-
铅卤钙钛矿可实现溶液法制备且具有诸多优异的光电特性, 在高效太阳能电池、发光二极管、激光、光探测器等光电子领域具有广阔的应用前景. 但是铅卤钙钛矿在光辐照下存在不稳定问题, 严重影响其相关光电器件的寿命和性能稳定性. 因此, 铅卤钙钛矿在持续光照下的不稳定性现象及其机理正受到越来越多的关注. 本文综述了铅卤钙钛矿在持续光照下的四类主要不稳定现象, 即光修复现象、光解现象、光致相分离现象以及光致相变现象, 并介绍目前已提出的相关机理, 分别从缺陷态、离子迁移、热力学原理、化学键等角度来解释其光照不稳定性. 最后, 本文简要讨论了钙钛矿中光稳定性研究的复杂性及未来需要解决的问题.Lead halide perovskites, which can be grown by solution process, are very suitable for various optoelectronic applications, such as in solar cells, light-emitting diodes, lasers and photo-detectors, due to their outstanding electrical and optical properties. However, lead halide perovskites are unstable under continuous light illumination, which severely degrades the performance and lifetime of optoelectronic devices based on them. Therefore, the photo-stability of perovskites and the mechanisms have received more and more attention. In this article we review the main phenomena in perovskites, induced by continuous light illumination, namely photo curing, photo dissociation, photo-induced phase separation, and photo-induced phase transition. The proposed mechanisms for the instability from the perspectives of defect states, ion migration, thermodynamics and chemical bonds, respectively, are also introduced. Finally, the complexity presented in the studying of photo-stability, and the issues should be addressed in the future are also briefly discussed.
-
Keywords:
- lead halide perovskite /
- photo-stability /
- mechanisms
[1] Sutherland B R, Sargent E H 2016 Nat. Photon. 10 295
 Google Scholar
Google Scholar
[2] Heo J H, Im S H, Noh J H, Mandal T N, Lim C S, Chang J A, Lee Y H, Kim H J, Sarkar A, Nazeeruddin M K, Grätzel M, Seok S I 2013 Nat. Photon. 7 486
 Google Scholar
Google Scholar
[3] Giorgi G, Fujisawa J I, Segawa H, Yamashita K 2013 J. Phys. Chem. Lett. 4 4213
 Google Scholar
Google Scholar
[4] Bisquert J 2013 J. Phys. Chem. Lett. 4 1051
 Google Scholar
Google Scholar
[5] Etgar L, Gao P, Xue Z, Peng Q, Chandiran A K, Liu B, Nazeeruddin M K, Grätzel M 2012 J. Am. Chem. Soc. 134 17396
 Google Scholar
Google Scholar
[6] Abu Laban W, Etgar L 2013 Energy Environ. Sci. 6 3249
 Google Scholar
Google Scholar
[7] Xing G, Mathews N, Sun S, Lim S S, Lam Y M, Grätzel M, Mhaisalkar S, Sum T C 2013 Science 342 344
 Google Scholar
Google Scholar
[8] Stranks S D, Eperon G E, Grancini G, Menelaou C, Alcocer M J P, Leijtens T, Herz L M, Petrozza A, Snaith H J 2013 Science 342 341
 Google Scholar
Google Scholar
[9] Yamada Y, Nakamura T, Endo M, Wakamiya A, Kanemitsu Y 2014 J. Am. Chem. Soc. 136 11610
 Google Scholar
Google Scholar
[10] Stranks S D, Burlakov V M, Leijtens T, Ball J M, Goriely A, Snaith H J 2014 Phys. Rev. Appl. 2 034007
 Google Scholar
Google Scholar
[11] Stoumpos C C, Malliakas C D, Kanatzidis M G 2013 Inorg. Chem. 52 9019
 Google Scholar
Google Scholar
[12] Yantara N, Bhaumik S, Yan F, Sabba D, Dewi H A, Mathews N, Boix P P, Demir H V, Mhaisalkar S 2015 J. Phys. Chem. Lett. 6 4360
 Google Scholar
Google Scholar
[13] Adachi M M, Fan F, Sellan D P, Hoogland S, Voznyy O, Houtepen A J, Parrish K D, Kanjanaboos P, Malen J A, Sargent E H 2015 Nat. Commun. 6 8694
 Google Scholar
Google Scholar
[14] Kojima A, Teshima K, Shirai Y, Miyasaka T 2009 J. Am. Chem. Soc. 131 6050
 Google Scholar
Google Scholar
[15] Kim H S, Lee C R, Im J H, Lee K B, Moehl T, Marchioro A, Moon S J, Humphry-Baker R, Yum J H, Moser J E, Grätzel M, Park N G 2012 Sci. Rep. 2 591
 Google Scholar
Google Scholar
[16] Yang W S, Park B W, Jung E H, Jeon N J, Kim Y C, Lee D U, Shin S S, Seo J, Kim E K, Noh J H, Seok S I 2017 Science 356 1376
 Google Scholar
Google Scholar
[17] Yao K, Li F, He Q Q, Wang X F, Jiang Y H, Huang H T, Jen A K Y 2017 Nano Energy 40 155
 Google Scholar
Google Scholar
[18] Chen R, Hou D, Lu C, Zhang J, Liu P, Tian H, Zeng Z, Xiong Q, Hu Z, Zhu Y, Han L 2018 Sustain. Energ. Fuels 2 1093
 Google Scholar
Google Scholar
[19] Jaramillo-Quintero O A, Sanchez R S, Rincon M, Mora-Sero I 2015 J. Phys. Chem. Lett. 6 1883
 Google Scholar
Google Scholar
[20] Kim Y H, Cho H, Heo J H, Kim T S, Myoung N, Lee C L, Im S H, Lee T W 2015 Adv. Mater. 27 1248
 Google Scholar
Google Scholar
[21] Li J Q, Bade S G R, Shan X, Yu Z B 2015 Adv. Mater. 27 5196
 Google Scholar
Google Scholar
[22] Tang B, Dong H, Sun L, Zheng W, Wang Q, Sun F, Jiang X, Pan A, Zhang L 2017 ACS Nano 11 10681
 Google Scholar
Google Scholar
[23] Yakunin S, Protesescu L, Krieg F, Bodnarchuk M I, Nedelcu G, Humer M, de Luca G, Fiebig M, Heiss W, Kovalenko M V 2015 Nat. Commun. 6 8056
 Google Scholar
Google Scholar
[24] Sutherland B R, Hoogland S, Adachi M M, Wong C T O, Sargent E H 2014 ACS Nano 8 10947
 Google Scholar
Google Scholar
[25] Fu Y, Zhu H, Stoumpos C C, Ding Q, Wang J, Kanatzidis M G, Zhu X, Jin S 2016 ACS Nano 10 7963
 Google Scholar
Google Scholar
[26] Domanski K, Tress W, Moehl T, Saliba M, Nazeeruddin M K, Gratzel M 2015 Adv. Funct. Mater. 25 6936
 Google Scholar
Google Scholar
[27] Sutherland B R, Johnston A K, Ip A H, Xu J, Adinolfi V, Kanjanaboos P, Sargent E H 2015 ACS Photon. 2 1117
 Google Scholar
Google Scholar
[28] Lian Z, Yan Q, Lv Q, Wang Y, Liu L, Zhang L, Pan S, Li Q, Wang L, Sun J L 2015 Sci. Rep. 5 16563
 Google Scholar
Google Scholar
[29] Adinolfi V, Ouellette O, Saidaminov M I, Walters G, Abdelhady A L, Bakr O M, Sargent E H 2016 Adv. Mater. 28 7264
 Google Scholar
Google Scholar
[30] Zhao D, Wang C, Song Z, Yu Y, Chen C, Zhao X, Zhu K, Yan Y 2018 ACS Energy Lett. 3 305
 Google Scholar
Google Scholar
[31] Lin K, Xing J, Quan L N, de Arquer F P G, Gong X, Lu J, Xie L, Zhao W, Zhang D, Yan C, Li W, Liu X, Lu Y, Kirman J, Sargent E H, Xiong Q, Wei Z 2018 Nature 562 245
 Google Scholar
Google Scholar
[32] Chiba T, Hayashi Y, Ebe H, Hoshi K, Sato J, Sato S, Pu Y J, Ohisa S, Kido J 2018 Nat. Photon. 12 681
 Google Scholar
Google Scholar
[33] Cao Y, Wang N, Tian H, Guo J, Wei Y, Chen H, Miao Y, Zou W, Pan K, He Y, Cao H, Ke Y, Xu M, Wang Y, Yang M, Du K, Fu Z, Kong D, Dai D, Jin Y, Li G, Li H, Peng Q, Wang J, Huang W 2018 Nature 562 249
 Google Scholar
Google Scholar
[34] Berhe T A, Su W N, Chen C H, Pan C J, Cheng J H, Chen H M, Tsai M C, Chen L Y, Dubale A A, Hwang B J 2016 Energy Environ. Sci. 9 323
 Google Scholar
Google Scholar
[35] Abdi-Jalebi M, Andaji-Garmaroudi Z, Cacovich S, Stavrakas C, Philippe B, Richter J M, Alsari M, Booker E P, Hutter E M, Pearson A J, Lilliu S, Savenije T J, Rensmo H, Divitini G, Ducati C, Friend R H, Stranks S D 2018 Nature 555 497
 Google Scholar
Google Scholar
[36] Frost J M, Butler K T, Brivio F, Hendon C H, van Schilfgaarde M, Walsh A 2014 Nano Lett. 14 2584
 Google Scholar
Google Scholar
[37] Yang J, Siempelkamp B D, Liu D, Kelly T L 2015 ACS Nano 9 1955
 Google Scholar
Google Scholar
[38] Christians J A, Miranda Herrera P A, Kamat P V 2015 J. Am. Chem. Soc. 137 1530
 Google Scholar
Google Scholar
[39] Li Y, Xu X, Wang C, Wang C, Xie F, Yang J, Gao Y 2015 J. Phys. Chem. C 119 23996
 Google Scholar
Google Scholar
[40] Song Z N, Abate A, Watthage S C, Liyanage G K, Phillips A B, Steiner U, Graetzel M, Heben M J 2016 Adv. Energy Mater. 6 1600846
 Google Scholar
Google Scholar
[41] Aristidou N, Sanchez-Molina I, Chotchuangchutchaval T, Brown M, Martinez L, Rath T, Haque S A 2015 Angew. Chem. Int. Ed. 54 8208
 Google Scholar
Google Scholar
[42] Bryant D, Aristidou N, Pont S, Sanchez-Molina I, Chotchunangatchaval T, Wheeler S, Durrant J R, Haque S A 2016 Energy Environ. Sci. 9 1655
 Google Scholar
Google Scholar
[43] Alberti A, Deretzis I, Pellegrino G, Bongiorno C, Smecca E, Mannino G, Giannazzo F, Condorelli G G, Sakai N, Miyasaka T, Spinella C, La Magna A 2015 Chem. Phys. Chem. 16 3064
 Google Scholar
Google Scholar
[44] Merdasa A, Bag M, Tian Y, Källman E, Dobrovolsky A, Scheblykin I G 2016 J. Phys. Chem. C 120 10711
 Google Scholar
Google Scholar
[45] Slotcavage D J, Karunadasa H I, McGehee M D 2016 ACS Energy Lett. 1 1199
 Google Scholar
Google Scholar
[46] Yang X, Yan X L, Wang W, Zhu X X, Li H, Ma W L, Sheng C X 2016 Org. Electron. 34 79
 Google Scholar
Google Scholar
[47] Hoke E T, Slotcavage D J, Dohner E R, Bowring A R, Karunadasa H I, McGehee M D 2015 Chem. Sci. 6 613
 Google Scholar
Google Scholar
[48] Yoon S J, Kuno M, Kamat P V 2017 ACS Energy Lett. 2 1507
 Google Scholar
Google Scholar
[49] Chen S, Wen X M, Huang S J, Huang F Z, Cheng Y B, Green M, Ho-Baillie A 2017 Solar RRL 1 Unsp 1600001
 Google Scholar
Google Scholar
[50] Mosconi E, Meggiolaro D, Snaith H J, Stranks S D, de Angelis F 2016 Energy Environ. Sci. 9 3180
 Google Scholar
Google Scholar
[51] Divitini G, Cacovich S, Matteocci F, Cinà L, Di Carlo A, Ducati C 2016 Nature Energy 1 15012
 Google Scholar
Google Scholar
[52] Kim N K, Min Y H, Noh S, Cho E, Jeong G, Joo M, Ahn S W, Lee J S, Kim S, Ihm K, Ahn H, Kang Y, Lee H S, Kim D 2017 Sci. Rep. 7 4645
 Google Scholar
Google Scholar
[53] Conings B, Drijkoningen J, Gauquelin N, Babayigit A, D'Haen J, D'Olieslaeger L, Ethirajan A, Verbeeck J, Manca J, Mosconi E, de Angelis F, Boyen H G 2015 Adv. Energy Mater. 5 1500477
 Google Scholar
Google Scholar
[54] Chen W, Wu Y, Yue Y, Liu J, Zhang W, Yang X, Chen H, Bi E, Ashraful I, Grätzel M, Han L 2015 Science 350 944
 Google Scholar
Google Scholar
[55] Kaltenbrunner M, Adam G, Głowacki E D, Drack M, Schwödiauer R, Leonat L, Apaydin D H, Groiss H, Scharber M C, White M S, Sariciftci N S, Bauer S 2015 Nat. Mater. 14 1032
 Google Scholar
Google Scholar
[56] You J, Meng L, Song T B, Guo T F, Yang Y, Chang W H, Hong Z, Chen H, Zhou H, Chen Q, Liu Y, De Marco N, Yang Y 2016 Nat. Nanotechnol. 11 75
[57] deQuilettes D W, Zhang W, Burlakov V M, Graham D J, Leijtens T, Osherov A, Bulović V, Snaith H J, Ginger D S, Stranks S D 2016 Nat. Commun. 7 11683
 Google Scholar
Google Scholar
[58] Gottesman R, Gouda L, Kalanoor B S, Haltzi E, Tirosh S, Rosh-Hodesh E, Tischler Y, Zaban A, Quarti C, Mosconi E, de Angelis F 2015 J. Phys. Chem. Lett. 6 2332
 Google Scholar
Google Scholar
[59] Domanski K, Roose B, Matsui T, Saliba M, Turren-Cruz S H, Correa-Baena J P, Roldan-Carmona C, Richardson G, Foster J M, de Angelis F, Ball J M, Petrozza A, Mine N, Nazeeruddin M K, Tress W, Gratzel M, Steiner U, Hagfeldt A, Abate A 2017 Energy Environ. Sci. 10 604
 Google Scholar
Google Scholar
[60] Zhou Y, You L, Wang S, Ku Z, Fan H, Schmidt D, Rusydi A, Chang L, Wang L, Ren P, Chen L, Yuan G, Chen L, Wang J 2016 Nat. Commun. 7 11193
 Google Scholar
Google Scholar
[61] Brivio F, Caetano C, Walsh A 2016 J. Phys. Chem. Lett. 7 1083
 Google Scholar
Google Scholar
[62] Zhao C, Chen B B, Qiao X F, Luan L, Lu K, Hu B 2015 Adv. Energy Mater. 5 1500279
 Google Scholar
Google Scholar
[63] Huang F, Jiang L, Pascoe A R, Yan Y, Bach U, Spiccia L, Cheng Y B 2016 Nano Energy 27 509
 Google Scholar
Google Scholar
[64] Deschler F, Price M, Pathak S, Klintberg L E, Jarausch D D, Higler R, Hüttner S, Leijtens T, Stranks S D, Snaith H J, Atatüre M, Phillips R T, Friend R H 2014 J. Phys. Chem. Lett. 5 1421
 Google Scholar
Google Scholar
[65] Tian Y, Merdasa A, Unger E, Abdellah M, Zheng K, McKibbin S, Mikkelsen A, Pullerits T, Yartsev A, Sundström V, Scheblykin I G 2015 J. Phys. Chem. Lett. 6 4171
 Google Scholar
Google Scholar
[66] Tian Y X, Peter M, Unger E, Abdellah M, Zheng K, Pullerits T, Yartsev A, Sundstrom V, Scheblykin I G 2015 Phys. Chem. Chem. Phys. 17 24978
 Google Scholar
Google Scholar
[67] Brenes R, Guo D, Osherov A, Noel N K, Eames C, Hutter E M, Pathak S K, Niroui F, Friend R H, Islam M S, Snaith H J, Bulović V, Savenije T J, Stranks S D 2017 Joule 1 155
 Google Scholar
Google Scholar
[68] Buin A, Pietsch P, Xu J, Voznyy O, Ip A H, Comin R, Sargent E H 2014 Nano Lett. 14 6281
 Google Scholar
Google Scholar
[69] Yin W J, Shi T, Yan Y 2014 Appl. Phys. Lett. 104 063903
 Google Scholar
Google Scholar
[70] Buin A, Comin R, Xu J, Ip A H, Sargent E H 2015 Chem. Mater. 27 4405
 Google Scholar
Google Scholar
[71] Mosconi E, de Angelis F 2016 ACS Energy Lett. 1 182
 Google Scholar
Google Scholar
[72] Meloni S, Moehl T, Tress W, Franckevičius M, Saliba M, Lee Y H, Gao P, Nazeeruddin M K, Zakeeruddin S M, Rothlisberger U, Graetzel M 2016 Nat. Commun. 7 10334
 Google Scholar
Google Scholar
[73] Haruyama J, Sodeyama K, Han L, Tateyama Y 2015 J. Am. Chem. Soc. 137 10048
 Google Scholar
Google Scholar
[74] Huang F, Dkhissi Y, Huang W, Xiao M, Benesperi I, Rubanov S, Zhu Y, Lin X, Jiang L, Zhou Y, Gray-Weale A, Etheridge J, McNeill C R, Caruso R A, Bach U, Spiccia L, Cheng Y B 2014 Nano Energy 10 10
 Google Scholar
Google Scholar
[75] Wen X M, Feng Y, Huang S J, Huang F Z, Cheng Y B, Green M, Ho-Baillie A 2016 J. Mater. Chem. C 4 793
 Google Scholar
Google Scholar
[76] Nie W, Blancon J C, Neukirch A J, Appavoo K, Tsai H, Chhowalla M, Alam M A, Sfeir M Y, Katan C, Even J, Tretiak S, Crochet J J, Gupta G, Mohite A D 2016 Nat. Commun. 7 11574
 Google Scholar
Google Scholar
[77] Gottesman R, Haltzi E, Gouda L, Tirosh S, Bouhadana Y, Zaban A, Mosconi E, de Angelis F 2014 J. Phys. Chem. Lett. 5 2662
 Google Scholar
Google Scholar
[78] Azpiroz J M, Mosconi E, Bisquert J, de Angelis F 2015 Energy Environ. Sci. 8 2118
 Google Scholar
Google Scholar
[79] Eames C, Frost J M, Barnes P R F, O’Regan B C, Walsh A, Islam M S 2015 Nat. Commun. 6 7497
 Google Scholar
Google Scholar
[80] Walsh A, Scanlon D O, Chen S Y, Gong X G, Wei S H 2015 Angew. Chem. Int. Ed. 54 1791
 Google Scholar
Google Scholar
[81] Zhang H M, Liang C J, Zhao Y, Sun M J, Liu H, Liang J J, Li D, Zhang F J, He Z Q 2015 Phys. Chem. Chem. Phys. 17 9613
 Google Scholar
Google Scholar
[82] Zu F S, Amsalem P, Salzmann I, Wang R B, Ralaiarisoa M, Kowarik S, Duhm S, Koch N 2017 Adv. Opt. Mater. 5 Unsp 1700139
 Google Scholar
Google Scholar
[83] Noh J H, Im S H, Heo J H, Mandal T N, Seok S I 2013 Nano Lett. 13 1764
 Google Scholar
Google Scholar
[84] Aharon S, Cohen B E, Etgar L 2014 J. Phys. Chem. C 118 17160
 Google Scholar
Google Scholar
[85] Gil-Escrig L, Miquel-Sempere A, Sessolo M, Bolink H J 2015 J. Phys. Chem. Lett. 6 3743
 Google Scholar
Google Scholar
[86] Bischak C G, Hetherington C L, Wu H, Aloni S, Ogletree D F, Limmer D T, Ginsberg N S 2017 Nano Lett. 17 1028
 Google Scholar
Google Scholar
[87] Barker A J, Sadhanala A, deschler F, Gandini M, Senanayak S P, Pearce P M, Mosconi E, Pearson A J, Wu Y, Srimath Kandada A R, Leijtens T, De Angelis F, Dutton S E, Petrozza A, Friend R H 2017 ACS Energy Lett. 2 1416
 Google Scholar
Google Scholar
[88] Draguta S, Sharia O, Yoon S J, Brennan M C, Morozov Y V, Manser J S, Kamat P V, Schneider W F, Kuno M 2017 Nat. Commun. 8 200
 Google Scholar
Google Scholar
[89] Beal R E, Slotcavage D J, Leijtens T, Bowring A R, Belisle R A, Nguyen W H, Burkhard G F, Hoke E T, McGehee M D 2016 J. Phys. Chem. Lett. 7 746
 Google Scholar
Google Scholar
[90] McMeekin D P, Sadoughi G, Rehman W, Eperon G E, Saliba M, Hörantner M T, Haghighirad A, Sakai N, Korte L, Rech B, Johnston M B, Herz L M, Snaith H J 2016 Science 351 151
 Google Scholar
Google Scholar
[91] Jaffe A, Lin Y, Beavers C M, Voss J, Mao W L, Karunadasa H I 2016 ACS Cent. Sci. 2 201
 Google Scholar
Google Scholar
[92] Hu M, Bi C, Yuan Y B, Bai Y, Huang J S 2016 Adv. Sci. 3 1500301
 Google Scholar
Google Scholar
[93] Wei T C, Wang H P, Li T Y, Lin C H, Hsieh Y H, Chu Y H, He J H 2017 Adv. Mater. 29 Unsp 1701789
 Google Scholar
Google Scholar
[94] Kirschner M S, Diroll B T, Guo P, Harvey S M, Helweh W, Flanders N C, Brumberg A, Watkins N E, Leonard A A, Evans A M, Wasielewski M R, Dichtel W R, Zhang X, Chen L X, Schaller R D 2019 Nat. Commun. 10 504
 Google Scholar
Google Scholar
[95] Cottingham P, Brutchey R L 2018 Chem. Mater. 30 6711
 Google Scholar
Google Scholar
[96] Xue J, Yang D, Cai B, Xu X, Wang J, Ma H, Yu X, Yuan G, Zou Y, Song J, Zeng H 2019 Adv. Funct. Mater. 29 1807922
[97] Yettapu G R, Talukdar D, Sarkar S, Swarnkar A, Nag A, Ghosh P, Mandal P 2016 Nano Lett. 16 4838
 Google Scholar
Google Scholar
[98] He Y, Matei L, Jung H J, McCall K M, Chen M, Stoumpos C C, Liu Z, Peters J A, Chung D Y, Wessels B W, Wasielewski M R, Dravid V P, Burger A, Kanatzidis M G 2018 Nat. Commun. 9 1609
 Google Scholar
Google Scholar
[99] Meng Y, Ahmadi M, Wu X, Xu T, Xu L, Xiong Z, Chen P 2019 Org. Electron. 64 47
 Google Scholar
Google Scholar
[100] Zhang Y, Sun H, Zhang S, Li S, Wang X, Zhang X, Liu T, Guo Z 2019 Opt. Mater. 89 563
 Google Scholar
Google Scholar
[101] Huang M Y, Veeramuthu L, Kuo C C, Liao Y C, Jiang D H, Liang F C, Yan Z L, Borsali R, Chueh C C 2019 Org. Electron. 67 294
 Google Scholar
Google Scholar
[102] Tan Z, Wu Y, Hong H, Yin J, Zhang J, Lin L, Wang M, Sun X, Sun L, Huang Y, Liu K, Liu Z, Peng H 2016 J. Am. Chem. Soc. 138 16612
 Google Scholar
Google Scholar
[103] Chen S, Shi G 2017 Adv. Mater. 29 1605448
 Google Scholar
Google Scholar
-
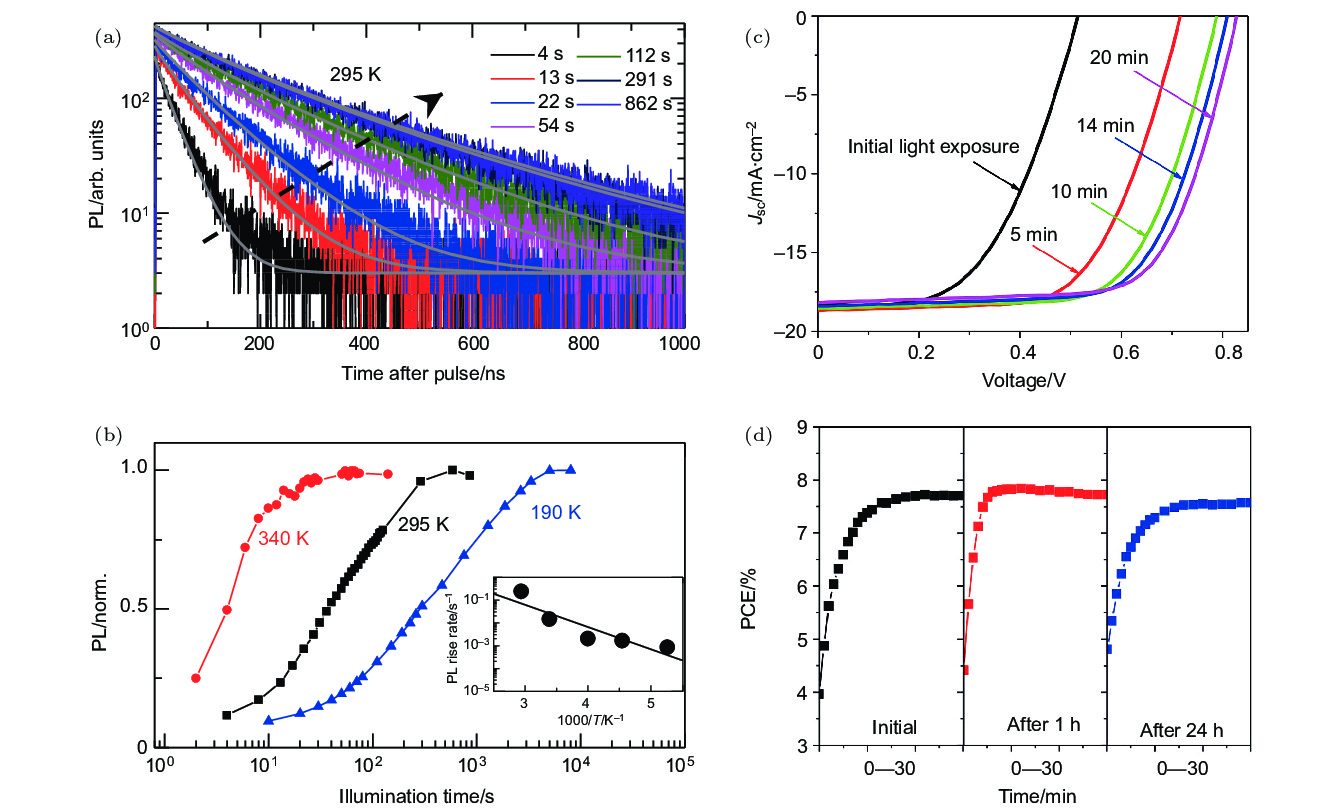
图 2 铅卤钙钛矿及其太阳能电池在持续光照下发生光修复现象时的性能变化 (a) CH3NH3PbI3薄膜在持续光照下的时间分辨PL衰减随光照时间的变化[57]; (b) CH3NH3PbI3薄膜在持续光照下以及不同温度下的PL强度随光照时间的变化[57]; (c) 铅卤钙钛矿太阳能电池ITO/PEDOT:PSS/CH3NH3PbI3–xClx/PCBM/Al在持续光照下的J-V曲线变化[62]; (d) 铅卤钙钛矿太阳能电池ITO/PEDOT:PSS/CH3NH3PbI3–xClx/PCBM/Al在持续光照下的光电转化效率的变化曲线[62]
Fig. 2. Performance changes of lead halide perovskite and its solar cells under continuous illunination: (a) A series of time-resolved PL decays from CH3NH3PbI3 films measured over time under continuous illumination[57]; (b) the normalized PL intensity of CH3NH3PbI3 films measured over time under continuous illumination at different temperatures[57]; (c) the J-V characteristics of lead halide perovskite solar cell with the architecture of ITO/PEDOT:PSS/CH3NH3PbI3–xClx/PCBM/Al under continuous illumination[62]; (d) the reversibility of power conversion efficiency of lead halide perovskite solar cell with the architecture of ITO/PEDOT:PSS/CH3NH3PbI3–xClx/PCBM/Al under continuous illumination[62].
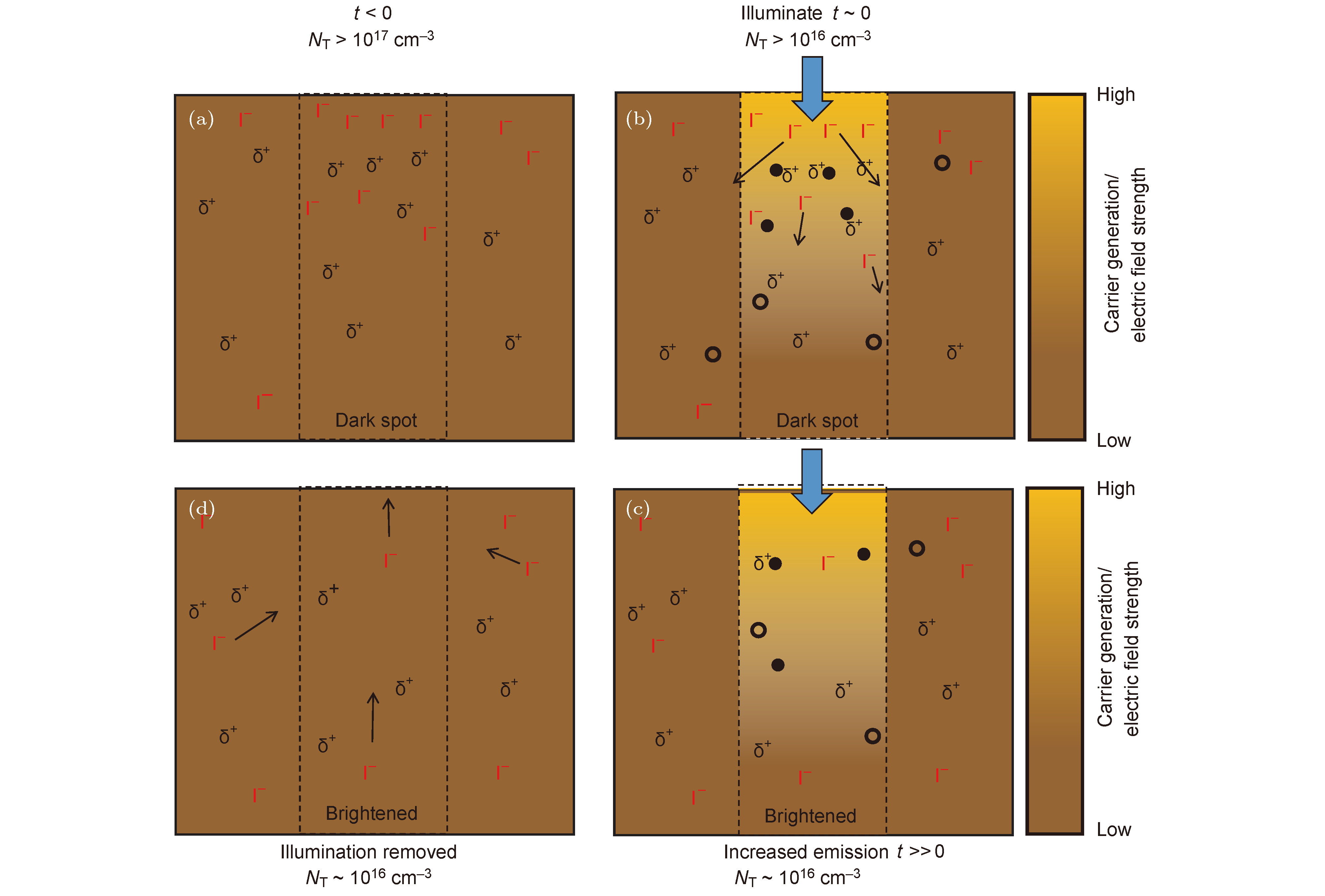
图 3 CH3NH3PbI3薄膜发生光修复现象的机理[57] (a) 在发光较弱的晶粒中分布有高浓度的缺陷态以及过量的碘离子; (b) CH3NH3PbI3薄膜经过光照后, 电子迅速填充缺陷, 产生电场, 造成碘离子从照射区域开始迁移, 填充空位; (c)照射区域中的缺陷态密度和碘离子浓度降低, 薄膜最终达到稳定的状态; (d) 当移除光照后, 碘离子存在浓度梯度, 驱使其扩散回弱发光区域, 直至达到新的碘离子浓度平衡
Fig. 3. Mechanism of photo curing in CH3NH3PbI3 film[57]: (a) Iodide in a "dark spot" is initially high with a corresponding excess of trap density; (b) electrons quickly fill traps under illumination, inducing an electric field that results in iodide to migrate away from the illuminated region and fill vacancies; (c) the fill will reaches a stabilized state, with the reduction of the density of the traps and the concentration of the iodide in illuminated region; (d) when the illumination is removed, there may be a concentration gradients driving some iodides in to the dark spot and the film will reaches a new equilibrium with a net redistributed iodide profile.
图 4 (a) 传统旋涂法制备的CH3NH3PbI3薄膜在激发密度0.2和0.65 W/cm2下的光致发光强度随时间的变化; (b) 气体辅助溶液工程技术制备的CH3NH3PbI3薄膜在激发密度0.90, 1.55和2.00 W/cm2下的光致发光强度随时间的变化; (c) 传统旋涂法以及气体辅助溶液工程技术制备的CH3NH3PbI3薄膜的光致发光谱和吸收光谱[49]
Fig. 4. (a) PL intensities over time of CH3NH3PbI3 film fabricated by conventional spin coating under laser excitations at 0.2 and 0.65 W/cm2; (b) PL intensities over time of CH3NH3PbI3 film fabricated by gas-assisted solution process technique under laser excitations at 0.9, 1.55 and 2.00 W/cm2; (c) absorption and PL spectra of samples fabricated conventional spin coating and gas-assisted solution process technique[49].
图 5 (a) CH3NH3PbI3薄膜在黑暗-光照-黑暗循环中的光致发光强度随时间的变化; (b) CH3NH3PbI3薄膜的光致发光谱随黑暗或光照下放置时间的变化; (c) CH3NH3PbI3薄膜的实验中和理论上的拉曼光谱以及CH3NH3PbI3薄膜在黑暗环境和光照20, 40和60 min后的拉曼光谱[58]
Fig. 5. (a) PL measurements performed throughout a dark-light-dark cycle, showing the effect of white-light illumination on the PL intensity of the CH3NH3PbI3 film; (b) a graph showing six PL measurements points from the dark-light-dark cycles at different times; (c) a graph showing Experimental (taken in the dark) and theoretical Raman spectra of a CH3NH3PbI3 film as well as the Raman spectra of the same film taken in the dark and following illumination for 20, 40 and 60 min[58]
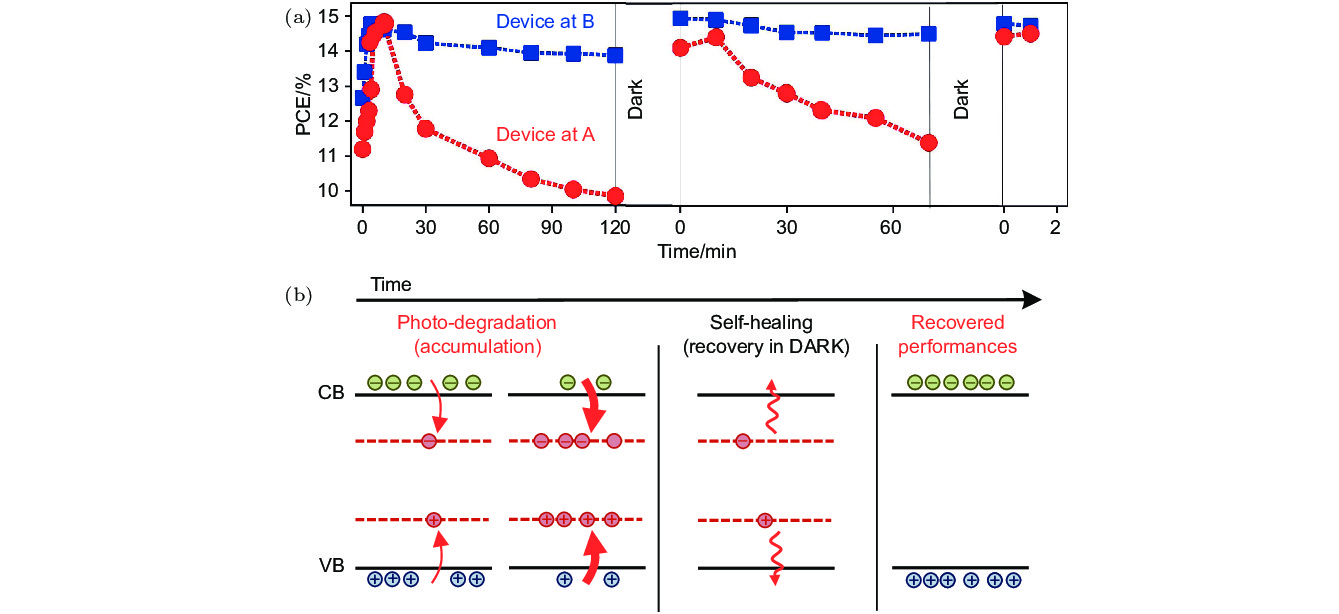
图 6 (a) 铅卤钙钛矿太阳能电池ITO/PEDOT:PSS/MAPbI3/PCBM/Al在持续光照下的光电转化效率的变化趋势, 其中工作条件A对应于J = 0, V = Voc; 工作条件B对应于J = Jsc, V = 0; (b) 铅卤钙钛矿材料发生可逆光解现象的机理[76]
Fig. 6. (a) Time evolution of power conversion efficiency of lead halide perovskite solar cell with the architecture of ITO/PEDOT:PSS/MAPbI3/PCBM/Al under constant 1-Sun illumination. The device is stressed at point A (red circles, J = 0, V = Voc) or point B (blue squares, J = Jsc, V = 0); (b) schematics of the photocurrent degradation and self-healing mechanism[76].
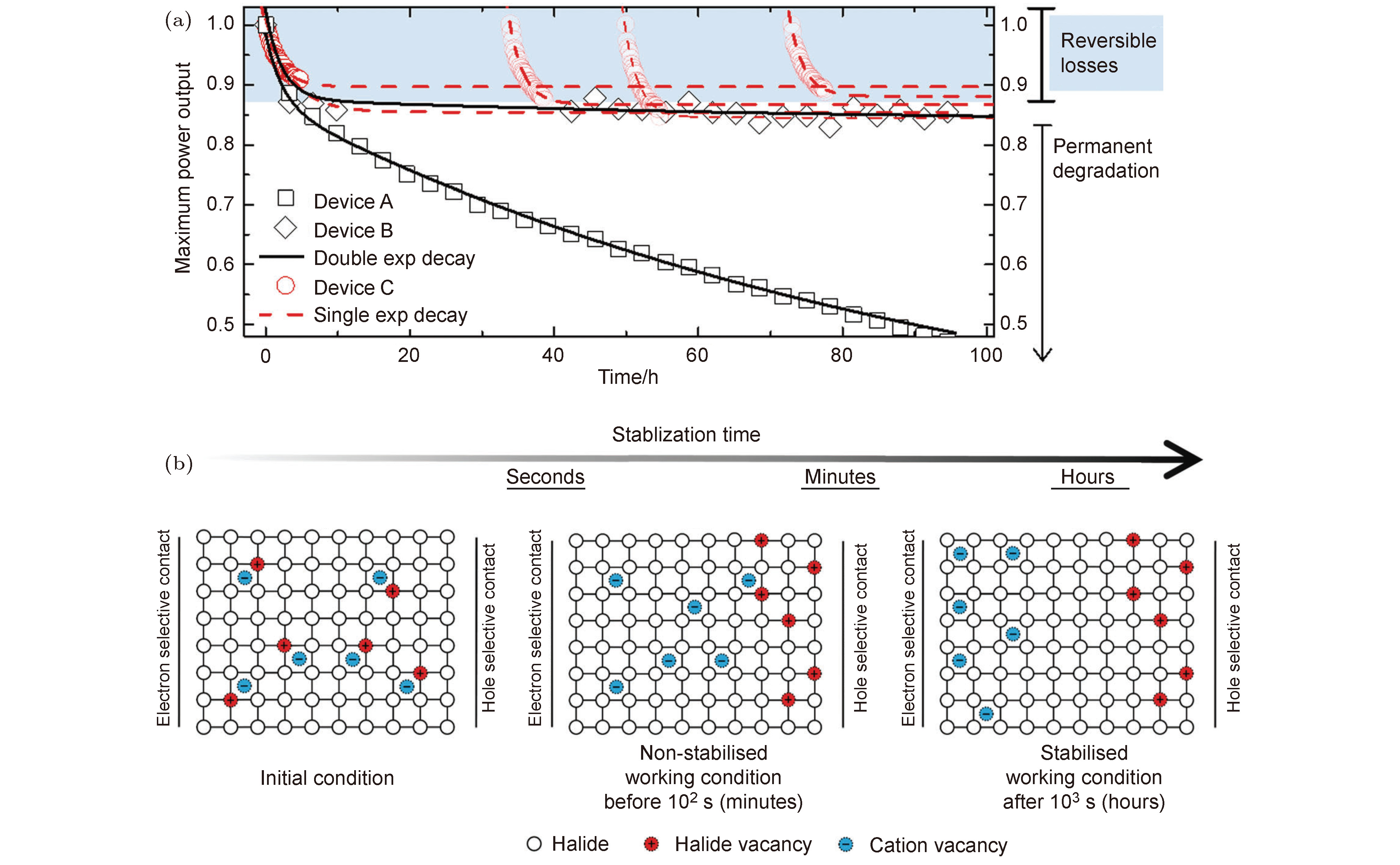
图 7 (a) 铅卤钙钛矿太阳能电池FTO/TiO2/mp-TiO2/(FAPbI3)x(MAPbBr3)y/spiro-OMeTAD/Au在滤过紫外光后的LED灯光持续照射下的最大功率输出的变化趋势; (b) 钙钛矿层中的离子分布的变化趋势[59]
Fig. 7. (a) Maximum power output tracking for 3 identically prepared perovskite solar cells (device A, B and C), with the architecture of FTO/TiO2/mp-TiO2/(FAPbI3)x(MAPbBr3)y/spiro-OMeTAD/Au, measured under UV-filtered 1-Sun LED illumination. Experimental data were fitted to an exponential decay (single or double). (b) Schematics of the evolution of ion distribution within the perovskite layer[59].
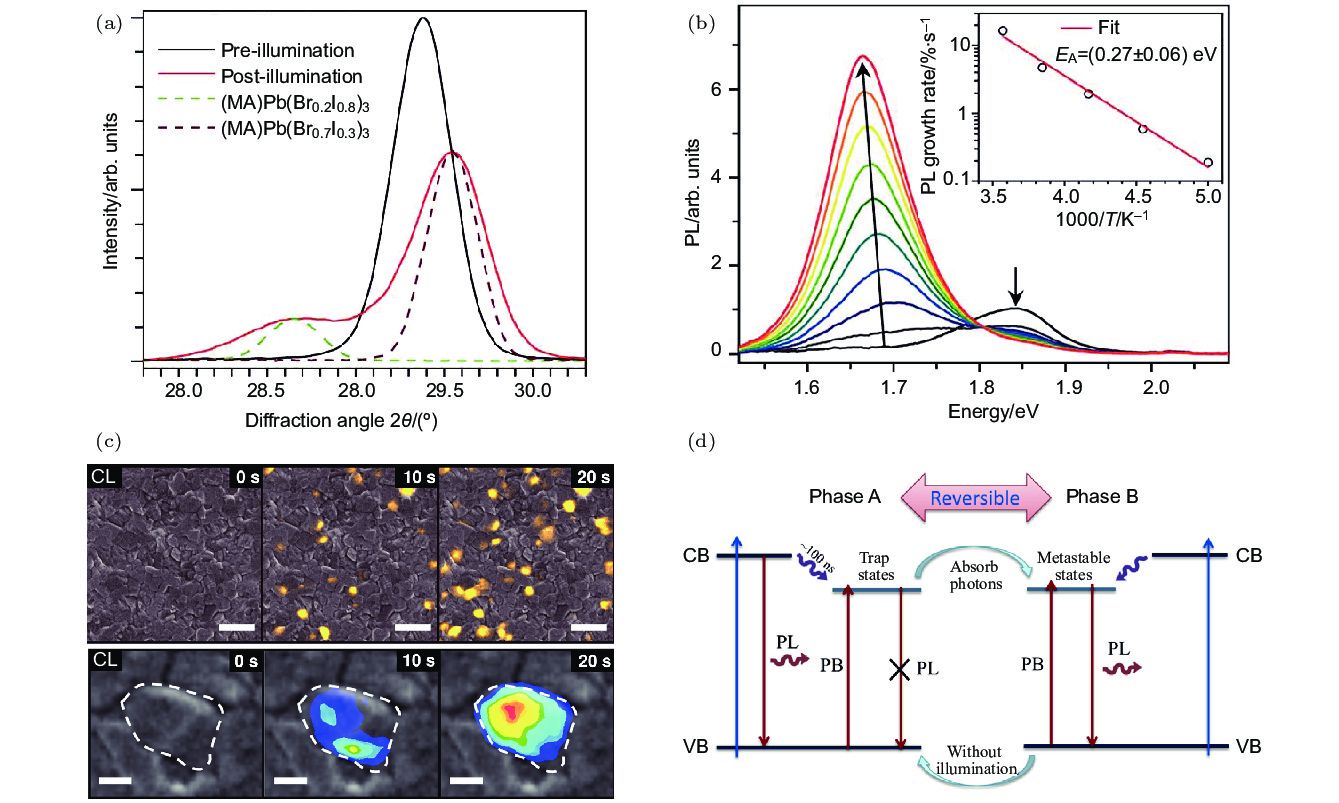
图 8 (a) CH3NH3Pb(Br0.6I0.4)3在激发密度50 mW/cm2下光照5 min前(黑线)和后(红线)的XRD图像, 其中虚线分别为CH3NH3Pb(Br0.2I0.8)3和CH3NH3Pb(Br0.7I0.3)3的XRD图像[47]; (b) CH3NH3Pb(Br0.4I0.6)3在激发密度为15 mW/cm2的457 nm激光持续照射下的光致发光谱的变化趋势[47]; (c) CH3NH3Pb(Br0.9I0.1)3在激发密度为50 mW/cm2的405 nm激光持续照射下的阴极发光图像随时间的变化[86]; (d) CH3NH3Pb(Br1–xIx)3在光照下发生光致相分解现象的机理[46]
Fig. 8. (a) The XRD peak of CH3NH3Pb(Br0.6I0.4)3 film before (black) and after (red) white-light soaking for 5 minutes 50 mW/cm2. XRD patterns of CH3NH3Pb(Br0.2I0.8)3 and CH3NH3Pb(Br0.7I0.3)3 films (dashed brown) are included for comparison[47]; (b) photoluminescence spectra of CH3NH3Pb(Br0.4I0.6)3 film over 45 s in 5 s increments under 457 nm, 15 mW/cm2 at 300 K[47]; (c) a series of cathodo luminescence image after light soaking under a 405 nm LED at 50 mW/cm2[86]; (d) the schematic of two-step mechanism for light-induced phase separation[46].
图 9 铅卤钙钛矿在持续光照下发生的主要四种不稳定现象 (a) 碘间隙离子/碘空位缺陷对的消除引起光修复现象; (b) 碘空位与阳离子空位的迁移引起光解现象; (c) 亥姆霍兹自由能最小化促使铅卤钙钛矿内部的卤素离子从无序排列向有序排列, 引起光致相分离现象; (d) 光生载流子迁移率差异产生电位差, 造成PBI64–六面体扭力方向的改变, 引起光致相变现象
Fig. 9. Main four kinds of instability of lead halide perovskites under continuous illumination: (a) Photo-curing induced by the elimination of iodine interstitial ions/iodine vacancies; (b) photo dissociation induced by the migration of iodine vacancies and cation vacancies; (c) phase separation induced by the minimization of Helmholtz free energy; (d) phase transition induced by the difference in the mobility of electrons and holes.
-
[1] Sutherland B R, Sargent E H 2016 Nat. Photon. 10 295
 Google Scholar
Google Scholar
[2] Heo J H, Im S H, Noh J H, Mandal T N, Lim C S, Chang J A, Lee Y H, Kim H J, Sarkar A, Nazeeruddin M K, Grätzel M, Seok S I 2013 Nat. Photon. 7 486
 Google Scholar
Google Scholar
[3] Giorgi G, Fujisawa J I, Segawa H, Yamashita K 2013 J. Phys. Chem. Lett. 4 4213
 Google Scholar
Google Scholar
[4] Bisquert J 2013 J. Phys. Chem. Lett. 4 1051
 Google Scholar
Google Scholar
[5] Etgar L, Gao P, Xue Z, Peng Q, Chandiran A K, Liu B, Nazeeruddin M K, Grätzel M 2012 J. Am. Chem. Soc. 134 17396
 Google Scholar
Google Scholar
[6] Abu Laban W, Etgar L 2013 Energy Environ. Sci. 6 3249
 Google Scholar
Google Scholar
[7] Xing G, Mathews N, Sun S, Lim S S, Lam Y M, Grätzel M, Mhaisalkar S, Sum T C 2013 Science 342 344
 Google Scholar
Google Scholar
[8] Stranks S D, Eperon G E, Grancini G, Menelaou C, Alcocer M J P, Leijtens T, Herz L M, Petrozza A, Snaith H J 2013 Science 342 341
 Google Scholar
Google Scholar
[9] Yamada Y, Nakamura T, Endo M, Wakamiya A, Kanemitsu Y 2014 J. Am. Chem. Soc. 136 11610
 Google Scholar
Google Scholar
[10] Stranks S D, Burlakov V M, Leijtens T, Ball J M, Goriely A, Snaith H J 2014 Phys. Rev. Appl. 2 034007
 Google Scholar
Google Scholar
[11] Stoumpos C C, Malliakas C D, Kanatzidis M G 2013 Inorg. Chem. 52 9019
 Google Scholar
Google Scholar
[12] Yantara N, Bhaumik S, Yan F, Sabba D, Dewi H A, Mathews N, Boix P P, Demir H V, Mhaisalkar S 2015 J. Phys. Chem. Lett. 6 4360
 Google Scholar
Google Scholar
[13] Adachi M M, Fan F, Sellan D P, Hoogland S, Voznyy O, Houtepen A J, Parrish K D, Kanjanaboos P, Malen J A, Sargent E H 2015 Nat. Commun. 6 8694
 Google Scholar
Google Scholar
[14] Kojima A, Teshima K, Shirai Y, Miyasaka T 2009 J. Am. Chem. Soc. 131 6050
 Google Scholar
Google Scholar
[15] Kim H S, Lee C R, Im J H, Lee K B, Moehl T, Marchioro A, Moon S J, Humphry-Baker R, Yum J H, Moser J E, Grätzel M, Park N G 2012 Sci. Rep. 2 591
 Google Scholar
Google Scholar
[16] Yang W S, Park B W, Jung E H, Jeon N J, Kim Y C, Lee D U, Shin S S, Seo J, Kim E K, Noh J H, Seok S I 2017 Science 356 1376
 Google Scholar
Google Scholar
[17] Yao K, Li F, He Q Q, Wang X F, Jiang Y H, Huang H T, Jen A K Y 2017 Nano Energy 40 155
 Google Scholar
Google Scholar
[18] Chen R, Hou D, Lu C, Zhang J, Liu P, Tian H, Zeng Z, Xiong Q, Hu Z, Zhu Y, Han L 2018 Sustain. Energ. Fuels 2 1093
 Google Scholar
Google Scholar
[19] Jaramillo-Quintero O A, Sanchez R S, Rincon M, Mora-Sero I 2015 J. Phys. Chem. Lett. 6 1883
 Google Scholar
Google Scholar
[20] Kim Y H, Cho H, Heo J H, Kim T S, Myoung N, Lee C L, Im S H, Lee T W 2015 Adv. Mater. 27 1248
 Google Scholar
Google Scholar
[21] Li J Q, Bade S G R, Shan X, Yu Z B 2015 Adv. Mater. 27 5196
 Google Scholar
Google Scholar
[22] Tang B, Dong H, Sun L, Zheng W, Wang Q, Sun F, Jiang X, Pan A, Zhang L 2017 ACS Nano 11 10681
 Google Scholar
Google Scholar
[23] Yakunin S, Protesescu L, Krieg F, Bodnarchuk M I, Nedelcu G, Humer M, de Luca G, Fiebig M, Heiss W, Kovalenko M V 2015 Nat. Commun. 6 8056
 Google Scholar
Google Scholar
[24] Sutherland B R, Hoogland S, Adachi M M, Wong C T O, Sargent E H 2014 ACS Nano 8 10947
 Google Scholar
Google Scholar
[25] Fu Y, Zhu H, Stoumpos C C, Ding Q, Wang J, Kanatzidis M G, Zhu X, Jin S 2016 ACS Nano 10 7963
 Google Scholar
Google Scholar
[26] Domanski K, Tress W, Moehl T, Saliba M, Nazeeruddin M K, Gratzel M 2015 Adv. Funct. Mater. 25 6936
 Google Scholar
Google Scholar
[27] Sutherland B R, Johnston A K, Ip A H, Xu J, Adinolfi V, Kanjanaboos P, Sargent E H 2015 ACS Photon. 2 1117
 Google Scholar
Google Scholar
[28] Lian Z, Yan Q, Lv Q, Wang Y, Liu L, Zhang L, Pan S, Li Q, Wang L, Sun J L 2015 Sci. Rep. 5 16563
 Google Scholar
Google Scholar
[29] Adinolfi V, Ouellette O, Saidaminov M I, Walters G, Abdelhady A L, Bakr O M, Sargent E H 2016 Adv. Mater. 28 7264
 Google Scholar
Google Scholar
[30] Zhao D, Wang C, Song Z, Yu Y, Chen C, Zhao X, Zhu K, Yan Y 2018 ACS Energy Lett. 3 305
 Google Scholar
Google Scholar
[31] Lin K, Xing J, Quan L N, de Arquer F P G, Gong X, Lu J, Xie L, Zhao W, Zhang D, Yan C, Li W, Liu X, Lu Y, Kirman J, Sargent E H, Xiong Q, Wei Z 2018 Nature 562 245
 Google Scholar
Google Scholar
[32] Chiba T, Hayashi Y, Ebe H, Hoshi K, Sato J, Sato S, Pu Y J, Ohisa S, Kido J 2018 Nat. Photon. 12 681
 Google Scholar
Google Scholar
[33] Cao Y, Wang N, Tian H, Guo J, Wei Y, Chen H, Miao Y, Zou W, Pan K, He Y, Cao H, Ke Y, Xu M, Wang Y, Yang M, Du K, Fu Z, Kong D, Dai D, Jin Y, Li G, Li H, Peng Q, Wang J, Huang W 2018 Nature 562 249
 Google Scholar
Google Scholar
[34] Berhe T A, Su W N, Chen C H, Pan C J, Cheng J H, Chen H M, Tsai M C, Chen L Y, Dubale A A, Hwang B J 2016 Energy Environ. Sci. 9 323
 Google Scholar
Google Scholar
[35] Abdi-Jalebi M, Andaji-Garmaroudi Z, Cacovich S, Stavrakas C, Philippe B, Richter J M, Alsari M, Booker E P, Hutter E M, Pearson A J, Lilliu S, Savenije T J, Rensmo H, Divitini G, Ducati C, Friend R H, Stranks S D 2018 Nature 555 497
 Google Scholar
Google Scholar
[36] Frost J M, Butler K T, Brivio F, Hendon C H, van Schilfgaarde M, Walsh A 2014 Nano Lett. 14 2584
 Google Scholar
Google Scholar
[37] Yang J, Siempelkamp B D, Liu D, Kelly T L 2015 ACS Nano 9 1955
 Google Scholar
Google Scholar
[38] Christians J A, Miranda Herrera P A, Kamat P V 2015 J. Am. Chem. Soc. 137 1530
 Google Scholar
Google Scholar
[39] Li Y, Xu X, Wang C, Wang C, Xie F, Yang J, Gao Y 2015 J. Phys. Chem. C 119 23996
 Google Scholar
Google Scholar
[40] Song Z N, Abate A, Watthage S C, Liyanage G K, Phillips A B, Steiner U, Graetzel M, Heben M J 2016 Adv. Energy Mater. 6 1600846
 Google Scholar
Google Scholar
[41] Aristidou N, Sanchez-Molina I, Chotchuangchutchaval T, Brown M, Martinez L, Rath T, Haque S A 2015 Angew. Chem. Int. Ed. 54 8208
 Google Scholar
Google Scholar
[42] Bryant D, Aristidou N, Pont S, Sanchez-Molina I, Chotchunangatchaval T, Wheeler S, Durrant J R, Haque S A 2016 Energy Environ. Sci. 9 1655
 Google Scholar
Google Scholar
[43] Alberti A, Deretzis I, Pellegrino G, Bongiorno C, Smecca E, Mannino G, Giannazzo F, Condorelli G G, Sakai N, Miyasaka T, Spinella C, La Magna A 2015 Chem. Phys. Chem. 16 3064
 Google Scholar
Google Scholar
[44] Merdasa A, Bag M, Tian Y, Källman E, Dobrovolsky A, Scheblykin I G 2016 J. Phys. Chem. C 120 10711
 Google Scholar
Google Scholar
[45] Slotcavage D J, Karunadasa H I, McGehee M D 2016 ACS Energy Lett. 1 1199
 Google Scholar
Google Scholar
[46] Yang X, Yan X L, Wang W, Zhu X X, Li H, Ma W L, Sheng C X 2016 Org. Electron. 34 79
 Google Scholar
Google Scholar
[47] Hoke E T, Slotcavage D J, Dohner E R, Bowring A R, Karunadasa H I, McGehee M D 2015 Chem. Sci. 6 613
 Google Scholar
Google Scholar
[48] Yoon S J, Kuno M, Kamat P V 2017 ACS Energy Lett. 2 1507
 Google Scholar
Google Scholar
[49] Chen S, Wen X M, Huang S J, Huang F Z, Cheng Y B, Green M, Ho-Baillie A 2017 Solar RRL 1 Unsp 1600001
 Google Scholar
Google Scholar
[50] Mosconi E, Meggiolaro D, Snaith H J, Stranks S D, de Angelis F 2016 Energy Environ. Sci. 9 3180
 Google Scholar
Google Scholar
[51] Divitini G, Cacovich S, Matteocci F, Cinà L, Di Carlo A, Ducati C 2016 Nature Energy 1 15012
 Google Scholar
Google Scholar
[52] Kim N K, Min Y H, Noh S, Cho E, Jeong G, Joo M, Ahn S W, Lee J S, Kim S, Ihm K, Ahn H, Kang Y, Lee H S, Kim D 2017 Sci. Rep. 7 4645
 Google Scholar
Google Scholar
[53] Conings B, Drijkoningen J, Gauquelin N, Babayigit A, D'Haen J, D'Olieslaeger L, Ethirajan A, Verbeeck J, Manca J, Mosconi E, de Angelis F, Boyen H G 2015 Adv. Energy Mater. 5 1500477
 Google Scholar
Google Scholar
[54] Chen W, Wu Y, Yue Y, Liu J, Zhang W, Yang X, Chen H, Bi E, Ashraful I, Grätzel M, Han L 2015 Science 350 944
 Google Scholar
Google Scholar
[55] Kaltenbrunner M, Adam G, Głowacki E D, Drack M, Schwödiauer R, Leonat L, Apaydin D H, Groiss H, Scharber M C, White M S, Sariciftci N S, Bauer S 2015 Nat. Mater. 14 1032
 Google Scholar
Google Scholar
[56] You J, Meng L, Song T B, Guo T F, Yang Y, Chang W H, Hong Z, Chen H, Zhou H, Chen Q, Liu Y, De Marco N, Yang Y 2016 Nat. Nanotechnol. 11 75
[57] deQuilettes D W, Zhang W, Burlakov V M, Graham D J, Leijtens T, Osherov A, Bulović V, Snaith H J, Ginger D S, Stranks S D 2016 Nat. Commun. 7 11683
 Google Scholar
Google Scholar
[58] Gottesman R, Gouda L, Kalanoor B S, Haltzi E, Tirosh S, Rosh-Hodesh E, Tischler Y, Zaban A, Quarti C, Mosconi E, de Angelis F 2015 J. Phys. Chem. Lett. 6 2332
 Google Scholar
Google Scholar
[59] Domanski K, Roose B, Matsui T, Saliba M, Turren-Cruz S H, Correa-Baena J P, Roldan-Carmona C, Richardson G, Foster J M, de Angelis F, Ball J M, Petrozza A, Mine N, Nazeeruddin M K, Tress W, Gratzel M, Steiner U, Hagfeldt A, Abate A 2017 Energy Environ. Sci. 10 604
 Google Scholar
Google Scholar
[60] Zhou Y, You L, Wang S, Ku Z, Fan H, Schmidt D, Rusydi A, Chang L, Wang L, Ren P, Chen L, Yuan G, Chen L, Wang J 2016 Nat. Commun. 7 11193
 Google Scholar
Google Scholar
[61] Brivio F, Caetano C, Walsh A 2016 J. Phys. Chem. Lett. 7 1083
 Google Scholar
Google Scholar
[62] Zhao C, Chen B B, Qiao X F, Luan L, Lu K, Hu B 2015 Adv. Energy Mater. 5 1500279
 Google Scholar
Google Scholar
[63] Huang F, Jiang L, Pascoe A R, Yan Y, Bach U, Spiccia L, Cheng Y B 2016 Nano Energy 27 509
 Google Scholar
Google Scholar
[64] Deschler F, Price M, Pathak S, Klintberg L E, Jarausch D D, Higler R, Hüttner S, Leijtens T, Stranks S D, Snaith H J, Atatüre M, Phillips R T, Friend R H 2014 J. Phys. Chem. Lett. 5 1421
 Google Scholar
Google Scholar
[65] Tian Y, Merdasa A, Unger E, Abdellah M, Zheng K, McKibbin S, Mikkelsen A, Pullerits T, Yartsev A, Sundström V, Scheblykin I G 2015 J. Phys. Chem. Lett. 6 4171
 Google Scholar
Google Scholar
[66] Tian Y X, Peter M, Unger E, Abdellah M, Zheng K, Pullerits T, Yartsev A, Sundstrom V, Scheblykin I G 2015 Phys. Chem. Chem. Phys. 17 24978
 Google Scholar
Google Scholar
[67] Brenes R, Guo D, Osherov A, Noel N K, Eames C, Hutter E M, Pathak S K, Niroui F, Friend R H, Islam M S, Snaith H J, Bulović V, Savenije T J, Stranks S D 2017 Joule 1 155
 Google Scholar
Google Scholar
[68] Buin A, Pietsch P, Xu J, Voznyy O, Ip A H, Comin R, Sargent E H 2014 Nano Lett. 14 6281
 Google Scholar
Google Scholar
[69] Yin W J, Shi T, Yan Y 2014 Appl. Phys. Lett. 104 063903
 Google Scholar
Google Scholar
[70] Buin A, Comin R, Xu J, Ip A H, Sargent E H 2015 Chem. Mater. 27 4405
 Google Scholar
Google Scholar
[71] Mosconi E, de Angelis F 2016 ACS Energy Lett. 1 182
 Google Scholar
Google Scholar
[72] Meloni S, Moehl T, Tress W, Franckevičius M, Saliba M, Lee Y H, Gao P, Nazeeruddin M K, Zakeeruddin S M, Rothlisberger U, Graetzel M 2016 Nat. Commun. 7 10334
 Google Scholar
Google Scholar
[73] Haruyama J, Sodeyama K, Han L, Tateyama Y 2015 J. Am. Chem. Soc. 137 10048
 Google Scholar
Google Scholar
[74] Huang F, Dkhissi Y, Huang W, Xiao M, Benesperi I, Rubanov S, Zhu Y, Lin X, Jiang L, Zhou Y, Gray-Weale A, Etheridge J, McNeill C R, Caruso R A, Bach U, Spiccia L, Cheng Y B 2014 Nano Energy 10 10
 Google Scholar
Google Scholar
[75] Wen X M, Feng Y, Huang S J, Huang F Z, Cheng Y B, Green M, Ho-Baillie A 2016 J. Mater. Chem. C 4 793
 Google Scholar
Google Scholar
[76] Nie W, Blancon J C, Neukirch A J, Appavoo K, Tsai H, Chhowalla M, Alam M A, Sfeir M Y, Katan C, Even J, Tretiak S, Crochet J J, Gupta G, Mohite A D 2016 Nat. Commun. 7 11574
 Google Scholar
Google Scholar
[77] Gottesman R, Haltzi E, Gouda L, Tirosh S, Bouhadana Y, Zaban A, Mosconi E, de Angelis F 2014 J. Phys. Chem. Lett. 5 2662
 Google Scholar
Google Scholar
[78] Azpiroz J M, Mosconi E, Bisquert J, de Angelis F 2015 Energy Environ. Sci. 8 2118
 Google Scholar
Google Scholar
[79] Eames C, Frost J M, Barnes P R F, O’Regan B C, Walsh A, Islam M S 2015 Nat. Commun. 6 7497
 Google Scholar
Google Scholar
[80] Walsh A, Scanlon D O, Chen S Y, Gong X G, Wei S H 2015 Angew. Chem. Int. Ed. 54 1791
 Google Scholar
Google Scholar
[81] Zhang H M, Liang C J, Zhao Y, Sun M J, Liu H, Liang J J, Li D, Zhang F J, He Z Q 2015 Phys. Chem. Chem. Phys. 17 9613
 Google Scholar
Google Scholar
[82] Zu F S, Amsalem P, Salzmann I, Wang R B, Ralaiarisoa M, Kowarik S, Duhm S, Koch N 2017 Adv. Opt. Mater. 5 Unsp 1700139
 Google Scholar
Google Scholar
[83] Noh J H, Im S H, Heo J H, Mandal T N, Seok S I 2013 Nano Lett. 13 1764
 Google Scholar
Google Scholar
[84] Aharon S, Cohen B E, Etgar L 2014 J. Phys. Chem. C 118 17160
 Google Scholar
Google Scholar
[85] Gil-Escrig L, Miquel-Sempere A, Sessolo M, Bolink H J 2015 J. Phys. Chem. Lett. 6 3743
 Google Scholar
Google Scholar
[86] Bischak C G, Hetherington C L, Wu H, Aloni S, Ogletree D F, Limmer D T, Ginsberg N S 2017 Nano Lett. 17 1028
 Google Scholar
Google Scholar
[87] Barker A J, Sadhanala A, deschler F, Gandini M, Senanayak S P, Pearce P M, Mosconi E, Pearson A J, Wu Y, Srimath Kandada A R, Leijtens T, De Angelis F, Dutton S E, Petrozza A, Friend R H 2017 ACS Energy Lett. 2 1416
 Google Scholar
Google Scholar
[88] Draguta S, Sharia O, Yoon S J, Brennan M C, Morozov Y V, Manser J S, Kamat P V, Schneider W F, Kuno M 2017 Nat. Commun. 8 200
 Google Scholar
Google Scholar
[89] Beal R E, Slotcavage D J, Leijtens T, Bowring A R, Belisle R A, Nguyen W H, Burkhard G F, Hoke E T, McGehee M D 2016 J. Phys. Chem. Lett. 7 746
 Google Scholar
Google Scholar
[90] McMeekin D P, Sadoughi G, Rehman W, Eperon G E, Saliba M, Hörantner M T, Haghighirad A, Sakai N, Korte L, Rech B, Johnston M B, Herz L M, Snaith H J 2016 Science 351 151
 Google Scholar
Google Scholar
[91] Jaffe A, Lin Y, Beavers C M, Voss J, Mao W L, Karunadasa H I 2016 ACS Cent. Sci. 2 201
 Google Scholar
Google Scholar
[92] Hu M, Bi C, Yuan Y B, Bai Y, Huang J S 2016 Adv. Sci. 3 1500301
 Google Scholar
Google Scholar
[93] Wei T C, Wang H P, Li T Y, Lin C H, Hsieh Y H, Chu Y H, He J H 2017 Adv. Mater. 29 Unsp 1701789
 Google Scholar
Google Scholar
[94] Kirschner M S, Diroll B T, Guo P, Harvey S M, Helweh W, Flanders N C, Brumberg A, Watkins N E, Leonard A A, Evans A M, Wasielewski M R, Dichtel W R, Zhang X, Chen L X, Schaller R D 2019 Nat. Commun. 10 504
 Google Scholar
Google Scholar
[95] Cottingham P, Brutchey R L 2018 Chem. Mater. 30 6711
 Google Scholar
Google Scholar
[96] Xue J, Yang D, Cai B, Xu X, Wang J, Ma H, Yu X, Yuan G, Zou Y, Song J, Zeng H 2019 Adv. Funct. Mater. 29 1807922
[97] Yettapu G R, Talukdar D, Sarkar S, Swarnkar A, Nag A, Ghosh P, Mandal P 2016 Nano Lett. 16 4838
 Google Scholar
Google Scholar
[98] He Y, Matei L, Jung H J, McCall K M, Chen M, Stoumpos C C, Liu Z, Peters J A, Chung D Y, Wessels B W, Wasielewski M R, Dravid V P, Burger A, Kanatzidis M G 2018 Nat. Commun. 9 1609
 Google Scholar
Google Scholar
[99] Meng Y, Ahmadi M, Wu X, Xu T, Xu L, Xiong Z, Chen P 2019 Org. Electron. 64 47
 Google Scholar
Google Scholar
[100] Zhang Y, Sun H, Zhang S, Li S, Wang X, Zhang X, Liu T, Guo Z 2019 Opt. Mater. 89 563
 Google Scholar
Google Scholar
[101] Huang M Y, Veeramuthu L, Kuo C C, Liao Y C, Jiang D H, Liang F C, Yan Z L, Borsali R, Chueh C C 2019 Org. Electron. 67 294
 Google Scholar
Google Scholar
[102] Tan Z, Wu Y, Hong H, Yin J, Zhang J, Lin L, Wang M, Sun X, Sun L, Huang Y, Liu K, Liu Z, Peng H 2016 J. Am. Chem. Soc. 138 16612
 Google Scholar
Google Scholar
[103] Chen S, Shi G 2017 Adv. Mater. 29 1605448
 Google Scholar
Google Scholar
计量
- 文章访问数: 24534
- PDF下载量: 660
- 被引次数: 0













 下载:
下载: