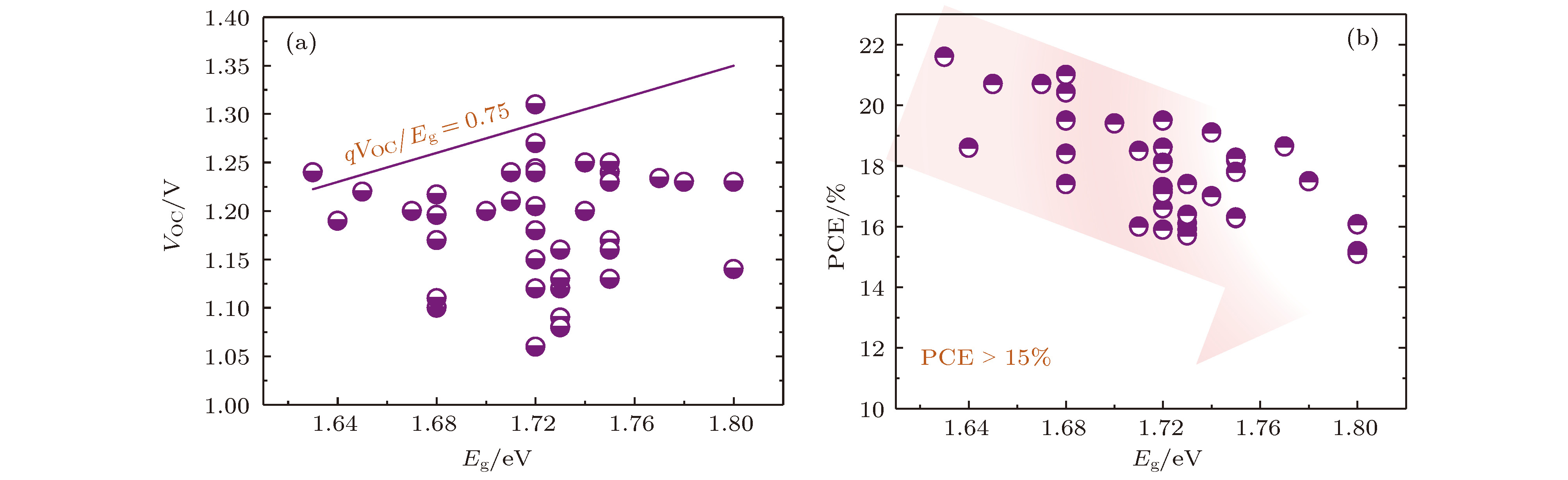
图 1 宽带隙钙钛矿太阳电池性能统计图(Eg ≥ 1.63 eV, PCE > 15%) (a) VOC与Eg之间的关系, 红色阴影部分表示的是qVOC与Eg的比值小于0.75, 其中q表示单位电荷量; (b) PCE和Eg之间的关系
Fig. 1. Performance statistics of WBG-PSCs (Eg ≥ 1.63 eV, PCE > 15%): (a) Relationship between VOC and Eg. The red shaded part indicates that the ratio between qVOC and Eg is less than 0.75, where q represents the unit charge; (b) relationship between PCE and Eg.
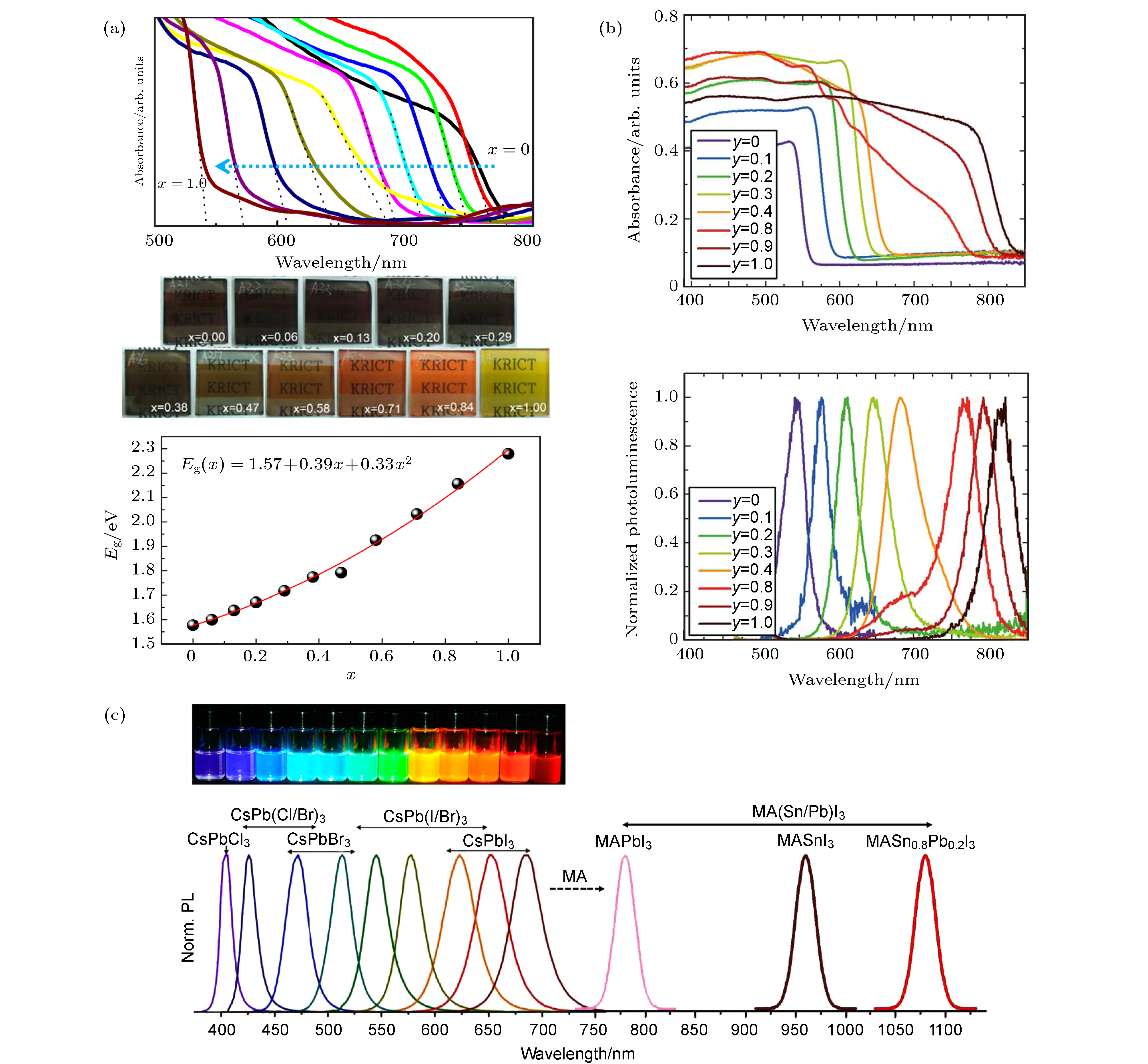
图 3 (a) MAPb(BrxI1–x)3的紫外可见吸收光谱、不同颜色钙钛矿薄膜照片以及带隙随Br含量变化的函数图[70]; (b) FAPb(Br1–yIy)3的紫外可见吸收光谱和光致发光(PL)光谱[71]; (c) CsPb(BrzI1–z)3的钙钛矿溶液和对应的光致发光(PL)谱[79]
Fig. 3. (a) UV-visible absorption spectra, photos of perovskite films with different colors, as well as functional graph between bandgap and bromine content of MAPb(BrxI1–x)3[70]; (b) UV-visible absorption spectra and photoluminescence (PL) spectra of FAPb(Br1–yIy)3[71]; (c) photos of CsPb(BrzI1–z)3 solutions and corresponding PL spectra[79].
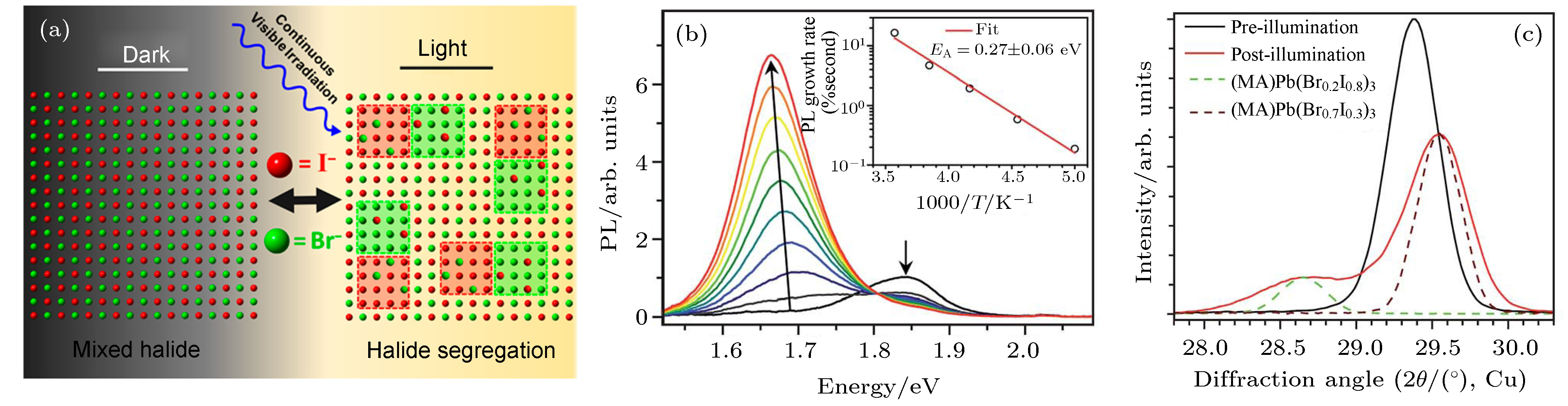
图 4 (a)卤素离子在光照下发生迁移和团簇示意图[90]; (b) MAPb(Br0.4I0.6)3在光照下的光致发光(PL)光谱, 插图表示初始PL增长率的温度依赖性[85]; (c)在约50 mW /cm2的条件下, MAPb(Br0.4I0.6)3膜在白光浸泡5 min前(黑色线)、后(红色线)的XRD图谱, 将MAPb(Br0.2I0.8)3膜(绿色虚线)和MAPb(Br0.7I0.3)3膜(棕色虚线)的XRD图谱进行比较[85]
Fig. 4. (a) Schematic illustration of halogen ion migration and clusters under light[90]; (b) photoluminescence (PL) spectra of MAPb(Br0.4I0.6)3 under light. The illustration shows the temperature dependence of the initial PL growth rate[85]; (c) the XRD pattern of MAPb(Br0.4I0.6)3 film before (black) and after (red) white-light soaking for 5 min at about 50 mW/cm2. XRD patterns of the MAPb(Br0.2I0.8)3 film (dashed green) and the MAPb(Br0.7I0.3)3 film (dashed brown) are included for comparison[85].
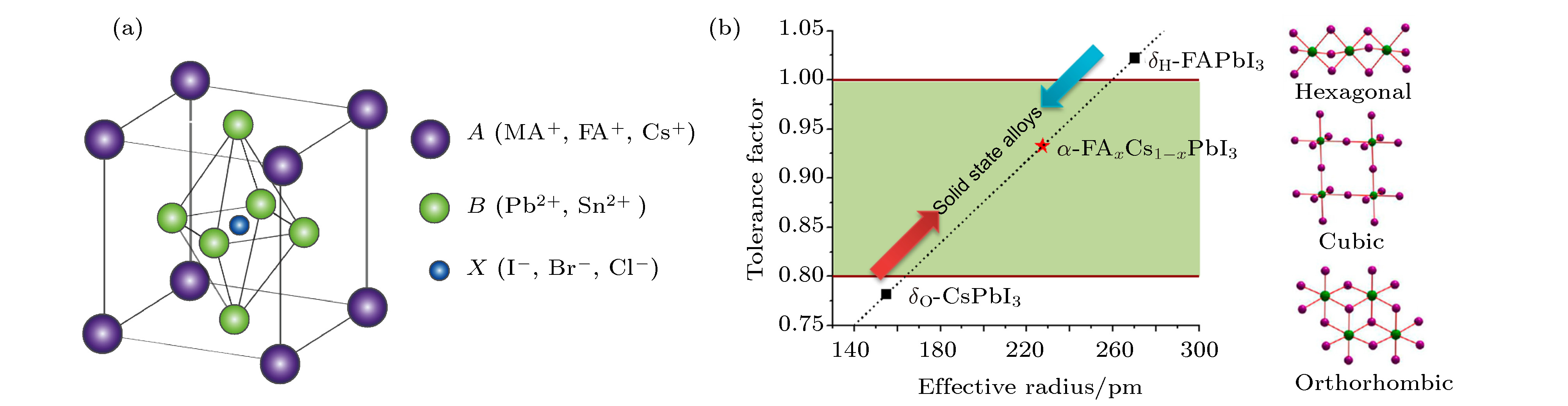
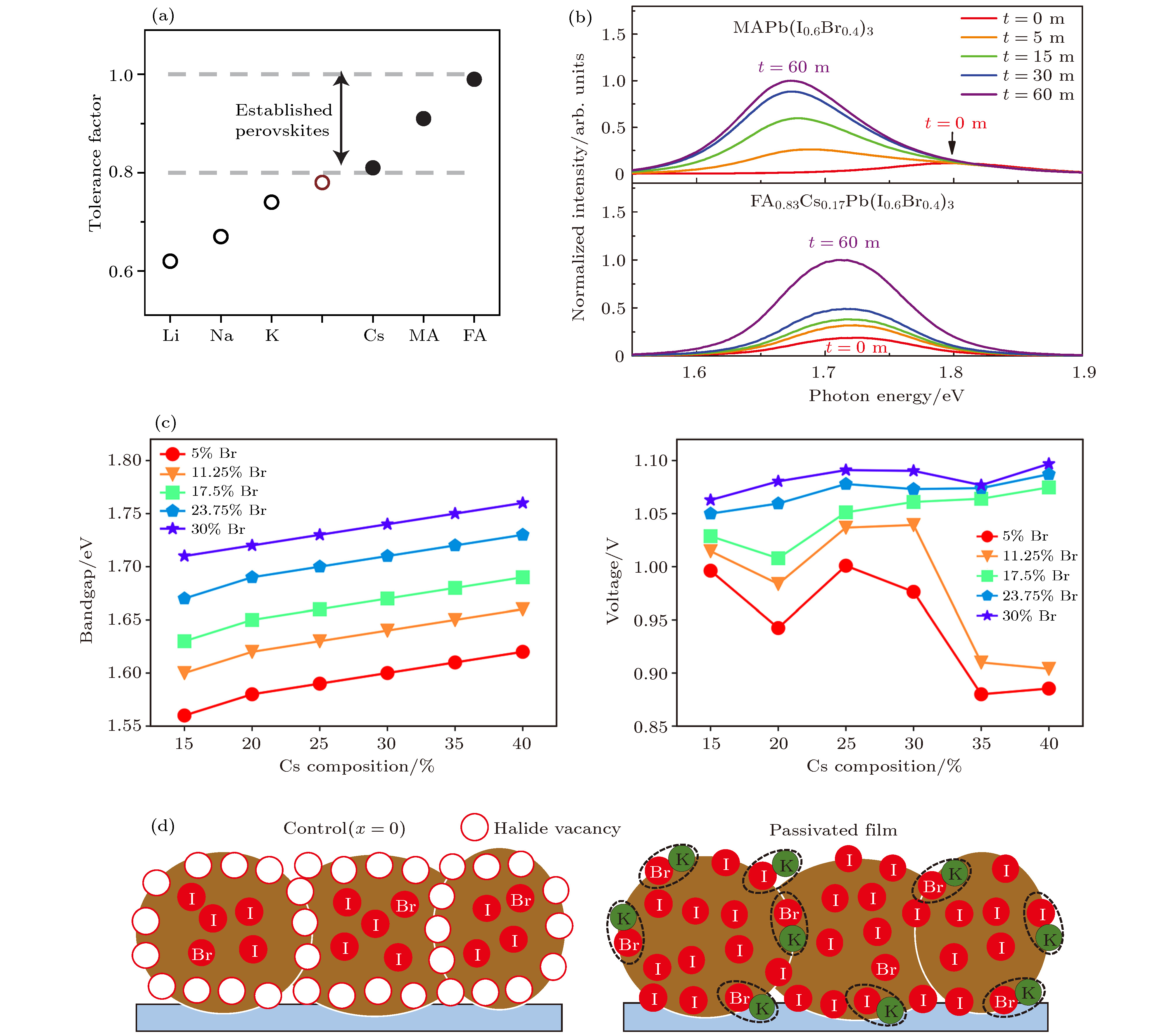
图 5 (a) APbI3钙钛矿的容差因子[56]; (b) FACs基钙钛矿光稳定性明显提高[57]; (c) CsxFA1–xPb(BryI1–y)3材料中的带隙和VOC 变化[37]; (d) K+钝化作用示意图[52]
Fig. 5. (a) Tolerance factor of APbI3[56]; (b) FACs-based perovskite light stability was improved obviously[57]; (c) changes of Eg and VOC in the CsxFA1–xPb(BryI1–y)3 compositions[37]; (d) schematic of K+ passivation[52].
图 6 (a) 3种阳离子的分子构型以及MA+空间旋转的示意图[55]; (b) CsFA和CSMAFA钙钛矿太阳电池最佳J-V曲线和EQE曲线[55]; (c) Cs+和GA+混合到钙钛矿晶格以及对带隙的调控曲线[102]; (d) DMA+对钙钛矿带隙的调整[43]
Fig. 6. (a) The molecular configurations of the three cations and the rotation of MA+ in space[55]; (b) J-V and EQE curves of the best-performing CsFA and CsMAFA PSCs[55]; (c) Cs+ and GA+ are mixed into the perovskite lattice and the tuning curves of the Eg[102]; (d) DMA+ adjusts the Eg of perovskite[43].
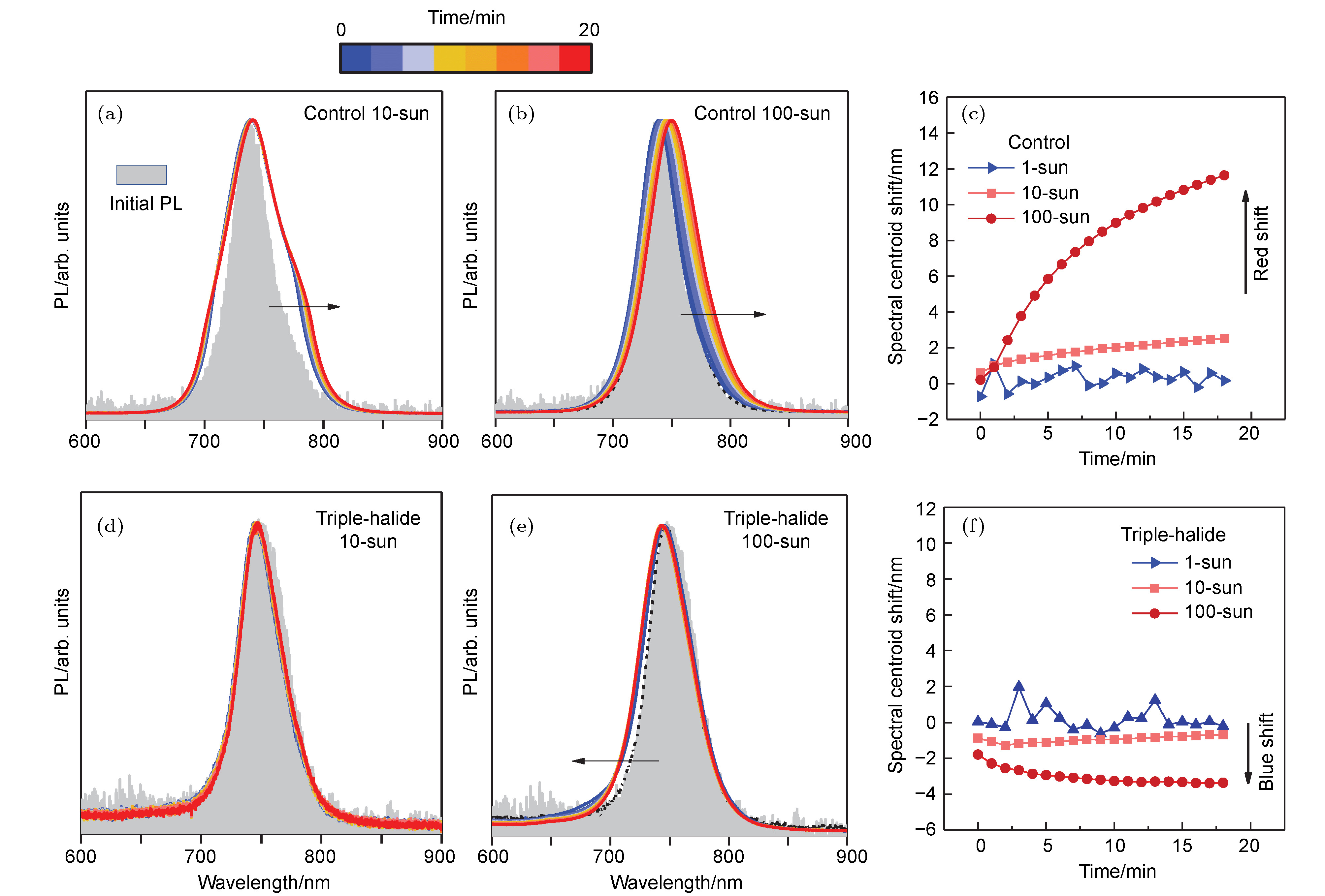
图 8 三卤化物钙钛矿的光稳定性 (a), (b)对照组钙钛矿薄膜(Cs25Br20)经过10倍和100倍太阳光照20 min后的PL光谱, 箭头表示PL峰位随时间变化的方向; (c)对照组薄膜的光谱中心随时间的移动, 在更强光照下, 红移变得更加明显; (d), (e)三卤钙钛矿薄膜(Cs25Br20+Cl3)分别经过10倍和100倍太阳光照20 min后的PL光谱; (f)三卤钙钛矿薄膜的光谱中心随时间的移动, 在更强光照下, 蓝移变得更加明显[44]
Fig. 8. Light stability of triple-halide perovskite: (a), (b) PL spectra of control perovskite films (Cs25Br20) under 10-sun and 100-sun illumination for 20 min, respectively. Arrows indicate the direction of the PL shift over time; (c) the shift of the spectral centroids of control films over time. The red shift becomes more obvious under higher injection; (d), (e) PL spectra of triple-halide perovskites (Cs22Br15+Cl3) under 10-sun and 100-sun illumination for 20 min, respectively; (f) the shift of the spectral centroids of triple-halide perovskites over time. The blue shift becomes more obvious under higher injection[44].
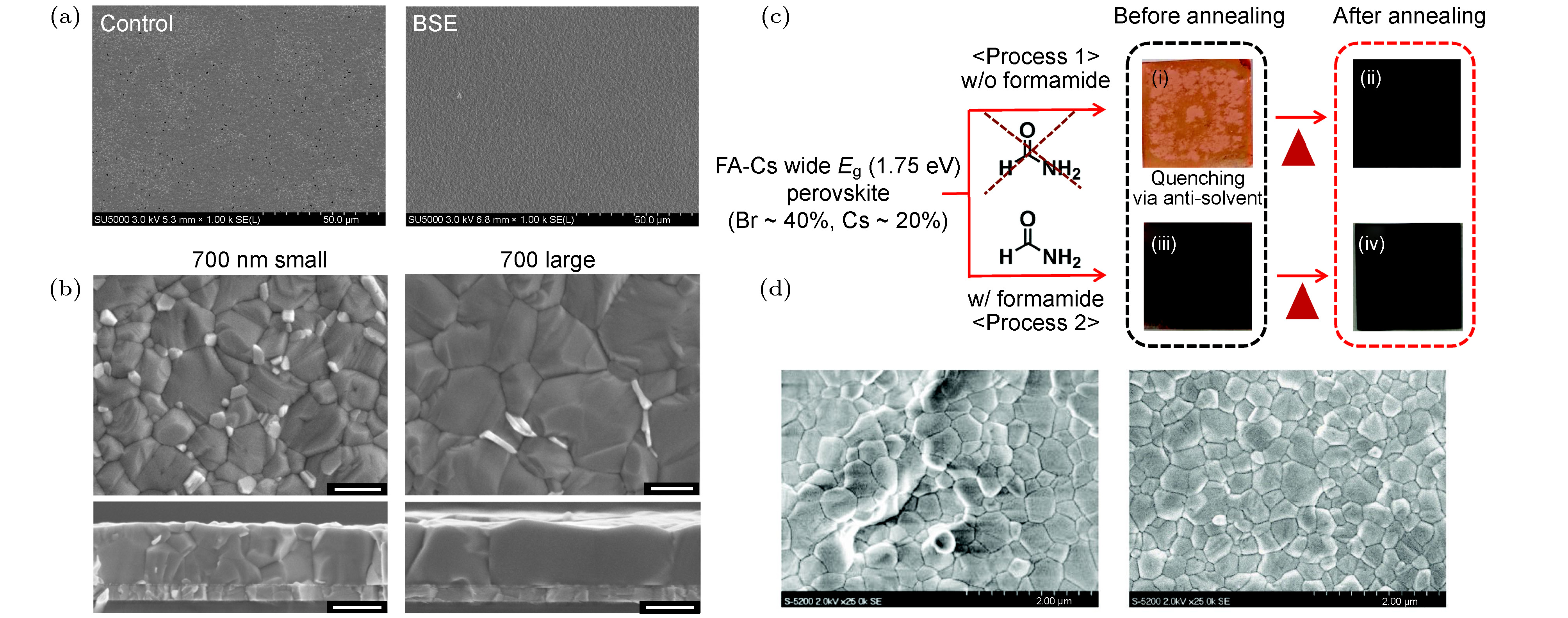
图 10 (a)非加快反溶剂萃取(左)和加快反溶剂萃取(右)制备的钙钛矿薄膜SEM图像[119]; (b)无尿素添加剂(左)和尿素添加剂(右)的700 nm厚钙钛矿薄膜SEM图像[119]; (c)甲酰胺诱导直接形成钙钛矿相, 抑制非钙钛矿相的形成[53]; (d)甲酰胺添加剂提高钙钛矿薄膜结晶质量(右)[53]
Fig. 10. (a) SEM images of perovskite films prepared using no-boosted solvent extraction (BSE) (left) and BSE (right) methods [119]; (b) SEM images of thick perovskite films without urea additives (left) and with urea additives (right) [119]; (c) formamide induces direct formation of perovskite phase and inhibits the formation of non-perovskite phase[53]; (d) improvement of perovskite film crystallization quality by formamide additives (right)[53].
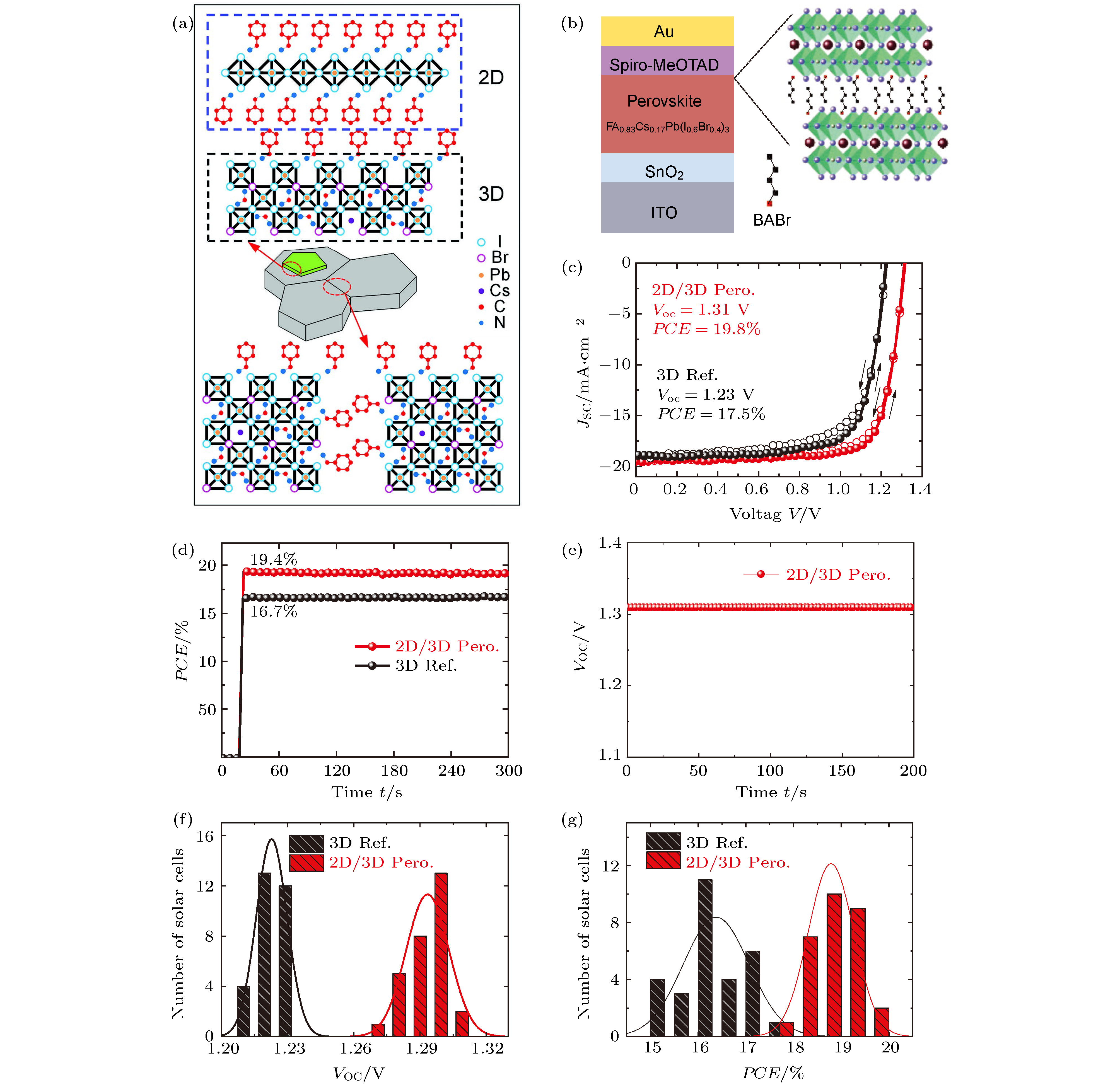
图 11 (a) BA分子与钙钛矿薄膜表面作用示意图[49]; (b)用BABr的异丙醇溶液处理钙钛矿薄膜表面形成二维钙钛矿薄层[59]; (c)经过BABr溶液处理的钙钛矿太阳电池J-V曲线59; (d) BABr溶液处理的钙钛矿太阳电池稳定功率输出曲线(SPCE)59; (e)在连续照明(AM 1.5 G)下测量的最优电池的稳态开路电压(VOC)59; (f), (g)两种电池的开路电压和效率统计[59]
Fig. 11. (a) Schematic of the impact of BA modification on the perovskite film[49]; (b) the perovskite film surface was treated with BABr solution to form a 2D perovskite thin layer[59]; (c) J-V and (d) SPCE curve of PSC with and without BABr treatment[59]; (e) steady-state VOC of the best-performing PSC measured under continuous illumination (AM 1.5 G)[59]; (f), (g) VOC and PCE statistics of two kinds of PSCs[59].
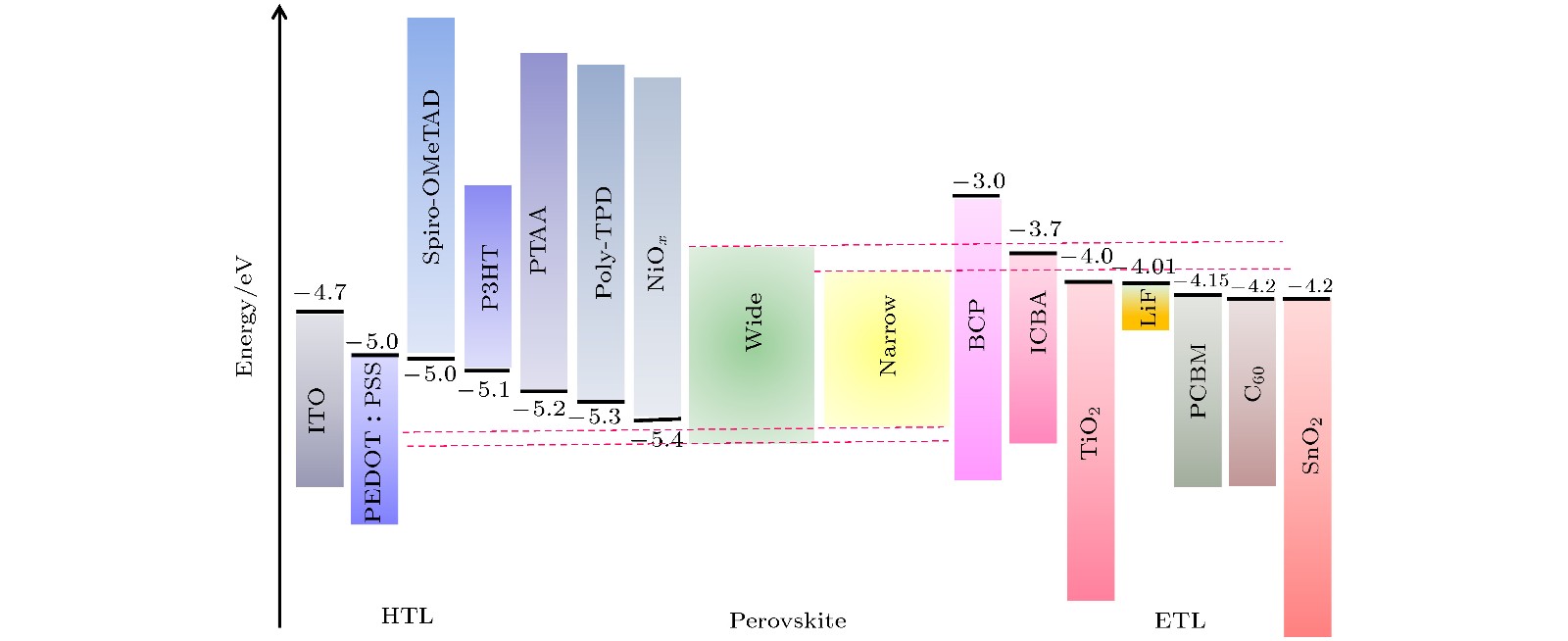
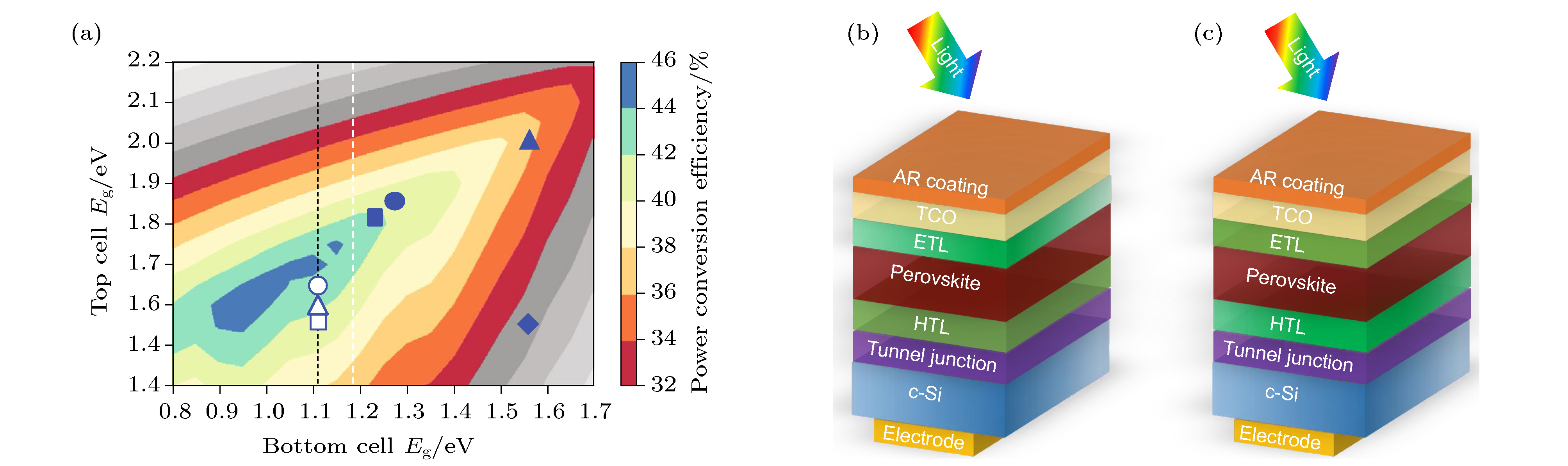
图 13 (a) 2T叠层太阳电池的理论效率图[81]; (b), (c) N-I-P型和P-I-N型2T钙钛矿/硅TSCs结构示意图(TCO: 透明导电氧化物, AR coating: 抗反射膜)
Fig. 13. (a) Theoretical efficiency limit for 2T tandem solar cells; (b), (c) schematics of device structures for N-I-P and P-I-N 2T perovskite/silicon TSCs (TCO: Transparent Conductive Oxide. AR coating: Antireflective coating).
表 1 宽带隙钙钛矿太阳电池性能统计(Eg ≥ 1.63 eV, PCE > 15%)
Table 1. WBG-PSCs performance statistics (Eg ≥ 1.63 eV and PCE > 15%).
Type Perovskite Eg/eV VOC/V qVOC/Eg JSC/mA·cm–2 FF/% PCE/% Ref. p-i-n MAPbI2.5Br0.5 1.72 1.060 0.61 18.30 78.2 16.60 [35] p-i-n (FA0.83MA0.17)0.95Cs0.05Pb(I0.6Br0.4)3 1.71 1.210 0.71 19.70 77.5 18.50 [36] p-i-n FA0.6Cs0.4Pb(I0.7Br0.3)3 1.75 1.170 0.67 17.50 80.0 16.30 [37] p-i-n FA0.83MA0.17Pb(I0.6Br0.4)3 1.72 1.150 0.67 19.40 77.0 17.20 [38] p-i-n FA0.8Cs0.2Pb(I0.7Br0.3)3 1.75 1.240 0.71 17.92 81.9 18.19 [39] p-i-n (FA0.65MA0.20Cs0.15)Pb(I0.8Br0.2)3 1.68 1.170 0.70 21.20 79.8 19.50 [27] p-i-n Cs0.15(FA0.83MA0.17)0.85Pb(I0.8Br0.2)3 1.64 1.190 0.73 19.50 80.2 18.60 [40] p-i-n CsPbI3 1.73 1.160 0.67 17.70 78.6 16.10 [41] p-i-n CsPbI2Br 1.80 1.230 0.67 15.26 78.0 15.19 [42] p-i-n FA0.6Cs0.3DMA0.1PbI2.4Br0.6 1.70 1.200 0.70 19.60 82.0 19.40 [43] p-i-n FA0.75Cs0.25Pb(I0.8Br0.2)3 1.68 1.217 0.72 20.18 83.6 20.42 [44] p-i-n (FA0.65MA0.2Cs0.15)Pb(I0.8Br0.2)3 1.67 1.200 0.72 NA NA 20.70 [45] p-i-n (FA0.64MA0.20Cs0.15)Pb0.99(I0.79Br0.2)3 1.68 1.196 0.71 21.65 81.5 21.00 [46] n-i-p Rb0.05(FA0.75MA0.15Cs0.1)0.95PbI2Br 1.73 1.120 0.71 19.40 73.0 15.90 [47] n-i-p FA0.83Cs0.17Pb(I0.6Br0.4)3 1.75 1.160 0.66 18.27 78.5 16.28 [48] n-i-p FA0.85Cs0.15Pb(I0.73Br0.27)3 1.72 1.240 0.72 19.83 73.7 18.13 [49] n-i-p FA0.8Cs0.2Pb(I0.7Br0.3)3 1.75 1.250 0.71 18.53 79.0 18.27 [50] n-i-p MAPb(Br0.2I0.8)3 1.72 1.120 0.65 17.30 82.3 15.90 [51] n-i-p K0.1(Cs0.06FA0.79MA0.15)0.9Pb(I0.4Br0.6)3 1.78 1.230 0.69 17.90 79.0 17.50 [52] n-i-p FA0.83Cs0.17Pb(I0.6Br0.4)3 1.75 1.230 0.70 18.34 79.0 17.80 [53] n-i-p Cs0.17FA0.83PbI2.2Br0.8 1.72 1.270 0.74 19.30 77.4 18.60 [54] n-i-p Cs0.12MA0.05FA0.83Pb(I0.6Br0.4)3 1.74 1.250 0.72 19.00 81.5 19.10 [55] n-i-p Rb5(Cs5MAFA)95Pb(I0.83Br0.17)3 1.63 1.240 0.76 22.80 81.0 21.60 [56] n-i-p FA0.83Cs0.17Pb(I0.6Br0.4)3 1.74 1.200 0.70 19.40 75.1 17.00 [57] n-i-p FA0.17Cs0.83PbI2.2Br0.8 1.72 1.244 0.72 19.80 75.0 18.60 [51] n-i-p Cs0.2FA0.8Pb(I0.75Br0.25)3 1.65 1.220 0.74 21.20 80.5 20.70 [55] n-i-p BA0.09(FA0.83 Cs0.17)0.91Pb(I0.6Br0.4)3 1.72 1.180 0.69 19.80 73.0 17.30 [38] n-i-p FA0.15Cs0.85Pb(I0.73Br0.27)3 1.72 1.240 0.72 19.83 73.7 18.10 [58] n-i-p FA0.83Cs0.17Pb(I0.6Br0.4)3 1.72 1.310 0.76 19.30 78.0 19.50 [59] n-i-p Rb0.05Cs0.095 MA0.1425 FA0.7125PbI2Br 1.72 1.205 0.70 18.00 78.9 17.10 [54] n-i-p CsPbI3 1.73 1.080 0.62 18.41 79.32 15.71 [60] n-i-p CsPbI2Br 1.80 1.230 0.68 16.79 77.81 16.07 [61] n-i-p β-CsPbI3 1.68 1.110 0.66 20.23 82.0 18.40 [62] n-i-p CsPbI3-xBrx 1.77 1.234 0.69 18.30 82.5 18.64 [63] n-i-p CsPbI2Br 1.80 1.270 0.71 15.40 79.0 15.50 [64] 注: NA表示文献中没有给出具体数值; FF表示填充因子. 序号 钙钛矿中常用离子 有效半径R/pm 1 胍离子(GA+) 278 2 二甲胺离子(DMA+) 272 3 甲脒离子(FA+) 253 4 甲胺离子(MA+) 217 5 铯离子(Cs+) 167 6 铷离子(Rb+) 152 7 钾离子(K+) 138 8 钠离子(Na+) 102 9 铅离子(Pb2+) 119 10 锡离子(Sn2+) 112 11 碘离子(I–) 220 12 溴离子(Br–) 196 13 氯离子(Cl–) 181 表 3 近年来典型的2T钙钛矿/硅TSCs的详细性能参数总结
Table 3. Summary of detailed performance of typical 2T perovskite/silicon TSCs in recent years.
Type Perovskite Eg/eV VOC/V Jsc/mA·cm–2 FF/% PCE/% Year Area/cm2 Ref. N-I-P MAPbI3 1.61 1.580 11.50 75.00 13.70 2015 1.00 [15] FA0.83MA0.17Pb(I0.84Br0.16)3 1.63 1.785 14.00 79.50 19.90 2016 0.16 [133] MAPbI3 1.60 1.692 15.80 79.90 21.40 2016 0.17 [134] MAPbI3 1.60 1.701 16.10 70.10 19.20 2016 1.22 [134] Cs0.19MA0.81PbI3 1.59 1.751 18.80 77.10 22.70 2018 0.25 [135] Cs0.19MA0.81PbI3 1.59 1.779 16.50 74.10 21.70 2018 1.43 [135] Cs0.19FA0.81Pb(I0.78Br0.22)3 1.63 1.769 16.50 65.40 19.10 2018 12.96 [135] MA0.37FA0.48Cs0.15PbI2.01Br0.99 1.69 1.703 15.26 79.20 20.57 2017 0.03 [136] FA0.5MA0.38Cs0.12PbI2.04Br0.96 1.69 1.655 16.50 81.10 22.22 2018 0.06 [137] FA0.75MA0.25 Pb(I0.76B0.24)3 1.65 1.710 15.49 71.00 18.81 2018 0.13 [138] Cs0.08FA0.74MA0.18Pb(I0.88Br0.12)3 1.65 1.780 17.82 75.00 23.73 2018 0.13 [139] Cs0.1(FA0.75MA0.25)0.9Pb(I0.78Br0.22)3 1.67 1.830 16.74 70.00 21.31 2019 0.13 [133] Cs 0.08FA0.69MA0.23Pb(I0.78Br22)3 1.67 1.750 16.89 74.18 21.93 2019 0.13 [140] CsRbFAMAPbI3-xBrx 1.62 1.763 17.80 78.10 24.50 2018 1.00 [132] P-I-N Cs0.17FA0.83Pb(Br0.17I0.83)3 1.63 1.650 18.10 79.00 23.60 2017 1.00 [141] FA 0.75Cs0.25Pb(I0.8Br0.2)3 1.68 1.770 18.40 77.00 25.00 2018 1.00 [142] Cs0.05(MA0.17FA0.83)Pb1.1(I0.83Br0.17)3 1.60 1.760 18.50 78.50 25.50 2018 0.81 [143] CsxFA1-xPb(I, Br)3 1.60 1.788 19.50 73.10 25.20 2018 1.42 [144] CsxFA1-x Pb(I, Br)3 1.60 1.741 19.50 74.70 25.40 2018 1.42 [145] Cs0.15(FA0.83MA0.17)0.85Pb(I0.8Br0.2)3 1.64 1.800 17.80 79.40 25.40 2018 0.49 [40] Cs0.05(FA0.83MA0.17)0.95Pb(I0.82Br0.18)3 1.63 1.792 19.02 74.60 25.43 2019 1.00 [146] Cs0.1MA0.9Pb(I0.9Br0.1)3 1.60 1.820 19.20 75.30 26.20 2020 NA [147] Cs 0.25FA0.75Pb(I0.85Br0.15Cl0.05)3 1.67 1.890 19.10 75.30 27.04 2020 1.00 [44] Cs0.05MA0.15FA0.8Pb(I0.75Br0.25)3 1.68 1.700 19.80 77.00 25.70 2020 0.83 [46] (FA0.65MA0.2Cs0.15)Pb(I0.8Br0.2)3 1.68 1.818 18.90 76.40 26.20 2020 1.00 [45] 注: NA表示文献中没有给出具体数值. -
[1] Xing G C, Mathews N, Lim S S, Yantara N, Liu X F, Sabba D, Grätzel M, Mhaisalkar S, Sum T C 2014 Nat. Mater. 13 476
 Google Scholar
Google Scholar
[2] Tan Z K, Moghaddam R S, Lai M L, Docampo P, Higler R, Deschler F, Price M, Sadhanala A, Pazos L M, Credgington D 2014 Nat. Nanotechnol. 9 687
 Google Scholar
Google Scholar
[3] Dou L, Yang Y, You J B, Hong Z, Chang W H, Li G, Yang Y 2014 Nat. Commun. 5 5404
 Google Scholar
Google Scholar
[4] Kojima A, Teshima K, Shirai Y, Miyasaka T 2009 J. Am. Chem. Soc. 131 6050
 Google Scholar
Google Scholar
[5] Kim M, Kim G H, Lee T K, Choi I W, Choi H W, Jo Y, Yoon Y J, Kim J W, Lee J Y, Huh D, Lee H, Kwak S K, Kim J Y, Kim D S 2019 Joule 3 2179
 Google Scholar
Google Scholar
[6] Jiang Q, Zhao Y, Zhang X W, Yang X L, Chen Y, Chu Z, Ye Q, Li X X, Yin Z G, You J B 2019 Nat. Photonics 13 460
 Google Scholar
Google Scholar
[7] Wang P Y, Li R J, Chen B B, Hou F H, Zhang J, Zhao Y, Zhang X D 2020 Adv. Mater. 32 1905766
 Google Scholar
Google Scholar
[8] Min H, Kim M, Lee S U, Kim H, Kim G, Choi K, Lee J Hee, Seok S I 2019 Science 366 749
 Google Scholar
Google Scholar
[9] Yoo J J, Wieghold S, Sponseller M C, Chua M R, Bertram S N, Hartono N T P, Tresback J S, Hansen E C, Correa-Baena J P, Bulovic V 2019 Energy Environ. Sci. 12 2192
 Google Scholar
Google Scholar
[10] Liu Y H, Akin S, Pan L F, Uchida R, Grätzel M 2019 Sci. Adv. 5 eaaw2543
 Google Scholar
Google Scholar
[11] Zhu P C, Gu S, Luo X, Gao Y, Li S L, Zhu J, Tan H R 2019 Adv. Energy Mater. 10 1903083
 Google Scholar
Google Scholar
[12] Zheng X P, Hou Y, Bao C X, Yin J, Yuan F L, Huang Z R, Song K P, Liu J K, Troughton J, Gasparini N, Zhou C, Lin Y B, Xue D J, Chen B, Johnston A K, Wei N N, Hedhili M N, Wei M, Alsalloum A Y, Maity P, Turedi B, Yang C, Baran D, Anthopoulos T D, Han Y, Lu Z H, Mohammed O F, Gao F, Sargent E H, Bakr O M 2020 Nat. Energy 5 131
 Google Scholar
Google Scholar
[13] Shockley W, Queisser H J 1961 J. Appl. Phys. 32 510
 Google Scholar
Google Scholar
[14] Beiley Z M, McGehee M D 2012 Energy Environ. Sci. 5 9173
 Google Scholar
Google Scholar
[15] Mailoa J P, Bailie C D, Johlin E C, Hoke E T, Akey A J, Nguyen W H, McGehee M D, Buonassisi T 2015 Appl. Phys. Lett. 106 121105
 Google Scholar
Google Scholar
[16] Albrecht S, Saliba M, Correa Baena J P, Lang F, Kegelmann L, Mews M, Steier L, Abate A, Rappich J, Korte L, Schlatmann R, Nazeeruddin M K, Hagfeldt A, Grätzel M, Rech B 2016 Energy Environ. Sci. 9 81
 Google Scholar
Google Scholar
[17] Altazin S, Stepanova L, Werner J, Niesen B, Ballif C, Ruhstaller B 2018 Opt. Express 26 A579
 Google Scholar
Google Scholar
[18] Fang Z M, Wang S Z, Yang S F, Ding L M 2018 Inorg. Chem. Front. 5 1690
 Google Scholar
Google Scholar
[19] Hu W P, He X, Fang Z M, Lian W T, Shang Y B, Li X C, Zhou W R, Zhang M M, Chen T, Lu Y L, Zhang L J, Ding L M, Yang S F 2020 Nano Energy 68 104362
 Google Scholar
Google Scholar
[20] Jia X, Ding L M 2018 Sci. China Mater. 62 54
 Google Scholar
Google Scholar
[21] Zuo C T, Ding L M 2017 Angew. Chem. Int. Ed. 56 6528
 Google Scholar
Google Scholar
[22] Chen B, Zheng X P, Bai Y, Padture N P, Huang J S 2017 Adv. Energy Mater. 7 1602400
 Google Scholar
Google Scholar
[23] Hu J N, Cheng Q, Fan R D, Zhou H P 2017 Sol. RRL 1 1700045
 Google Scholar
Google Scholar
[24] Lal N N, Dkhissi Y, Li W, Hou Q C, Cheng Y B, Bach U 2017 Adv. Energy Mater. 7 1602761
 Google Scholar
Google Scholar
[25] Jošt M, Kegelmann L, Korte L, Albrecht S 2020 Adv. Energy Mater. 10 1904102
 Google Scholar
Google Scholar
[26] Al-Ashouri A, Magomedov A, Roß M, et al. 2019 Energy Environ. Sci. 12 3356
 Google Scholar
Google Scholar
[27] Kim D H, Muzzillo C P, Tong J, et al. 2019 Joule 3 1734
 Google Scholar
Google Scholar
[28] Lin R, Xiao K, Qin Z Y, Han Q L, Zhang C F, Wei M Y, Saidaminov M I, Gao Y, Xu J, Xiao M, Li A D, Zhu J, Sargent E H, Tan H R 2019 Nat. Energy 4 864
 Google Scholar
Google Scholar
[29] McMeekin D P, Mahesh S, Noel N K, Klug M T, Lim J, Warby J H, Ball J M, Herz L M, Johnston M B, Snaith H J 2019 Joule 3 387
 Google Scholar
Google Scholar
[30] Xue Q F, Xia R X, Brabec C J, Yip H L 2018 Energy Environ. Sci. 11 1688
 Google Scholar
Google Scholar
[31] Henemann A 2008 Renew. Energy Focus 9 14
 Google Scholar
Google Scholar
[32] Shi B, Duan L R, Zhao Y, Luo J S, Zhang X D 2020 Adv. Mater. 32 1806474
 Google Scholar
Google Scholar
[33] Park S, Chang W J, Lee C W, Park S, Ahn H Y, Nam K T 2016 Nat. Energy 2 16185
 Google Scholar
Google Scholar
[34] 陈为, 魏伟, 孙予罕 2017 中国科学: 化学 47 1251
 Google Scholar
Google Scholar
Chen W, Wei W, Sun Y H 2017 Sci. China: Chem. 47 1251
 Google Scholar
Google Scholar
[35] Hu M, Bi C, Yuan Y B, Bai Y, Huang J S 2016 Adv. Sci. 3 1500301
 Google Scholar
Google Scholar
[36] Lin Y Z, Chen B, Zhao F W, Zheng X P, Deng Y H, Shao Y C, Fang Y J, Bai Y, Wang C R, Huang J S 2017 Adv. Mater. 29 1700607
 Google Scholar
Google Scholar
[37] Bush K A, Frohna K, Prasanna R, Beal R E, Leijtens T, Swifter S A, McGehee M D 2018 ACS Energy Lett. 3 428
 Google Scholar
Google Scholar
[38] Wang Z P, Lin Q Q, Chmiel F P, Sakai N, Herz L M, Snaith H J 2017 Nat. Energy 2 17135
 Google Scholar
Google Scholar
[39] Chen C, Song Z N, Xiao C X, Zhao D W, Shrestha N, Li C W, Yang G, Yao F, Zheng X L, Ellingson R J, Jiang C S, Al-Jassim M, Zhu K, Fang G J, Yan Y F 2019 Nano Energy 61 141
 Google Scholar
Google Scholar
[40] Chen B, Yu Z S, Liu K, Zheng X P, Liu Y, Shi J W, Spronk D, Rudd P N, Holman Z, Huang J S 2019 Joule 3 177
 Google Scholar
Google Scholar
[41] Wang J, Zhang J, Zhou Y Z, Liu H B, Xue Q F, Li X S, Chueh C C, Yip L P, Zhu Z L, Jen A K Y 2020 Nat. Commun. 11 177
 Google Scholar
Google Scholar
[42] Liu C, Yang Y Z, Zhang C L, Wu S H, Wei L Y, Guo F, Arumugam G M, Hu J L, Liu X Y, Lin J, Schropp R E L, Mai Y H 2020 Adv. Mater. 32 1907361
 Google Scholar
Google Scholar
[43] Palmstrom A F, Eperon G E, Leijtens T, et al. 2019 Joule 3 2193
 Google Scholar
Google Scholar
[44] Xu J X, Boyd C C, Yu Z J, et al. 2020 Science 367 1097
 Google Scholar
Google Scholar
[45] Kim D, Jung H J, Park I J, Larson B W, Dunfield S P, Xiao C X, Kim J, Tong J H, Boonmongkolras P, Ji S G, Zhang F, Pae S R, Kim M, Kang S B, Dravid V, Berry J J, Kim J Y, Zhu K, Kim D H, Shin B 2020 Science 368 155
 Google Scholar
Google Scholar
[46] Ye J Y, Tong J H, Hu J, et al. 2020 Sol. RRL 4 2000082
 Google Scholar
Google Scholar
[47] Duong T, Wu Y L, Shen H P, et al. 2017 Adv. Energy Mater. 7 1700228
 Google Scholar
Google Scholar
[48] Yang M J, Kim D H, Yu Y, Li Z, Reid O G, Song Z N, Zhao D W, Wang C L, Li L W, Meng Y, Guo T, Yan Y F, Zhu K 2018 Mater. Today Energy 7 232
 Google Scholar
Google Scholar
[49] Zhou Y, Wang F, Cao Y, Wang J P, Fang H H, Loi M A, Zhao N, Wong C P 2017 Adv. Energy Mater. 7 1701048
 Google Scholar
Google Scholar
[50] Yu Y, Wang C L, Grice C R, Shrestha N, Zhao D W, Liao W Q, Guan L, Awni R A, Meng W W, Cimaroli A J, Zhu K, Ellingson R J, Yan Y F 2017 ACS Energy Lett. 2 1177
 Google Scholar
Google Scholar
[51] Zhou Y, Jia Y H, Fang H H, Loi M A, Xie F Y, Gong L, Qin M C, Lu X H, Wong C P, Zhao N 2018 Adv. Funct. Mater. 28 1803130
 Google Scholar
Google Scholar
[52] Abdi-Jalebi M, Andaji-Garmaroudi Z, Cacovich S, et al. 2018 Nature 555 497
 Google Scholar
Google Scholar
[53] Kim J, Saidaminov M I, Tan H R, et al. 2018 Adv. Mater. 30 1706275
 Google Scholar
Google Scholar
[54] Duong T, Pham H, Kho T H, et al. 2019 Adv. Energy Mater. 10 1903553
 Google Scholar
Google Scholar
[55] Tan H R, Che F L, Wei M Y, Zhao Y C, Saidaminov M I, Petar T, Danny B, Grant W, Tan F R, Zhuang T T 2018 Nat. Commun. 9 3100
 Google Scholar
Google Scholar
[56] Saliba M, Matsui T, Domanski K, Seo J Y, Ummadisingu A, Zakeeruddin S M, Correa-Baena J P, Tress W R, Abate A, Hagfeldt A, Grätzel M 2016 Science 354 206
 Google Scholar
Google Scholar
[57] McMeekin D P, Sadoughi G, Rehman W, Eperon G E, Saliba M, Hörantner M T, Haghighirad A, Sakai N, Korte L, Rech B, Johnston M B, Herz L M, Snaith H J 2016 Science 351 151
 Google Scholar
Google Scholar
[58] Zhuang J, Mao P, Luan Y G, Yi X H, Tu Z Y, Zhang Y Y, Yi Y P, Wei Y Z, Chen N L, Lin T, Wang F Y, Li C, Wang J Z 2019 ACS Energy Lett. 4 2913
 Google Scholar
Google Scholar
[59] Gharibzadeh S, Abdollahi Nejand B, Jakoby M, et al. 2019 Adv. Energy Mater. 9 1803699
 Google Scholar
Google Scholar
[60] Wang P Y, Zhang X W, Zhou Y Q, Jiang Q, Ye Q F, Chu Z M, Li X X, Yang X L, Yin Z G, You J B 2018 Nat. Commun. 9 2225
 Google Scholar
Google Scholar
[61] Zhang J, Bai D L, Jin Z W, Bian H, Wang K, Sun J, Wang Q, Liu S Z F 2018 Adv. Energy Mater. 8 1703246
 Google Scholar
Google Scholar
[62] Wang Y, Dar M I, Ono L K, Zhang T Y, Kan M, Li Y W, Zhang L J, Wang X T, Yang Y G, Gao X Y, Qi Y B, Grätzel M, Zhao Y X 2019 Science 365 591
 Google Scholar
Google Scholar
[63] Ye Q F, Zhao Y, Mu S Q, Ma F, Gao F, Chu Z M, Yin Z G, Gao P Q, Zhang X W, You J B 2019 Adv. Mater. 1 1905143
 Google Scholar
Google Scholar
[64] Xiao Q, Tian J J, Xue Q F, Wang J, Xiong B J, Han M M, Li Z, Zhu Z L, Yip H L, Li Z 2019 Angew. Chem. Int. Ed. 58 17724
 Google Scholar
Google Scholar
[65] Green M A, Ho-Baillie A, Snaith H J 2014 Nat. Photonics 8 506
 Google Scholar
Google Scholar
[66] Kim H S, Im S H, Park N G 2014 J. Phys.Chem. C 118 5615
 Google Scholar
Google Scholar
[67] Li Z, Yang M J, Park J S, Wei S H, Berry J, Zhu K 2015 Chem. Mater. 28 284
 Google Scholar
Google Scholar
[68] Chang Y H, Park C H 2004 J. Korean Phys. Soc. 44 889
[69] Anaya M, Correabaena J P, Lozano G L, Saliba M, Anguita P, Roose B, Abate A, Steiner U, Grätzel M, Calvo M 2016 J. Mater. Chem. A 4 11214
 Google Scholar
Google Scholar
[70] Noh J H, Im S H, Heo J H, Mandal T N, Seok S I 2013 Nano Lett. 13 1764
 Google Scholar
Google Scholar
[71] Eperon G E, Stranks S D, Menelaou C, Johnston M B, Herz L M, Snaith H J 2014 Energy Environ. Sci. 7 982
 Google Scholar
Google Scholar
[72] Saliba M, Correa-Baena J P, Grätzel M, Hagfeldt A, Abate A 2017 Angew. Chem. Int. Ed. 57 2554
 Google Scholar
Google Scholar
[73] Unger E L, Bowring A R, Tassone C J, Pool V L, Gold-Parker A, Cheacharoen R, Stone K H, Hoke E T, Toney M F, McGehee M D 2014 Chem. Mater. 26 7158
 Google Scholar
Google Scholar
[74] Dong Q, Yuan Y B, Shao Y C, Fang Y J, Wang Q, Huang J S 2015 Energy Environ. Sci. 8 2464
 Google Scholar
Google Scholar
[75] Lee B, Hwang T, Lee S, Shin B, Park B 2019 Sci. Rep. 9 4803
 Google Scholar
Google Scholar
[76] Zhang C P, Li Z P, Liu J, Xin Y C, Shao Z P, Cui G, Pang S P 2018 ACS Energy Lett. 3 1801
 Google Scholar
Google Scholar
[77] Ma T, Wang S W, Zhang Y W, Zhang K X, Yi L X 2020 J. Mater. Sci. 55 464
 Google Scholar
Google Scholar
[78] Tong G, Ono L K, Qi Y B 2019 Energy Technol. 8 1900961
 Google Scholar
Google Scholar
[79] Protesescu L, Yakunin S, Bodnarchuk M I, et al. 2015 Nano Lett. 15 3692
 Google Scholar
Google Scholar
[80] Mehrabian M, Dalir S, Mahmoudi G, Miroslaw B, Safin D A 2019 Eur. J. Inorg. Chem. 2019 3699
 Google Scholar
Google Scholar
[81] Leijtens T, Bush K A, Prasanna R, McGehee M D 2018 Nat. Energy 3 828
 Google Scholar
Google Scholar
[82] Mahesh S, Ball J M, Oliver R D J, McMeekin D P, Nayak P K, Johnston M B, Snaith H J 2020 Energy Environ. Sci. 13 258
 Google Scholar
Google Scholar
[83] Levine I, Vera O G, Kulbak M, Ceratti D-R, Rehermann C, Márquez J A, Levcenko S, Unold T, Hodes G, Balberg I, Cahen D, Dittrich T 2019 ACS Energy Lett. 4 1150
 Google Scholar
Google Scholar
[84] Song Z, Chen C, Li C, Awni R A, Zhao D, Yan Y 2019 Semicond. Sci. Technol. 34 093001
 Google Scholar
Google Scholar
[85] Hoke E T, Slotcavage D J, Dohner E R, Bowring A R, Karunadasa H I, McGehee M D 2015 Chem. Sci. 6 613
 Google Scholar
Google Scholar
[86] Slotcavage D J, Karunadasa H I, McGehee M D 2016 ACS Energy Lett. 1 1199
 Google Scholar
Google Scholar
[87] Yun J S, Seidel J, Kim J, Soufiani A M, Huang S, Lau J, Jeon N J, Seok S I, Green M A, Ho-Baillie A 2016 Adv. Energy Mater. 6 1600330
 Google Scholar
Google Scholar
[88] Beal R E, Hagström N Z, Barrier J, Gold-Parker A, Prasanna R, Bush K A, Passarello D, Schelhas L T, Brüning K, Tassone C J, Steinrück H G, McGehee M D, Toney M F, Nogueira A F 2020 Matter 2 207
 Google Scholar
Google Scholar
[89] Bischak C G, Hetherington C L, Wu H, Aloni S, Ogletree D F, Limmer D T, Ginsberg N S 2017 Nano Lett. 17 1028
 Google Scholar
Google Scholar
[90] Brennan M C, Draguta S, Kamat P V, Kuno M 2017 ACS Energy Lett. 3 204
 Google Scholar
Google Scholar
[91] Liu S, Guan Y J, Sheng Y S, Hu Y, Rong Y G, Mei A Y, Han H W 2020 Adv. Energy Mater. 10 1902492
 Google Scholar
Google Scholar
[92] Gao F, Zhao Y, Zhang X W, You J B 2020 Adv. Energy Mater. 10 1902650
 Google Scholar
Google Scholar
[93] Han J H, Luo S P, Yin X W, Zhou Y, Nan H, Li J B, Li X, Oron D, Shen H P, Lin H 2018 Small 14 1801016
 Google Scholar
Google Scholar
[94] Bai S, Da P M, Li C, Wang Z P, Yuan Z C, Fu F, Kawecki M, Liu X J, Sakai N, Wang J T W, Huettner S, Buecheler S, Fahlman M, Gao F, Snaith H J 2019 Nature 571 245
 Google Scholar
Google Scholar
[95] Bi D Q, Yi C Y, Luo J S, Décoppet J D, Zhang F, Zakeeruddin Shaik M, Li X, Hagfeldt A, Grätzel M 2016 Nat. Energy 1 16142
 Google Scholar
Google Scholar
[96] Noel N K, Abate A, Stranks S D, Parrott E S, Burlakov V M, Goriely A, Snaith H J 2014 ACS Nano 8 9815
 Google Scholar
Google Scholar
[97] Correa-Baena J B, Luo Y Q, Brenner T M, Snaider J, Sun S J, Li X Y, Jensen M A, Hartono N P T, Nienhaus L, Wieghold S, Poindexter J R, Wang S, Meng Y S, Wang T, Lai B, Holt M V, Cai Z H, Bawendi M G, Huang L B, Buonassisi T, Fenning D P 2019 Science 363 627
 Google Scholar
Google Scholar
[98] Kieslich G, Sun S, Cheetham A K 2014 Chem. Sci. 12 4712
 Google Scholar
Google Scholar
[99] Kubicki D, Prochowicz D, Hofstetter A, Saski M, Yadav P, Bi D, Pellet N, Lewiński J, Zakeeruddin S M, Grätzel M 2018 J. Mater. Chem. A 140 3345
 Google Scholar
Google Scholar
[100] Jodlowski A D, Roldán-Carmona C, Grancini G, Salado M, Ralaiarisoa M, Ahmad S, Koch N, Camacho L, de Miguel G, Nazeeruddin M K 2017 Nat. Energy 2 972
 Google Scholar
Google Scholar
[101] Chen H, Wei Q, Saidaminov M I, Wang F, Johnston A, Hou Y, Peng Z J, Xu K M, Zhou W J, Liu Z H, Qiao L, Wang X, Xu S W, Li J Y, Long R, Ke Y Q, Sargent E H, Ning Z J 2019 Adv. Mater. 31 e1903559
 Google Scholar
Google Scholar
[102] Stoddard R J, Rajagopal A, Palmer R L, Braly I L, Jen A K Y, Hillhouse H W 2018 ACS Energy Lett. 3 1261
 Google Scholar
Google Scholar
[103] Yu H, Wang F, Xie F Y, Li W W, Chen J, Zhao N 2014 Adv. Funct. Mater. 24 7102
 Google Scholar
Google Scholar
[104] Chae J S, Dong Q F, Huang J S, Centrone A 2015 Nano Lett. 15 8114
 Google Scholar
Google Scholar
[105] Gao C, Liu J, Liao C, Ye Q Y, Zhang Y Z, He X L, Guo X W, Mei J, Lau W 2015 RSC Adv. 5 26175
 Google Scholar
Google Scholar
[106] Xie Y L, Yu H Y, Duan J S, Xu L, Hu B 2020 ACS Appl. Mater. Interfaces 12 11190
 Google Scholar
Google Scholar
[107] Abate A, Saliba M, Hollman D J, Stranks S D, Wojciechowski K, Avolio R, Grancini G, Petrozza A, Snaith H J 2014 Nano Lett. 14 3247
 Google Scholar
Google Scholar
[108] Heo D Y, Lee T H, Iwan A, Kavan L, Omatova M, Majkova E, Kamarás K, Jang H W, Kim S Y 2020 J. Power Sources 458 228067
 Google Scholar
Google Scholar
[109] Tong J H, Song Z N, Kim D H, et al. 2019 Science 364 475
 Google Scholar
Google Scholar
[110] Tang G, Yang C, Stroppa A, Fang D N, Hong J W 2017 J. Chem. Phys. 146 224702
 Google Scholar
Google Scholar
[111] Ke W J, Xiao C X, Wang C L, et al. 2016 Adv. Mater. 28 5214
 Google Scholar
Google Scholar
[112] Xu J X, Buin A, Ip A H, et al. 2015 Nat. Commun. 6 7081
 Google Scholar
Google Scholar
[113] Liang P W, Chueh C C, Williams S T, Jen A K Y 2015 Adv. Energy Mater. 5 1402321
 Google Scholar
Google Scholar
[114] Gatti T, Menna E, Meneghetti M, Maggini M, Petrozza A, Lamberti F 2017 Nano Energy 41 84
 Google Scholar
Google Scholar
[115] Fang Y J, Bi C, Wang D, Huang J S 2017 ACS Energy Lett. 2 782
 Google Scholar
Google Scholar
[116] Cui C H, Li Y W, Li Y F 2017 Adv. Energy Mater. 7 1601251
 Google Scholar
Google Scholar
[117] Lee J W, Park N G 2019 Adv. Energy Mater. 10 1903249
 Google Scholar
Google Scholar
[118] Lee J W, Kim H S, Park N G 2016 Acc. Chem. Res. 49 311
 Google Scholar
Google Scholar
[119] Xue D J, Hou Y, Liu S C, Wei M, Chen B, Huang Z, Li Z, Sun B, Proppe A H, Dong Y, Saidaminov M I, Kelley S O, Hu J S, Sargent E H 2020 Nat. Commun. 11 1514
 Google Scholar
Google Scholar
[120] Jan S, Robby P, Rolf B 2018 Sol. Energy Mater. Sol. Cells 187 39
 Google Scholar
Google Scholar
[121] Luo D Y, Yang W Q, Wang Z P, et al. 2018 Science 360 1442
 Google Scholar
Google Scholar
[122] Bu X N, Westbrook R J E, Lanzetta L, Ding D, Chotchuangchutchaval T, Aristidou N, Haque S A 2019 Sol. RRL 3 1800282
 Google Scholar
Google Scholar
[123] Wang Q, Zheng X P, Deng Y H, Zhao J J, Chen Z L, Huang J S 2017 Joule 1 371
 Google Scholar
Google Scholar
[124] Zheng X P, Chen B, Dai J, Fang Y J, Bai Y, Lin Y Z, Wei H T, Zeng X C, Huang J S 2017 Nat. Energy 2 17102
 Google Scholar
Google Scholar
[125] Elgamel H E, Barnett A M, Rohatgi A, Chen Z, Vinckier C, Nijs J, Mertens R 1995 J. Appl. Phys. 78 3457
 Google Scholar
Google Scholar
[126] Jaysankar M, Raul B A L, Bastos J, Burgess C, Weijtens C, Creatore M, Aernouts T, Kuang Y, Gehlhaar R, Hadipour A, Poortmans J 2018 ACS Energy Lett. 4 259
 Google Scholar
Google Scholar
[127] Stolterfoht M, Caprioglio P, Wolff C M, et al. 2019 Energy Environ. Sci. 12 2778
 Google Scholar
Google Scholar
[128] Bian H, Bai D L, Jin Z W, Wang K, Liang L, Wang H R, Zhang J R, Wang Q, Liu S Z F 2018 Joule 2 1500
 Google Scholar
Google Scholar
[129] Zeng Q, Liu L, Xiao Z, Liu F Y, Hua Y, Yuan Y B, Ding L M 2019 Sci. Bull. 64 885
 Google Scholar
Google Scholar
[130] Zhao D W, Ding L M 2020 Sci. Bull. 65 1144
 Google Scholar
Google Scholar
[131] Zhao D W, Chen C, Wang C L, Junda M M, Song Z N, Grice C R, Yu Y, Li C W, Subedi B, Podraza N J, Zhao X Z, Fang G J, Xiong R G, Zhu K, Yan Y F 2018 Nat. Energy 3 1093
 Google Scholar
Google Scholar
[132] Shen H P, Omelchenko S T, Jacobs D A, et al. 2018 Sci. Adv. 4 eaau9711
 Google Scholar
Google Scholar
[133] Hou F H, Yan L L, Shi B, et al. 2019 ACS Appl. Energy Mater. 2 243
 Google Scholar
Google Scholar
[134] Werner J, Weng C H, Walter A, Fesquet L, Seif J P, De Wolf S, Niesen B, Ballif C 2016 J. Phys. Chem. Lett. 7 161
 Google Scholar
Google Scholar
[135] Sahli F, Kamino B A, Werner J, et al. 2018 Adv. Energy Mater. 8 1701609
 Google Scholar
Google Scholar
[136] Fan R D, Z N, Zhang L, Yang R, Meng Y, Li L W, Guo T, Chen Y H, Xu Z Q, Zheng G H J, Huang Y, Li L, Qin L, Qiu X H, Chen Q, Zhou H P 2017 Sol. RRL 1 1700149
 Google Scholar
Google Scholar
[137] Qiu Z W, Xu Z Q, Li N X, Zhou N, Chen Y H, Wan X X, Liu J L, Li N, Hao X T, Bi P Q, Chen Q, Cao B Q, Zhou H P 2018 Nano Energy 53 798
 Google Scholar
Google Scholar
[138] Zhu S J, Y X, Ren Q S, et al. 2018 Nano Energy 45 280
 Google Scholar
Google Scholar
[139] Zhu S J, Hou F H, Huang W, et al. 2018 Sol. RRL 2 1800176
 Google Scholar
Google Scholar
[140] Hou F H, Han C, Isabella O, et al. 2019 Nano Energy 56 234
 Google Scholar
Google Scholar
[141] Bush K A, Palmstrom A F, Yu Z J, et al. 2017 Nat. Energy 2 17009
 Google Scholar
Google Scholar
[142] Bush K A, Manzoor S, Frohna K, Yu Z J, Raiford J A, Palmstrom A F, Wang H-P, Prasanna R, Bent S F, Holman Z C, McGehee M D 2018 ACS Energy Lett. 3 2173
 Google Scholar
Google Scholar
[143] Marko Jošt, Köhnen E, Morales-Vilches A B, Lipovšek B, Jäger K, Macco B, Al-Ashouri A, Krč J, Korte L, Rech B, Schlatmann R, Topič M, Stannowski B, Albrecht S 2018 Energy Environ. Sci. 11 3511
 Google Scholar
Google Scholar
[144] Sahli F, Werner J, Kamino B A, et al. 2018 Nat. Mater. 17 820
 Google Scholar
Google Scholar
[145] Nogay G, Sahli F, Werner J, Monnard R, Boccard M, Despeisse M, Haug F J, Jeangros Q, Ingenito A, Ballif C 2019 ACS Energy Lett. 4 844
 Google Scholar
Google Scholar
[146] Mazzarella L, Lin Y H, Kirner S, Morales-Vilches A B, Korte L, Albrecht S, Crossland E, Stannowski B, Case C, Snaith H J, Schlatmann R 2019 Adv. Energy Mater. 9 1803241
 Google Scholar
Google Scholar
[147] Chen B, Yu Z J, Manzoor S, Wang S, Weigand W, Yu Z H, Yang G, Ni Z Y, Dai X Z, Holman Z C, Huang J S 2020 Joule 4 850
 Google Scholar
Google Scholar
[148] Berhe T A, Su W N, Chen C H, Pan C J, Cheng J H, Chen H M, Tsai M C, Chen L Y, Dubale A A, Hwang B J 2016 Energy Environ. Sci. 9 323
 Google Scholar
Google Scholar
[149] Li Z, Xiao C X, Yang Y, et al. 2017 Energy Environ. Sci. 10 1234
 Google Scholar
Google Scholar
计量
- 文章访问数: 44621
- PDF下载量: 1525
- 被引次数: 0













 下载:
下载: